Research Article 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Anti-oxidant and Anti-free Radical Activities of Natural Oil Blend Formulation: Implication for Immune Regulation and Potential Adjuvant Treatment in Infectious Diseases
*Corresponding author: Haig Babikian, Department of Pharmaceutical Sciences, Pharmaceuticals Scineses Pte. Ltd, Gedung Pri Matari 2, Indonesia
Received: August 12, 2020; Published: September 07, 2020
DOI: 10.34297/AJBSR.2020.10.001491
Highlights
- The obtained result has great implication for immune regulation and adjuvant treatment in infectious diseases.
- Natural Oil Blend is a combination of the following oils Gardenia jasminoides, Commiphora myrrhea, Boswellia serrata, Foeniculum vulgarae, and Daucus carrota with the purpose of viral inactivating properties.
- Using quantitative flow cytometry, the influence of Natural Oil Blend on neutrophils phagocytosis and their respiratory burst induction were studied.
- Natural Oil Blend enhances human neutrophils phagocytosis but dramatically decreases chemotactic peptide N-formyl-Met-Leu-Phe (fMLP)-, phorbol 12-myristate 13-acetate (PMA)-, and E. coli-induced oxidative burst.
- Natural Oil Blend like alfa-tocopherol was able to promote the scavenging of free radicals, displaying a high dose–response representing a significant anti-oxidant activity.
- Natural Oil Blend may serve as a potent pharmaceutical agent with combined anti-inflammatory, antimicrobial and anti-oxidant activity capable restoring impaired intracellular production of ROS and down-modulation of overwhelming inflammatory response of innate immune cells to chemotaxis- and phagocytosis-mediated respiratory burst induction.
Abstract
In the present work, the human peripheral blood polymorphonuclear neutrophils phagocytosis, neutrophils oxidative burst regulation, and anti-free radical activity of Natural Oil Blend (NOB) contained Gardenia jasminoides, Commiphora myrrha, Boswellia serrata, Foeniculum vulgarea and Daucus carota essential oils evaluated. Using quantitative flow cytometry in this study we investigated the influence of NOB on neutrophils phagocytosis, and also on their respiratory burst induction. We demonstrated that NOB enhances human neutrophils phagocytosis but dramatically decreases chemotactic peptide N-formyl-Met-Leu-Phe (fMLP)-, phorbol 12-myristate 13-acetate (PMA)-, and E. coli-induced oxidative burst. Furthermore, the antioxidant activity of NOB confirmed by in vitro studies the anti-free radical scavenging activity.
We observed that OB like alfa-tocopherol was able to promote the scavenging of free radicals, displaying a high dose-response representing a significant anti-oxidant activity. Therefore, our results suggest that NOB may serve as a potent pharmaceutical agent with combined antiinflammatory, antimicrobial and anti-oxidant activity capable restoring impaired intracellular production of ROS and down-modulation of the overwhelming inflammatory response of innate immune cells to chemotaxis- and phagocytosis-mediated respiratory burst induction. This result has great implications for immune regulation and adjuvant treatment in infectious diseases.
Introduction
Natural oils are a mixture of volatile compounds (mainly mono- and sesquiterpenoids, benzenoids, amino acid- derivatives, phenylpropanoids, etc.). Natural oils also obtain a wide range of pharmacological actions, e.g. antimicrobial, antifungal, anti-inflammatory, anti-oxidant, hyperemic, anti-nociceptive, spasmolytic, diuretic, choleretic, and carminative effects [1,2]. For centuries, natural oils used for combating infection in various cultures. Nowadays, the use of natural antimicrobial agents is noticeably growing for a wide variety of purposes including human and animal medication, food industry, and environmental protection [3].
Previously, we demonstrated that the natural oil blend formulation (NOBF) which prepared from a mixture of Gardenia jasminoides, Commiphora myrrha, Boswellia serrata, Foeniculum vulgarea and Daucus carota, as well as other combination of natural oils [4], act as a potent antimicrobial and antifungal agent. The natural oil blend formulation (NOBF) inhibited the growth of several tested microorganisms, including gram-positive Staphylococcus aureus, Staphylococcus epidermidis, Micrococcus luteus, gram-negative Pseudomonas aeruginosa, as well as the strains of fungi Candida albicans. The most prominent antimicrobial activity of NOBF observed against Staphylococcus epidermidis and Pseudomonas aeruginosa. Besides, NOBF demonstrated a prominent anti-inflammatory effect as reflected by the suppression of IL-1β, TNFα, IL-6, and IFNγ production in a dose-dependent manner by LPS-stimulated human mononuclear cells.
The phagocytic cells of the innate immune system such as neutrophils and macrophages are key components of antimicrobial host defense, which is because these cells are capable of generating large amounts of highly toxic molecules - reactive oxygen species (ROS) [5,6]. When phagocytes are activated, the production of ROS is initiated by NADPH oxidase because of the translocation of several cytosolic proteins (gp67phox, gp40phox, gp47phox, and the Rho-family GTPase, Rac2) to the membrane-bound complex carrying cytochrome b558 (gp91phox-gp22phox-Rap1a). The primary product of the reaction catalyzed by NADPH oxidase is superoxide (O2-), which can be converted to H2O2 by superoxide dismutase, to hydroxyl radicals (OH) and hydroxyl anion (OH-). ROS generated by phagocytes can damage DNA and several chemical moieties and are of central importance in host defense and inflammation [7,8].
Oxygen radicals generated in excess in a diverse array of microbial and viral infections. Emerging concepts in free radical biology are now shedding light on the pathogenesis of various diseases. Reactive oxygen species generated by infiltrating phagocytic cells and xanthine oxidase expressed in inflamed tissues and they believed to contribute to nonspecific (innate) and immunological host defense as well [9]. Free radical-induced pathogenicity in virus infections is of great importance, because evidence suggests that oxygen radicals such as superoxide are key molecules in the pathogenesis of various infectious diseases including severe acute respiratory syndrome coronavirus (SARS-CoV) induced acute respiratory distress syndrome (ARDS) [10].
Therefore, the NOBF -induced production of ROS upon phagocytic activation of effector cells and oxidative burst may be natural for killing several micro-organisms, as shown by the resistance of NOBF -treated animals to bacterial and viral infection. Using quantitative flow cytometry in this study we investigated the influence of NOBF on human peripheral blood polymorphonuclear neutrophils (PMN) phagocytosis, and also on their respiratory burst induction. We demonstrated that NOBF enhances human PMN phagocytosis but dramatically decreases chemotactic peptide N-formyl-Met-Leu-Phe (fMLP)-, phorbol 12-myristate 13-acetate (PMA)-, and E. coli-induced oxidative burst. Furthermore, the anti-oxidant activity of NOBF confirmed by in vitro studies the anti-free radical scavenging activity.
Materials and Methods
Natural Oil Blend formulation (NOBF) preparation and composition
Each single oil Gardenia jasminoides, Commiphora myrrha, Boswellia serrata, Foeniculum vulgarea and Daucus carota obtained from the vendors who comply with the strictest industry practices: Demeter Agro Research and Improvements Pty Ltd, New Directions Australia Pty Ltd and Australian Botanical Products Pty Ltd. Each natural oil is obtained through the steam distillation process and should undergo thorough checking for the quality and chemical compositions based on European Pharmacopeia. After the natural oils are declared to pass the quality checking, the mixture of the NOs conducted with the following sequence and percentage: Gardenia jasminoides, Commiphora myrrha, Boswellia serrata, Foeniculum vulgarea and Daucus carota are added in equal quantities to form the oil mixture they mixed with extra light olive oil. Hence, we get 1% concentration of the final product.
Peripheral blood sampling
After signed informed consent, 30 ml of venous blood samples obtained from 6 healthy subjects, 30 ml of venous blood samples 3 men (mean age of 25 ± 3.1 years) and another 30 ml from 3 women (mean age of 26 ± 3.4 years). The Ethical Committee approved the experiment of the Institute of Molecular Biology of the NAS RA (IRB IORG0003427). Healthy volunteers selection was conducted based on reviews of medical history with the following exclusion criteria 1) studied subjects were not taking any medication at the time of the experiments; 2) none of the individuals suffered from acute/chronic diseases or abnormalities; 3) smoking, consumption of alcohol, drugs and current or past use of any active substance; 4) pregnancy or lactating mothers.
Cytotoxicity of NOBF on human whole blood cells
Assessment of Natural oil blend formulation (NOBF) cytotoxicity performed with trypan blue dye exclusion and flow cytometry. Both techniques used to determine the number of viable cells present in a whole blood samples after 0 -2 h incubation with NOBF and PMA. Cytotoxicity <30% was accepted as a criterion for valid positive result in this assay.
Neutrophil Phagocytosis
Assessment of natural oil blend formulation (NOBF) cytotoxicity performed with trypan blue dye exclusion and flow cytometry. Both techniques used to determine the number of viable cells present in whole blood samples after 0 -2 h incubation with NOBF and PMA. Cytotoxicity <30% was accepted as a criterion for a valid positive result in this assay. Whole blood samples were incubated with 6×10-3 and 6×10-4 µg/µl NOBF or vehicle and incubated in a horizontal shaking water bath at 37oC for 1-10 min and phagocytosis assay performed using dextran-FITC for engulfment. Dextran-FITC (5 mg, Sigma Chemical. St. Louis, Mo.) added to 100 ml of whole blood and the samples were normalized with the absolute content of neutrophils and incubated in a horizontal shaking water bath at 37 oC for 10 min [11]. Control samples left on the ice. After erythrocyte removal by lysis solution to avoid artifacts occurrence, upon aggregated cells determination, phagocyte DNA was stained with propidium iodide. Phagocytosis monitored by determining the percentage of fluorescing cells on a FACSCaliburTM instrument using CellQuestTM software (Becton Dickinson).
Polymorphonuclear neutrophils respiratory burst
The effect of NOBF on the respiratory burst of human PMN studied by quantitative flow cytometry determination of dihydroergotamine-123 (DHR123) fluorescence intensity that reacts with ROS (superoxide anion, hydrogen peroxide, and hypochlorous acid) in the presence or absence of respiratory burst stimulators; fMLP, PMA, and opsonized E.coli [7,8]. The assay performed with the Bursttest-kit (PHAGOBURST™). 20 ng/ml PMA or 100 ng/ml fMLP or 2 × 107 opsonized E.coli and 6×10-3 and 6×10-4 µg/µl NOBF were added to 100 μl of whole blood and incubated at 37°C for 10 min. DHR123 solution added and the tubes incubated for an additional 10 min at 37°C. After erythrocyte removal by lysing solution the DNA of the remaining cells was stained with propidium iodide and immediately subjected to flow cytometry analysis (10,000 events). The respiratory burst monitored by determining the percentage of fluorescing cells on a FACSCaliburTM instrument using CellQuestTM software (Becton Dickinson).
Antioxidant assay
The antioxidant activity of the NOBF was evaluated by DPPH radical scavenging assay using methodologies previously described [12]. A stock solution of DPPH reagent (0.5 mM) prepared in methanol. The solution was diluted with methanol (60 μM approx.) to yield an initial absorbance of 0.62 ± 0.02 at 517 nm. The reaction mixture was composed of 1950 μl of DPPH solution and 50 μl of the oil, diluted with different proportions of methanol. For each sample, a methanol blank also used. we used Alfa-tocopherol as a standard antioxidant. The absorbance measured after 10 and 60 min for the samples and standard, respectively. The DPPH-inhibition percentage calculated the radical-scavenging activity according to the equation: I% = 100 (A-B)/A, where A and B are the blank and sample absorbance values, at the end of the reaction. Linear regression determined the concentration of antioxidant required for 50 % scavenging of DPPH radicals (half-maximal effective concentration or EC50). The analysis performed in triplicate and the result presented as mean value ± standard deviation.
Statistics
Statistical analyses performed using the statistical software Graph Pad Prism 5.01 (Graph Pad Software, USA). Data compared using the One-way Repeated Measure ANOVA test. Normal distribution was checked visually from distributions and with Shapiro-Wilk's W test. P values ≤ 0.05 considered as significant. Results expressed as the mean and standard error of the mean.
Results
The effect of natural oil blend formulation (NOBF) on PMN phagocytosis
First, we tested the cytotoxicity of different concentrations of NOBF on human whole blood cells by using both trypan blue dye exclusion and flow cytometry. natural oil blend formulation (NOBF) showed cytotoxic effects at the highest 6×10-2 µg/µl concentration and the absence of toxic effect of NOBF at 6×10-3 - 6×10-5 µg/µl concentrations. Next, we investigated the influence of NOBF on human peripheral blood PMN phagocytosis using quantitative flow cytometry. The obtained results of the flow cytometry study of PMN phagocytos is provided in Figure 1. We observed that NOBF significantly increased (p<0.05) the PMN phagocytic function (i.e., percentage of cells able to undergo phagocytosis) comparing with the untreated control in the presence of both 6×10-3 and 6×10-4 µg/µl concentrations [Figure 1]. We did not observe significant differences between 6×10-3 and 6×10-4 µg/µl concentrations of NOBF on PMN phagocytic function in the whole blood model. Therefore, NOBF increased PMN phagocytic function which could be the innate immune basic mechanism underling antimicrobial activity of NOBF previously observed in vivo animal models [13-15].
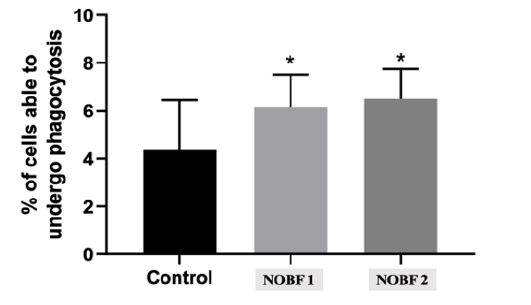
Figure 1: NOBF effect on normal donors whole blood PMN phagocytosis. Results are the means SD. NOBF 1 - 6×10-3 μg/μl, NOBF 2 - (6×10-4 μg/μl concentrations. *p<0.05, comparing with untreated control.
The effect of natural oil blend formulation (NOBF) on the respiratory burst of PMN.
Although PMN cell population exerts antimicrobial activity against extracellular bacteria, not only through phagocytic function, there is also more antimicrobial activity through the utilization of ROS production for the control of intracellular microbes. The results of the flow cytometric determination of PMN respiratory burst, using the fluorescence probe DHR123, which is highly specific to superoxide anion, hydrogen peroxide, and hypochlorous acid [7,8], are presented in Table 1. According to the presented data, the NOBF in both used concentrations didn't influence the number of PMN undergoing respiratory burst in the absence of ROS stimulators. However, we observed that NOBF dramatically decreases chemotactic peptide N-formyl-Met-Leu-Phe (fMLP)-, phorbol 12-myristate 13-acetate (PMA)-, and E. coli-induced oxidative burst comparing to the untreated whole blood control (p<0.05). Again, we did not observe significant differences between 6×10-3 and 6×10-4 µg/µl concentrations of NOBF on PMN respiratory burst in the whole blood model. Together, these results suggest that NOBF possesses a pronounced ability to down-regulate PMN oxidative burst induced by strong pro-inflammatory stimulators like fMLP, PMA, and bacterial cells.
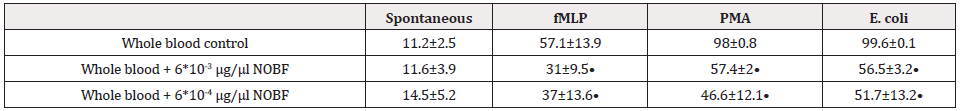
Table 1: Oxidative burst induction of whole blood PMN and Natural oil blend formulation (NOBF) effects.
Results are the means ± SD. • -pt ≤ 0.05 versus non-NOBF -treated control.
Antioxidant activity
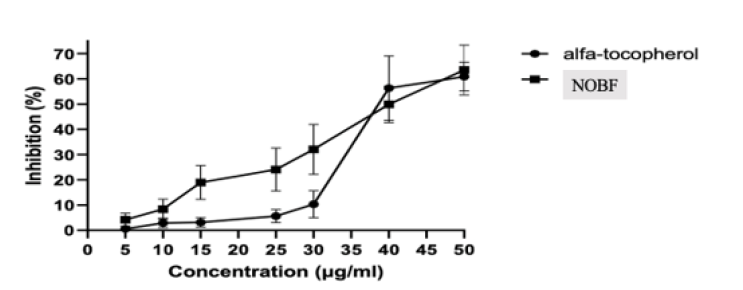
In the present work, the in vitro antioxidant activity of the NOBF evaluated by DPPH radical scavenging. The oil interacted with the DPPH by transfer of electron or hydrogen, neutralizing its free radical character [12]. The oil was able to promote the scavenging of DPPH radical, displaying a high dose-response (r2 = 0.96), with inhibition varying from 5 to 65 %, at concentrations from 5 to 50μg/ml. The half-maximal effective concentration (EC50) was 40.0 ± 3.5 μg/ml, calculated by linear regression (p < 0.05), representing a significant antioxidant activity and comparable to the standard alfa-tocopherol (38.8 ± 2.9 μg/ml), as can be seen in Figure 2.
Discussion
Natural oil blend formulation (NOBF) inhibited the growth of several tested microorganisms in vitro, including gram-positive Staphylococcus aureus, Staphylococcus epidermidis, Micrococcus luteus, gram-negative Pseudomonas aeruginosa, as well as the strains of fungi Candida albicans [4]. The most prominent antimicrobial activity of NOBF observed against Staphylococcus epidermidis and Pseudomonas aeruginosa.
While inflammatory response required for effective host defense during infections, exaggerated or uncontrolled inflammation is the main cause of healthy tissue destruction and chronic pathologies. Natural Oil Blend formulation (NOBF) was designed not solely as a formulation with an antimicrobial and antifungal potency but also as an anti-inflammatory agent, capable to limit pathogen-induced inflammation [4]. Natural oil blend formulation (NOBF) demonstrated a prominent anti-inflammatory effect as reflected by the suppression of IL-1β, TNFα, IL-6, and IFNγ production in a dose-dependent manner by LPS-stimulated human mononuclear cells. Production of anti-inflammatory IL-10 by LPS-primed cells was also suppressed by NOBF in a dose-dependent manner [4].
The ROS and reactive nitrogen species (RNS) as central components of the "respiratory burst" in activated leukocytes, play an natural role in the host immune defenses against pathogens [13,14]. However, based on their nonspecific and highly reactive nature, products of redox metabolism (e.g., O2⋅-, OH⋅-, ONOO⋅-, HClO, H2O2, and NO) may be potentially harmful to host cells [12, 20]. Apart from virulence factors expressed by pathogenic bacteria and viruses, the excessive production of ROS and RNS by activated immune cells creates a highly cytotoxic milieu that contributes to the direct damage of target organs in the context of unbalanced inflammatory responses [14-17]. Therefore, the modulation of the host's redox system as an antimicrobial therapeutic strategy is challenging. The up regulation of ROS and RNS production may increase cytotoxicity and organ damage, while the downregulation of these molecules may favor the survival and spread of pathogenic microorganisms, leading to opposite scenarios with a similar endpoint, including higher mortality of the host [16,17].
Here we demonstrated that NOBF up-regulated human PMN phagocytic function but dramatically down-regulated fMLP- and PMA- as well as bacteria (opsonized E. coli)-induced, an oxidative burst of neutrophils. Increased level of ROS production in PMN during inflammatory conditions characterized by the impairment of PMA (protein kinase C)-dependent regulation of ROS production and also the over-regulation of chemotaxis/inflammation-mediated respiratory burst induction as well as phagocytosis-dependent activation of ROS production. In the present study we show that NOBF is a differentially regulated oxidative burst induction in PMN. In this study we demonstrated that NOBF does not affect human PMN spontaneous oxidative burst but dramatically decreases fMLP- and PMA-induced, an oxidative burst of cells [Figure 1 & Table 1]. Accordingly, NOBF could serve as a potent antioxidant capable of restoring impaired intracellular production of ROS in PMN and down-modulation of the overwhelming inflammatory response of PMN to chemotaxis- and phagocytosis-mediated respiratory burst induction.
The cells of living organisms generate free radicals as a result of pathophysiological and biochemical processes in response to factors such as, radiation, chemicals, toxins, and bacterial and viral infection. The NOBF creates an imbalance in the formation and neutralization of prooxidants that subsequently seek stability through electron pairing with biological macromolecules such as proteins, lipids, and DNA, leading to oxidative stress in the physiological system [18]. Furthermore, these effects lead to lipid peroxidation as well as protein or DNA damage in human cells, which consequently lead to aging and several chronic diseases such as cancer, diabetes, and atherosclerosis as well as cardiovascular, inflammatory, and other degenerative diseases in humans [18,19]. The ability of certain phytochemical extracts to inhibit or delay the oxidation of other molecules by suppressing the initiation or propagation of oxidizing chain reactions have made them active alternatives in complementary medicine [19-22]. These naturally occurring antioxidants have reported to exhibit antioxidant and anticancer activities [21,22].
In the present study, the in vitro antioxidant activity of the Therefore, the NOBF -induced production of ROS upon phagocytic activation of effector cells and oxidative burst may be essential for killing several micro-organisms, as shown by the resistance of NOBF -treated animals to bacterial and viral infection. Using quantitative flow cytometry in this study we investigated the influence of NOBF on human peripheral blood polymorphonuclear neutrophils (PMN) phagocytosis, and also on their respiratory burst induction. We demonstrated that NOBF enhances human PMN phagocytosis but dramatically decreases chemotactic peptide N-formyl-Met-Leu-Phe (fMLP)-, phorbol 12-myristate 13-acetate (PMA)-, and E. coli-induced oxidative burst. Furthermore, the anti-oxidant activity of NOBF confirmed by in vitro studies the anti-free radical scavenging activity. It was evaluated by DPPH radical scavenging [Figure 2]. The NOBF like alfa-tocopherol was able to promote the scavenging of DPPH radical, displaying a high dose-response representing a significant antioxidant activity.
Antioxidant systems seem to play a crucial role in maintaining the morphological and functional integrity of all microorganisms and viral particles. Accordingly, disruptors of redox balance (e.g., inductors of oxidative stress or inhibitors of antioxidant molecules) proposed as candidates to new antimicrobial drugs [23]. Several drugs developed for different conditions (e.g., anti-inflammatory, antineoplastic, antidepressant, anxiolytic, and antipsychotic) also exhibit antimicrobial properties [23]. In many cases, these properties based on the disruption of microbial redox metabolism, a pharmacological effect that opens new avenues for drug repurposing and development of new strategies for the treatment of infectious diseases [24].
Cytokine storm is an excessive immune response to external stimuli. The pathogenesis of the cytokine storm is complex. The disease progresses rapidly, and mortality is high. Certain evidence shows that, during the coronavirus disease 2019 (COVID-19) epidemic, the severe deterioration of some patients has closely related to the cytokine storm in their bodies [24,25]. The pathogens then mediate the recruitment of immune cells, which eliminates the pathogens and ultimately leads to tissue repair and restoration of homeostasis. However, SARS-CoV-2 induces excessive and prolonged cytokine/chemokine responses in some infected individuals, known as the cytokine storm [26]. Cytokine storm causes ARDS or multiple-organ dysfunction, which leads to physiological deterioration and death. Timely control of the cytokine storm in its early stage through such means as immunomodulators and cytokine antagonists, as well as the reduction of lung inflammatory cell infiltration caused by ROS, would facilitate in improving the treatment success rate of patients with infectious diseases.
Conclusion
Natural oils used for our blend thought to have a strong biological potency associated with a suppression of unwanted inflammation and bacterial and viral infection. Disruption of the host excessive production of ROS and redox metabolism pronounced antioxidant activity and the ability to increase a key component of antimicrobial host defense.
The phagocytic activity of the innate immune system pronounced such as neutrophils, a pharmacological effect that opens new venues for Natural oil blend formulation ( N O B F ) development of new strategies for the adjuvant treatment of infectious diseases. Therefore, it has anticipated that overlapping action of different natural compounds will amplify antimicrobial potency, antioxidant activity and influence the magnitude of immune responses and restrict exaggerated production of ROS and cytokines, supporting the regulation of immune response to inflammatory stressors.
Acknowledgment
We would like to convey our gratitude to PT. Central Proteina Prima for the continuous support to our research. Special thanks to Laboratory of Molecular and Cellular Immunology, Institute of Molecular Biology, National Academy of Sciences, Yerevan 0014, Armenia and Faculty of Pharmacy, Universitas Padjadjaran (Padjajaran University), Indonesia for facilitating the laboratories.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Bakkali F, Averbeck S, Averbeck D, Idaomar M (2008) Biological effects of essential oils-a review. Food Chem Toxicol 46: 446-475.
- Edris AE (2007) Pharmaceutical and therapeutic potentials of essential oils and their individual volatile constituents: a review. Phytother Res 21(4): 308-323.
- Smith-Hall C, Larsen HO, Pouliot M (2012) People, plants and health: a conceptual framework for assessing changes in medicinal plant consumption. J Ethnobiol Ethnomed 8: Pp43.
- Babikian H (2019) Antibacterial and Anti-Inflammatory Properties of Natural Herbal Oil Formulation (NHOF). Jacobs J Exp Cardiol Res 6(1): Pp025.
- Bogdan Ch, Rollinghoff M, Diefenbach A (2000) Reactive oxygen and reactive nitrogen intermediates in innate and specific immunity. Curr Opin Immunol 12(1): 64-76.
- Hampton M B, Kettle A J, Winterbourn Ch (1998) Inside the neutrophil phagosome: Oxidants, myeloperoxidase, and bacterial killing. Blood 92(9): 3007-3017.
- Davtyan TK, Manukyan HM, Hakobyan GS (2005) Hypothalamic proline-rich polypeptide is a regulator of oxidative burst in human neutrophils and monocytes. Neuro Immuno Modulation 12: 270-284.
- Davtyan TK, Manukyan HM, Hakopyan GS, Mkrtchyan NR, Avetisyan SA, et al. (2005) Hypothalamic Proline-Rich Polypeptide is an Oxidative Burst Regulator. Neurochem res 30(3): 297-309.
- Takaaki Akaike (2001) Role of free radicals in viral pathogenesis and mutation. Rev Med Virol 11: 87-101.
- Chen H I, Kao S J, Wang D, Lee R P, Su Ch F, et al. (2003) Acute respiratory distress syndrome. Journal of Biomedical Science 10: 588-592.
- Davtyan T K, Avetisyan S A, Hakobyan G (2013) F-actin Dynamics in Familial Mediterranean Fever: The Unequal Effect of Colchicine on Activated Neutrophils. Antiinflamm Antiallergy Agents Med Chem 12(2): 165-72.
- Sarrazin SLF, da Silva LA, Oliveira RB (2105) 2 Antibacterial action against food-borne microorganisms and antioxidant activity of carvacrol-rich oil from Lippia origanoides Kunth. Lipids in Health and Disease. 14: Pp145.
- Davtyan TK, Mkrtchyan NR, Manukyan HM, Avetisyan S A, Galoyan A A, et al. (2006) Dexamethasone, colchicine and iodine-lithium-a-dextrin act differentially on the oxidative burst and endotoxin tolerance induction in vitro in patients with Behcet’s disease. International Immunopharmacology 6: 396-407.
- Pohanka M (2013) Role of oxidative stress in infectious diseases. A review. Folia Microbiol 58(6): 503-13.
- Sorci G, Faivre B (2009) “Inflammation and oxidative stress in vertebrate host-parasite systems,” Philosophical Transactions of the Royal Society B: Biological Sciences 364(1513): 71-83.
- Ivanov AV, Bartosch B, Isaguliants MG (2017) Oxidative stress in infection and consequent disease. Oxidative Medicine and Cellular Longevity.
- Novaes RD, Santos EC, Fialho CQ (2017) Nonsteroidal anti-inflammatory is more effective than anti-oxidant therapy in counteracting oxidative/nitrosative stress and heart disease in T. cruzi-infected mice. Parasitology 144(7): 904-916.
- Kehrer JP, Klotz LO (2015) Free radicals and related reactive species as mediators of tissue injury and disease: implications for Health. Crit Rev Toxicol 45(9): 765-798.
- Sharma GN, Gupta G, Sharma P (2018) A Comprehensive Review of Free Radicals, Antioxidants, and Their Relationship with Human Ailments. Crit Rev Eukaryot Gene Expr 28(2): 139-154.
- Al-Abd NM, Nor ZM, Mansor M, Azhar F (2015) Antioxidant, antibacterial activity, and phytochemical characterization of Melaleuca cajuputi extract. BMC Complementary and Alternative Medicine 15: Pp385.
- Nagmoti DM, Khatri DK, Juvekar PR, Juvekar AR (2012) Antioxidant activity free radical-scavenging potential of Pithecellobium dulcei Benth seed extracts. Free Rad Antiox 2(2):37-43.
- Novaes RD, Teixeira AL, de Miranda AS (2019) Oxidative Stress in Microbial Diseases: Pathogen, Host, and Therapeutics. Oxidative Medicine and Cellular Longevity.
- Beltran-Hortelano I, Perez-Silanes S, Galiano S (2017) Trypanothione reductase and superoxide dismutase as current drug targets for Trypanosoma cruzi: an overview of compounds with activity against Chagas disease. Current Medicinal Chemistry 24(11): 1066-1138.
- Qing Ye MD, Bili Wang MS, Jianhua Mao MD (2020) Cytokine Storm in COVID-19 and Treatment. J Infect 80(6): 607-613.
- Sin-Yee Fung, Kit-San Yuen, Zi-Wei Ye (2020) A tug-of-war between severe acute respiratory syndrome coronavirus 2 and host antiviral defense: lessons from other pathogenic viruses. Emerg Microbes Infect 9(1): 558-570.
- Chih-Cheng Lai, Tzu-Ping Shih, Wen-Chien Ko, Hung-Jen Tang, Po-Ren Hsueh, et al. (2020) Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and corona virus disease-2019 (COVID-19): the epidemic and the challenges. Int J Antimicrob Agents 55(3): Pp105924.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.