Research Article 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Microarray profiling of long noncoding RNAs and mRNA expression in the kidney of rats damaged by cadmium
*Corresponding author: Shaoxin Huang, School of Basic Medicine, Jiujiang University, Jiujiang, China.#These authors contributed equally
Received: August 27, 2020; Published: September 09, 2020
DOI: 10.34297/AJBSR.2020.10.001493
Abstract
Cadmium (Cd) is a toxic heavy metal that can induce nephrotoxicity with the unclear molecular mechanism. Microarray technology is a tool in the global analysis of gene expression, allowing for the simultaneous investigation of hundreds or thousands of genes in a sample. Long noncoding RNAs (lncRNAs) represent a novel class of noncoding RNAs that are involved in a variety of biological processes and human diseases. For the gene expression analysis, kidney tissues from both normal(fed with physiological saline) and cadmium treated rats(fed with 6.6mg/kg cadmium for 4 weeks) were collected. Total RNA was extracted and subjected to the CapitalBio Technology Rat LncRNA Array v1, KEGG, Gene Ontology (GO) analysis were applied to screen and analyze differentially regulated genes. This study identified 322 lncRNAs and 495 mRNAs that were differentially expressed after cadmium-induced kidney damage, indicating a potential role for lncRNAs and mRNAs. Enrichment analysis of differentially expressed mRNAs and the results could be enriched to inflammatory mediator regulation of TRP channels, which may be related to kidney damage caused by cadmium. Subsequently, the mRNA expression in this channel was verified by RT-qPCR. A lncRNA-mRNA-transcription factor co-expression network was constructed to relate lncRNAs to regulatory factors and pathways that may be important in the kidney by cadmium damage. This study analyzed differentially expressed genes in the kidney tissues of normal and cadmium damage by microarray analysis and bioinformatics, and to determine the potential molecular mechanisms of nephrotoxicity damaged by cadmium.
Keywords: LncRNA; Cadmiun; Nephrotoxicity; Mechanism
Abbreviations: Cd: Cadmium; lncRNAs: Long Noncoding RNAs; GO: Gene Ontology; Cd-MT: Cadmium-metallothionein; Nrf2: Nuclear Factor Erythroid 2-Related Factor 2; qRT-PCR: Quantitative Real-time PCR; dsDNA: Double-stranded Complementary DNA; KEGG: Kyoto Encyclopedia of Genes and Genomes; CNC: Coding-noncoding; TFs: Transcription Factors; ROS: Generate Reactive Oxygen Species
Introduction
Cadmium (Cd) is a naturally present and global environmental pollutant widely distributed into the surrounding environment [1- 3]. A wide spectrum of deleterious effects has been associated with prolonged cadmium exposure [4-7]. Kidney damage has long since been described to be the main problem for patients chronically exposed to cadmium. Meanwhile, the kidney is the most sensitive and affected organ [8]. Almost 30% of body cadmium absorbed is deposited in the kidney tubule region [9]. Cadmium reaches the kidney in the form of cadmium-metallothionein (Cd-MT) [1]. Cd- MT is filtrated in the glomerulus. and Cd-MT could fgsubsequently be reabsorbed in the proximal tubules. Then it could remain in the tubules cells [10]. The amount of cadmium in the kidney tubules cells increases during every person’s life span. Once the deposition of cadmium in the kidney reaches a certain level, it causes pathological changes in the renal tubular area and it could induce renal dysfunction often persists for many years [11].
The mechanism of chronic kidney damage is still unclear. Some research suggests that one of the causative mechanisms of chronic kidney disease is oxidative stress [12,13]. Cadmium induces oxidative stress, this nephron segment is sensitive to damage by oxidative stress [12,14,15]. Some research suggests that Cd induce kidney damage and dysfunction, along with autophagy, both in vivo and in vitro [16,17]. but the molecular mechanisms involved in the cell damage from oxidative stress in cadmium-induced chronic kidney disease are not well understood [18].
Long noncoding RNAs (lncRNAs) represent a novel class of noncoding RNAs that are involved in a variety of biological processes and human diseases. Recently, more and more evidence indicated that lncRNAs exerted critical roles in a series of biological processes, involved in the regulation of gene transcriptional, chromatin methylation, post-transcriptional levels, and another biological progression [19]. Previous research is recently revealed that long non-coding RNA (lncRNA) MT1DP induction under Cd stress decreases the nuclear factor erythroid 2-related factor 2 (Nrf2) level to evoke oxidative stress through the elevation of miR- 365, which acted to repress the Nrf2 level via direct binding to its 3’UTR, which may be involved in the mechanism of cadmiuminduced liver toxicity through oxidative stress [20]. lncRNAMALAT1 regulates cell proliferation, apoptosis, migration and invasion, and which may serve as a novel valuable biomarker of cadmium exposure and cadmium toxicity [21].
Reports suggest that the kidney is the most important target organ for cadmium-induced toxicity and carcinogenicity, and cadmium may lead to different mRNAs and LncRNAs expression profiles, which play important roles in cadmium nephrotoxicity [22,23]. However, few studies have addressed the expression and functional significance of differentially expressed LncRNA and mRNA in kidney damage induced by cadmium using gene microarray technology.
This study clarified the roles of differentially expressed mRNAs and lncRNAs in cadmium-damage kidney tissue. This is the first comprehensive study using microarray and bioinformatics to analyze the transcriptomic alterations of both LncRNAs and mRNAs in the kidney after cadmium-induced kidney damage. Enrichment analysis of differentially expressed mRNAs, some differentially expressed mRNAs can be enriched to inflammatory mediator regulation of TRP channels. By predicting the target genes of differentially expressed LncRNAs, it was found that the corresponding target gene mRNAs can also be enriched in inflammatory mediator regulation of TRP channels. This channel may be related to kidney damage caused by cadmium. Furthermore, the expressions of these RNAs are verified by quantitative realtime PCR (qRT-PCR). This study can help to clarify the molecular mechanism of cadmium-induced nephrotoxicity, which also may provide potential biomarkers and therapeutic targets for cadmium nephrotoxicity.
Materials and Methods
Ethics statement
The study's animal experimental protocols were approved by the Animal Care and Use Committee of Jiujiang University. All efforts were made to minimize the number used and their suffering. All rats were kept in a facility free of specific pathogens and had easy access to food and water. Animals were euthanized using the broken-neck method under deeply anesthetized conditions.
Animal protocol and histological examination
The animal protocol used for these studies is a well-established and well-characterized sub-chronic treatment protocol for producing Cd-induced nephrotoxicity in the rats. Sprague-Dawley rats weighing 100-150g (specific-pathogen-free rat, Hunan slake experimental animals co. LTD, China) were maintained in individual plastic cages on a 14/10 h light/dark cycle. Rats in the cadmium treatment group (n=3) received daily intragastric administration of CdCl2 at a dose of 6.6mg/kg in 1ml isotonic saline for up to 4 weeks,and named cadmium group. Control group animals (n=3) received daily injections of the saline vehicle alone and named the control group. The animals were allowed free access to water at all times. This treatment protocol was repeated three times and data were pooled whenever possible. At week 4, rats were put to death and their kidneys were then dissected and placed in 4% paraformaldehyde solution. Histological changes were subsequently observed by H&E staining by using conventional optical microscopes.
Microarray
LncRNA and mRNA expression profiles of kidney tissue were examined between the experimental group and the control group. Rats in the experimental group were fed with 6.6mg/kg cadmium for 4 weeks, and in the control group, we fed rats with physiological saline. Total RNA was extracted from the kidney of rats tissue using the Trizol reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer's instructions. RNA integrity was determined by capillary electrophoresis using the RNA 6000 Nano Lab-on-a-Chip kit and the Bioanalyzer 2100 (Agilent Technologies, Santa Clara, CA, USA). Only RNA extracts with RNA integrity number values >6 underwent in further analysis. Then RT Reagent Kit (Macherey - Nagel, Germany) was used to synthesize double-stranded complementary DNA (dsDNA). The dsDNA products were purified using a PCR NucleoSpin Extract II Kit (Macherey - Nagel, Germany) and eluted with 30 elution buffer.
Complementary RNA was synthesized from the eluted dsDNA products using a T7 Enzyme Mix at 37 °C for 14h and was purified using the RNA Clean-up Kit (Macherey - Nagel, Germany). 4μg random primer was used for amplifying RNA (2μg), and denatured at 65 °C for 5min on ice. Then, 4 × first-strand buffer(5μl ), 0.1 M DTT(2μl ), and 1.5μl CbcScript II reverse transcriptase were added. The mixtures were incubated at 25 °C for 10min, then at 37 °C for 90min. The cDNA products were purified using a PCR NucleoSpin Extract II Kit ( Macherey - Nagel, Germany)and the vacuum evaporated to 14μl. The cDNA was mixed with 4μg random primer, heated to 95 °C for 3min, and cooled on ice for 5min. Then, 5μl Klenow buffer, dNTP, and Cy3-dCTP (GE Healthcare) were added to final concentrations of 240 M dATP, 240 M dGTP, 240 M dTTP, 120M dCTP, and 40M Cy-dCTP. Next, the Klenow enzyme labeling strategy was adopted, and the reaction was performed at 37 °C for 90min. Labeled cDNA was purified with a PCR NucleoSpin Extract II Kit (MN) and resuspended in the elution buffer. Array hybridization was performed in a CapitalBio BioMixerTM II Hybridization Station overnight at a rotation speed of 8rpm and a temperature of 42 °C and washed with 0.2% SDS, 2 × SSC at 42 °C for 5min, and 0.2 × SSC for 5min at room temperature.
Double-stranded cDNA was labeled and hybridized to the CapitalBio Technology Rat LncRNA Array v1, 8x60K. The CapitalBio Technology Rat LncRNA Array v1 was designed with four identical arrays per slide (8 x 60K format), with each array containing probes interrogating about 22,020 rat lncRNAs and about 30,254 rat mRNAs. These lncRNA and mRNA target sequences were merged from the existing databases, such as RefSeq and Ensembl. After hybridization and washing, the processed slides were scanned with the Agilent Microarray Scanner. The lncRNA+mRNA array data were analyzed for data summarization, normalization and quality control by using the GeneSpring software V13.0 (Agilent Technologies, Inc). The data was Log2 transformed and median centered by genes using the Adjust Data function of CLUSTER 3.0 software University of Tokyo, Human Genome Center, Tokyo, Japan) then further analyzed with hierarchical clustering with average linkage. Finally, we performed tree visualization by using Java Treeview (Stanford University School of Medicine, Stanford, CA, USA).
Gene ontology (GO) and pathway enrichment analysis
Functional analysis, including gene ontology (GO) analysis and pathway analysis, was carried out by the standard enrichment computation method. GO analysis is frequently used in functional enrichment studies of large-scale genes. Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analysis was performed to analyze the biological pathways, involving the differentially expressed mRNAs. The PANTHER classifications are the results of sophisticated bioinformatics algorithms, and details of the methods can be found in the previous studies [24]. In the current study, David software was used to investigate the functional enrichment condition for the up and down-regulated differentially expressed mRNAs. This analysis helped us to determine the biological pathway in which there was significant enrichment of the differentially expressed mRNAs between the cadmium-group and control group. An FDR was calculated to correct the p-value and FDR < 0.05 was selected as the threshold.
The construction of lncRNA-mRNA weighted co-expression network
To associate the lncRNAs with the direct regulated expression of target mRNAs, we superimposed lncRNA target predictions onto the lncRNA-mRNA correlation network. The resulting network was defined as a lncRNA-mRNA regulatory network. Several differentially expressed lncRNAs and mRNAs were selected for coding-noncoding (CNC) gene co-expression analysis by Cytoscape software (v2.8.1) as previously described [25]. To make a visual representation, Pearson's correlation coefficients equal to >0.99 were applied as a criterion to confirm the coexpression relationships.
LncRNA
prediction
The Cis-acting lncRNA prediction and Trans-acting lncRNA prediction were used to predict the target mRNA of LncRNA. The Cis-acting lncRNA prediction was performed by their tight correlation (Pearson’s correlation coefficient minimum of 0.99) to a group of expressed protein-coding genes. The lncRNA resided at genomic loci where a protein-coding gene and a lncRNA gene were within 10 kb of each other along the genome [26]. and Trans-acting lncRNA prediction was conducted using blat tools (Standalone BLAT v. 35x1 fast sequence search command-line tool, down) compare the full sequence of the lncRNA with the 3' UTR of its co-expression mRNAs with the default parameter setting.
The mRNA and LncRNA co-expression analysis network
The basic method of correlation analysis was used to calculate and verify the correlation coefficients, and p values were calculated by using the standardized signal values. The correlation algorithm adopts Pearson. The P-value was not multi-corrected by default. The total results of the co-expression analysis were significantly correlated. The selection criteria was correlation > 0.99 or correlation<-0.99, and P value<0.05. 1, 1000 pairs of related genes with the highest correlation coefficient were selected from the co-expression results, and their network map was plotted with Cytoscape software.
Transcription factor prediction
According to Guttman et al. [27], specific lncRNAs participating in certain biological pathways were regulated by key transcription factors (TFs), and regulate these pathways. Thus, we compared the mRNAs that co-expressed with these lncRNAs with the mRNAs to categorize lncRNAs that possibly have trans-regulating functions which are regulatory targets of certain TFs. We predicted that these lncRNAs possibly participate in pathways regulated by these TFs, If the intersection of these 2 groups is large enough (P<0.05; and the P-value was calculated by hypergeometric cumulative distribution function and FDR <0.05). The Match 1.0 public transcription factor prediction tool was used to predict the transcription factor of LncRNA (TFs_predict). The graph of the lncRNA-TF network was drawn with the help of Cytoscape 3.01. to predict the binding status of 2000bp upstream and 500bp downstream of each lncRNA starting a site with transcription factors.
RNA extraction and RT-qPCR
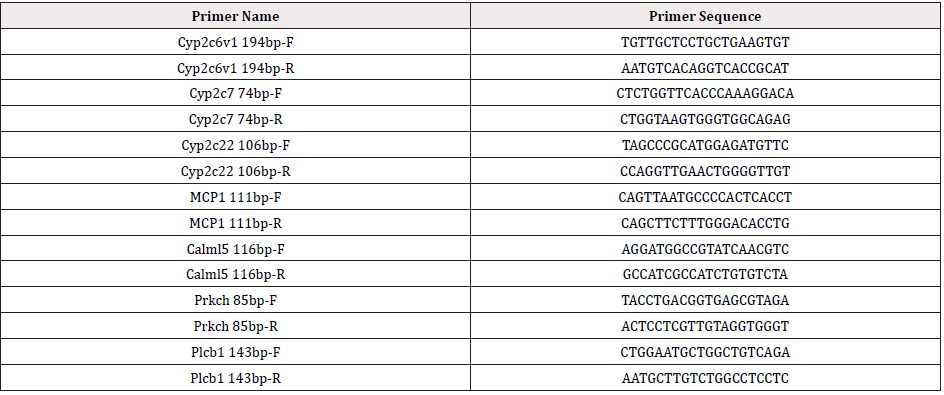
Total RNA was extracted from cells using TRIzol reagent (Life Technologies, Grand Island, NY, USA) according to the manufacturer's instructions. The extracted mRNA then stored at -80˚C until use. qPCR was performed with an Applied Biosystems StepOne™ real-time PCR system (Applied Biosystems; Thermo Fisher Scientific, Inc.). The fast SYBR® Green Master Mix was obtained from Applied Biosystems (Thermo Fisher Scientific, Inc.). The following primers used in this step were shown in Table 1.
The expression of each mRNA was represented as fold change using 2−∆∆ Ct methods. The expression differences of mRNAs between the models and the controls were analyzed using Student's t-test. P-value < 0.05 was considered as significant.
Statistical Analysis
All the statistical data are shown as the mean±standard error from at least three separate experiments and were analyzed using SPSS 22.0 software (Chicago, IL, USA) and R software (Version 3.2.3). Student’s t-test was applied for comparisons between two groups, and differences with P < 0.05 were considered statistically significant.
Results
The kidney damage model of rats caused by cadmium exposure was successfully constructed by gavage
Rats in the cadmium group (n=6) received daily intragastric administration of CdCl2 at a dose of 6.6mg/kg in 1ml isotonic saline for up to 8 weeks. The control group, received daily injections of the saline vehicle alone. After executing the rats at 8 weeks, the kidneys were taken for pathological examination. There was a significant lesion in the kidney. These pathological conditions reveal kidney tissues of rats were heavily necrotic as the Figure 1B. There was no nucleus in epithelial cells, and tubulin was observed in the renal tubule. Renal medullary hyperemia, enlargement of blood vessels and capillaries, swelling of capillary endothelial cells and the cytoplasmic residues of dead cells were observed in most renal tubules. Hyperemia was evident in the renal cortex, and there were a large number of inflammatory cells, and the renal tubular epithelium shows water- like degeneration and appears cellular cavitation (Figure 1).
Identification of differentially expressed lncRNAs and mRNAs
LncRNAs can directly regulate gene expression independent of inter-pathway interactions. Therefore, we tried to seek for answers via lncRNA microarray analysis, and we generated normalized gene expression data from 22,020 rat lncRNAs and about 30,254 mRNA probes. High-throughput microarray analysis is an efficient approach that facilitated our investigation of lncRNA and mRNA expression signatures in muscle development. Based on the cut-off criteria, compared differentially expressed genes in the cadmium damage group and control group. The results of the global profiling of differentially expressed lncRNAs and mRNAs between cadmium damage group and control group were performed in Figure 2. The volcano plot of these probe-matched gene expressions of mRNA and LncRNA shows the up-regulated and down-regulated mRNAs (Figure 2C) and LncRNAs (Figure 2E) profiling across groups. The blue and red dots stand for down-regulated and up-regulated expressions, respectively. In our microarray analysis, a total of 495 mRNA transcripts was changed by added cadmium in the kidney, including 180 mRNA expression were up-regulated, and 315 mRNA were down-regulated. 322 lncRNA transcripts were differentially expressed in the cadmium group, with 142 up-regulated and 180 down-regulated. Among these dysregulated lncRNA transcripts, NONRATT022879 was the most up-regulated, with an FC of genes was 2.4190 and the P-value was 0.0095. whereas NONRATT019758 was the most down-regulated, and the FC of the gene was 7.28 and the P-value was 0.00574. Heat map of those dysregulated lncRNAs was generated by unsupervised hierarchical clustering analysis (Figure 3).
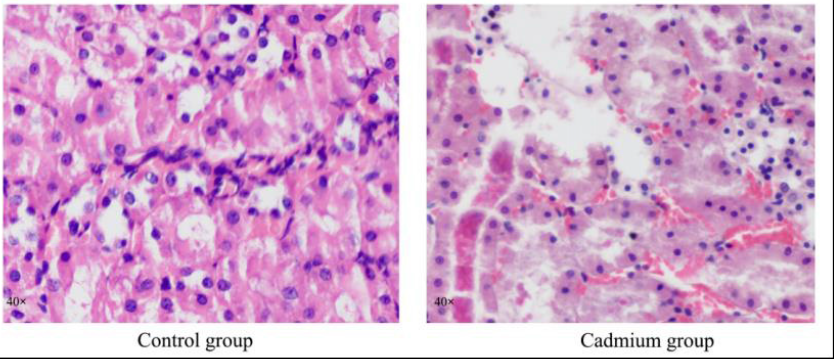
Figure 1: Histological examination of kidney tissues on the control group and cadmium group which damaged by cadmium as shown by HE staining (magnification × 40).
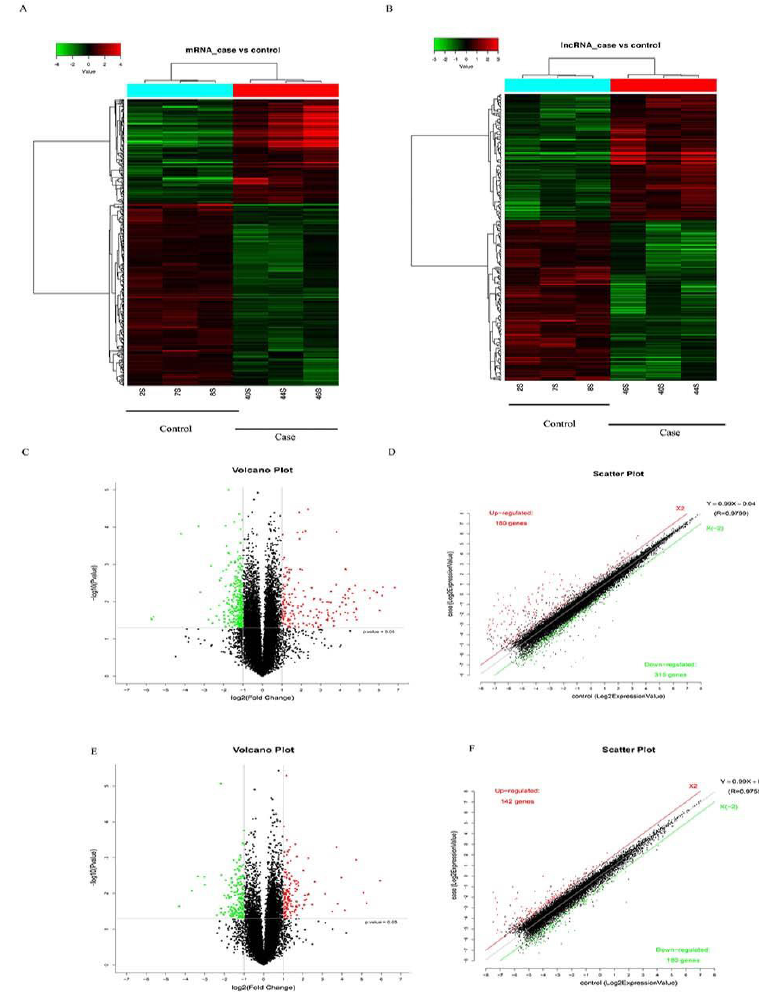
Figure 2: Global profiling of differently expressed lncRNAs and mRNAs in the cadmium damage group and control group. Heat map of the expression profiles of (A) mRNAs and (B) lncRNAs. (C) Scatter plots present differences in the expression of mRNAs and the volcano plot of the differentially expressed mRNA probes. (D) Scatter plots present differences in the expression of LncRNAs probes. (E) The volcano plot of the differentially expressed mRNA probes. (F) The values plotted on the x- and y-axes are the averaged normalized signal values of each group (log2 scaled).
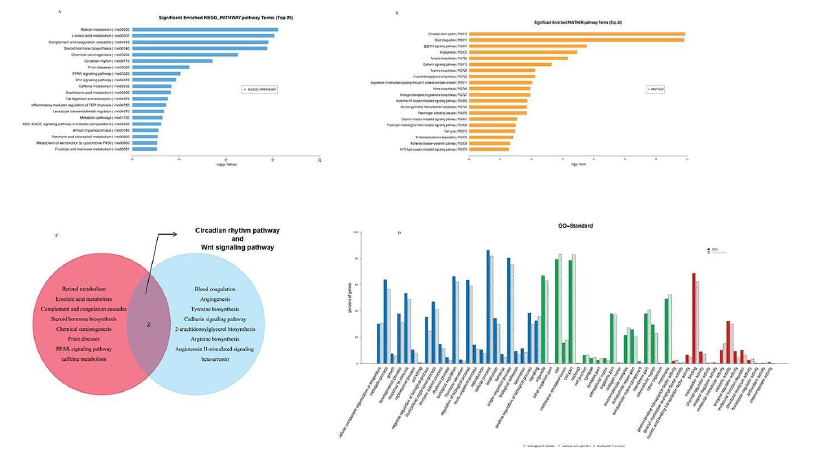
Figure 3: GO analysis and pathway analysis of differently expressed mRNAs. (A) KEGG analysisand the related pathways were sorted by p value. (B) Panther analysis and the related pathways were sortedg by p value. (C) Venn of KEGG and panther pathway analysis. (D) The left vertical is the percentage of genes, the right vertical is the number of genes that different, and the grey data is the number of background genes (usually all on the chip). Solid post stands for different genes, and dotted column stands for background gene. The figure shows the enrichment of different genes compared with background genes
GO and pathway enrichment analysis
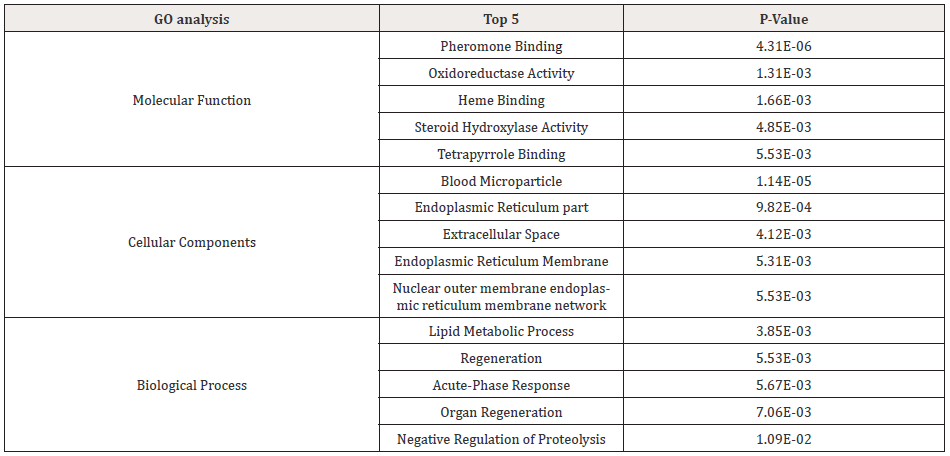
GO analysis was performed to investigate differently expressed mRNA attributes in any organism, including molecular function, biological processes, and cellular components. Among the upregulated mRNAs in the case group, there were 127 groups of genes involved in biological processes, 26 groups of genes involved in cellular component and 55 groups of genes involved in molecular function. Among the downregulated mRNAs, there were 225 groups of genes involved in the biological process, 25 groups of genes involved in cellular components and 14 groups of genes involved in molecular function. And the p values denote the significance of GO term enrichment in the different groups. The significance of GO term was analyzed as p <0.05. As a result, Figure 4 was shown as the GO analysis. And top 5 regulated genes of GO analysis was listed as Table 2. KEGG mapping pathways were used for pathway analysis. p values were denoted the significance of the pathway correlation. The lower p-value, the more significant of the pathway (the p-value cutoff was 0.05).
Pathway enrichment analysis of differentially expressed mRNAs showed the top 20 enrichment score values of the significantly enriched pathways by KEGG pathway (Figure 2A) analysis and panther pathway analysis (Figure 2B). The regulated mRNAs were associated with “Retinol metabolism”, “Linoleic acid metabolism”, “Complement and coagulation cascades”, “Steroid hormone biosynthesis”, “Chemical carcinogenesis”, “Circadian rhythm”, “Prion diseases”, “PPAR signaling pathway”, “Wnt signaling pathway”, and these were listed top10 in KEGG pathway. We also used the panther pathway to analyzed the differential genes. “Circadian clock system”, “Blood coagulation”, “Wnt signaling pathway”, “Angiogenesis”, “Tyrosine biosynthesis”, “Cadherin signaling pathway”, “2-arachidonoylglycerol biosynthesis”, “Arginine biosynthesis”, “Angiotensin II-stimulated signaling through G proteins ”and “beta-arrestin” was the top10 terms in panther pathway. Through two methods of path analysis, the results showed that the effects of cadmium on rat kidneys were involved in Circadian rhythm and Wnt signaling pathway.
The construction of lncRNA-mRNA weighted co-expression network
We first constructed general coding and noncoding genes co-expression networks between the cadmium group and control group with all different expression of LncRNAs and mRNAs in this study. Those LncRNAs and mRNAs had Pearson correlation coefficients equal to or > 0.99 were selected, and subsequently, a network including top 1000 correlation coefficient was constructed in each of the above-mentioned groups using cytoscape program for co-expression network. The results were shown in Figure 4. The data was showed that the co-expression network was composed of top 1000 network nodes and 44677 connections between 10 lncRNAs and coding genes .
The target mRNA of LncRNA prediction and functional analysis
The network showed that one mRNA was correlated with one or more lncRNAs. The target genes of lncRNAs with top 10 correlation coefficient were predicted by lncRNA prediction as Figure 4A. Meanwhile, as Figure 4B reveals, the network was operative in some meaningful pathways related to the development of “Linoleic acid metabolism”, “Steroid hormone biosynthesis”, “Retinol metabolism”, “Arachidonic acid metabolism”, “Chemical carcinogenesis”, “Inflammatory mediator regulation of TRP channels”, “Serotonergic synapse” and “Dorso-ventral axis formation” , etc. The results of interacted with mRNAs involving in the “Steroid hormone biosynthesis”, and 17 lncRNAs were interacted with 2 mRNAs in the KEGG of “Steroid hormone biosynthesis”, this was consistent with the results of KEGG analysis for differentially expressed mRNA. Meanwhile, 13 lncRNAs interacting with 2 mRNAs in the KEGG of “Linoleic acid metabolism”. Furthermore, predicted function of lncRNAs was annotated using GO terms analysis of co-expressed mRNAs. For example, lncRNA NONRATT002903 and was associated with positive regulation of Cyp2c7 production and related with “response to external biotic stimulus” in Biological Process and Cyp2c7 production also related with “arachidonic acid monooxygenase activity” in Molecular Function, etc. (Figure 5).
Transcription factor
prediction
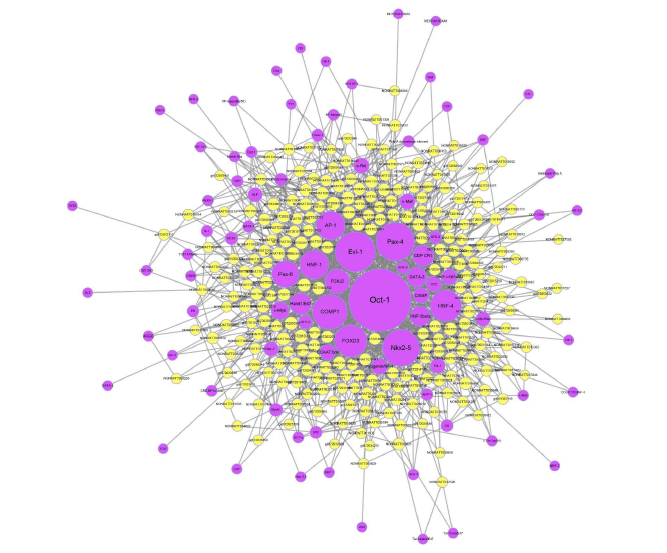
Transcription factors (TFs) are key regulators of the transcriptional expression of multiple genes in diverse biological processes [28]. 1893 lncRNA-Transcription factor(TF) pairs were found by using the threshold of P<0.05 and FDR <0.05, corresponding to 1449 TFs. Then, we generated a core network using the top 1000 lncRNA-TF pairs with the most credentiality (lowest P-values and FDRs), as shown in Figure 6, we found that most of these potential trans-regulatory lncRNAs participate in pathways, which regulated by top5 TFs: FOXJ2, Pax-6, 1-0ct, HNF-4 and COMP1.
Validation of differentially expressed mRNA involving inflammatory mediator regulation of TRP channels by qRT-PCR
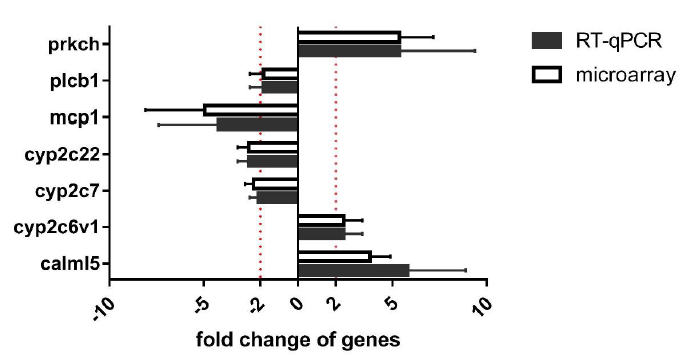
Through enrichment analysis of differentially expressed mRNAs, it was found that differentially expressed mRNAs could be enriched to inflammatory mediator regulation of TRP channels after cadmium exposure caused kidney damage in rats. Similarly, after predicting the differentially expressed LncRNA of the target gene, it was found that its corresponding mRNA can also be enriched to inflammatory mediator regulation of TRP channels. Therefore, research suggests that this pathway may be related to kidney damage caused by cadmium, and also involves LncRNA regulation. To verify the microarray results, we selected all differentially expressed mRNAs (7 genes) involving in inflammatory mediator regulation of TRP channels in this study, and validated their expression levels by RT-qPCR analysis. As shown in Figure 7. the gene expression of prkch, cyp2c6v1 and calml5 were significantly up-regulated in cadmium group compared with control, while the gene expression of plcb1, mcp1,cyp2c22 and cyp2c7 and were significantly down-regulated. The expression patterns of these six mRNA from qRT-PCR results were all in accordance with the microarray analysis.
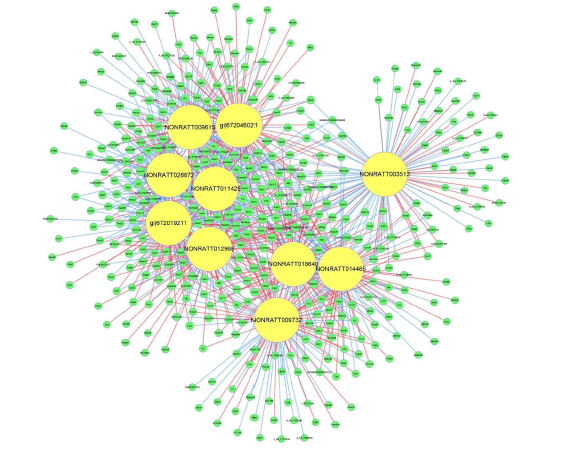
Figure 4: Total LncRNA-mRNA-Network. Interaction networks of differently expressed genes. The yellow circle represents LncRNA, the green circle represents mRNA, the size of the circle represents the degree (number of neighbors) of genes in the network, the red line represents the positive correlation, and the blue line represents the negative correlation.
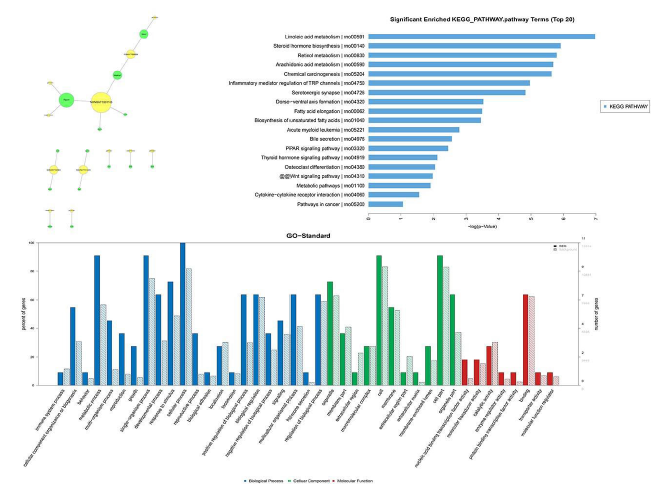
Figure 5: LncRNA prediction and the Functional analysis of LncRNA target genes. (A) LncRNA prediction using trans prediction. The yellow circle represents LncRNA, the green circle represents mRNA. (B) Top 20 significantly enriched. KEGG pathway of differentially expressed lncRNAs prediction genes (C) GO analysis for target genes of LncRNA prediction
Discussion
Cadmium is a toxic chemical that adversely affects kidney function, and cadmium exposure can induce renal toxicity [29,30]. Cadmium occurs in all food, but agricultural crops (particu-larly irrigated rice) generally account for most of the intake. Therefore, in this study, gastric was conducted in the form of digestive tract to rat. Exposure to cadmium through prolonged gavage was used to imitate the human experience of ingesting cadmium through food over a long period of time. A wide range of cadmium effects has been reported at the cellular and molecular level. Kidney function is to excrete metabolic products, adjust the balance of water, electrolyte and acid-base, and secret active substances to maintain homeostasis [31]. Prior researches have demonstrated that there was a certain degree disorder of metabolism with injured kidney functions by added cadmium. In some cases the mechanism are controversial, however, and in recent years there has been an ongoing discussion on the mechanism of general effects and chronic toxicity for cadmium. Several mechanisms have also been proposed to account for cadmium-induced nephrotoxicity, including oxidative stress, cell apoptosis, and glomerular contraction [32]. But its mechanism of action remains to be fully elucidated. There have been no studies on gene microarray for kidney damage caused by cadmium exposure. In the current work, we studied the expression of genes and metabolic pathways specifically modulated by cadmium. For this purpose, a gene microarray analysis of mRNA and LncRNA was performed in the kidney of rats. We investigated that the gene expression modulation in the kidney tissue of rats after exposure to 6.6mg/kg cadmium for 8 weeks by using an Agilent microarray.
After exposure to cadmium, Cd accumulates in the kidney via a receptor-mediated endocytosis and as Cd-MT complexes, in the proximal tubules. Internalized Cd-MTs are degraded in endosomes and lysosomes and, by releasing free Cd2+ into the cytosol, generate reactive oxygen species (ROS) and activate cell death pathways [33]. In this study, the pathological conditions reveal kidney tissues of rats were heavily necrotic.
The renal medullary hyperemia, and both blood vessels and capillaries are enlarged. Capillary endothelial cells swell and swell thickening. Blood vessel basement membrane was thickened. Cytoplasmic residues of dead cells can be seen in most renal tubules. Neoplasia, apoptosis and necrosis was appearing in renal tubule cells (mainly proximal convoluted tubule). The renal cortex appears hyperemia obviously, and there were lots of inflammatory cells. The renal tubule epithelium is hydrophilic and vacuolar. Under this pathological conditions, the image showed that we successfully constructed a model of kidney injury that induced by cadmium in rats. Due to increased permeability of the glomerular basement membrane, a large amount of protein enters the renal tubule from the glomerular. In the convoluted tubule and collecting duct distal part of the kidney, the protein coagulates and precipitates in the renal tubular cavity, forming a tubular shape.
There have been no studies on gene microarray for kidney damage caused by cadmium exposure. In the current study, we demonstrated that there was a significantly altered lncRNA and mRNA expression profile using microarray technology in kidney tissue of rats exposure to cadmium. In this study, we identified 180 significantly up and 315 down-regulated gene. Gene ontology (GO) and pathway enrichment analyses were performed to analyze the biological functions and pathways for the differentially expressed mRNAs. KEGG and panther pathway analysis were used for enrichment the gene. We found that the pathway of wnt signaling pathway and circadian rhythm pathway were involved. Some previous studies also have found that cadmium exposure is associated with circadian rhythm. This was consistent with the results of these studies. The results still have indirect implications to some extent that cadmium exposure might affect the differentially express of genes related to circadian rhythm. Previous research has found that oxidative stress and immunotoxicity were more serious when fish were exposed to cadmium in the evening than in the morning, which highlight the importance of circadian rhythm in Cd-induced neurotoxicity in fish [34]. In this study, the expression of mRNA involving circadian rhythm was also verified by RT-qPCR, and the results were consistent with those of Microarray, and the results were shown in Figure 7. We also submitted these genes to STRING to analyze the relationship among the genes, and drawn it by cytoscape software.
The GO analysis showed that pheromone binding, oxidoreductase activity, heme binding, steroid hydroxylase activity, tetrapyrrole binding were the top5 significantly enriched in molecular function. Lipid metabolic process, regeneration, acute-phase response, organ regeneration and negative regulation of proteolysis were top 5 enriched in biological process. Cellular macromolecule localization, single-organism cellular localization, protein localization to membrane, cytoplasmic transport, cell surface receptor signaling pathway were significantly enriched at top 5 of cellular components. Interesting findings: Cadmium exposure alters the expression of lipid metabolism-related genes. In our study, the expression of 42 genes related to lipid metabolic process were changed. Previous studies have also found that cadmium exposure might cause some changes in lipid metabolism, and for example, cadmium increased the lipid synthesis in liver [35]. Some research suggested that the effects caused by cadmium toxicity in the tissues were considered to be associated with the impairment in anti-oxidant defense system, and lipid peroxidation was increases after cadmium exposure [36]. MDA is the end product of peroxidation of unsaturated fatty acids in the cells, cadmium exposure could increase free radicals, and which might cause an increase in MDA production [37]. As is known to all, the MDA level is known to reflect oxidative stress and the status of the anti-oxidant system [38]. The down-regulation in these genes transcription may be responsible for reduced the ability of fatty acid β-oxidation after cadmium exposure. In our study, 16 differentially genes were involved in regeneration pathway. That's the implication that cadmium exposure might influence regeneration of kidney tissue. Liu [39] demonstrated that cadmium exposure induced apoptosis and subsequent regeneration of the renal tubular epithelium in chicken kidney [39]. The mechanisms involved in the regeneration of kidney damage by cadmium exposure are still not clear and need further investigation.
To obtain new insights into the function of lncRNAs in kidney of rats with cadmium exposure, ten lncRNAs were selected for network construction. The co-expression analysis is based on the correlation in mathematics, looking for LncRNA-mRNA relationship pairs with similar expression profiles from the level of gene expression. Therefore, co-expression analysis could help to discover the possible functional relationship between lncRNA and mRNA, and found that the lncRNA which affects mRNA expression regulation, meanwhile, this study could discover the lncRNA which plays a central regulatory role in the network and the possible new functional mechanism of lncRNA [40].
The Cis-acting lncRNA prediction and Trans-acting lncRNA prediction were used to predict the target mRNA of LncRNA. The target genes of differentially lncRNAs with top 10 correlation coefficient were predicted by lncRNA prediction as Figure 5A. and KEGG and panther pathway were used for analyzing the pathway enriched by these genes. It was found that the target genes of the differentially expressed LncRNA involved linoleic acid metabolism and steroid ormone biosynthesis etc. From these results, we found that some target genes of differentially LncRNA, for example NONRATT007029, NONRATT001106 and NONRATT014465 were enriched into the pathways of inflammatory mediator regulation of TRP channels [41]. TRP channels can be modulated indirectly by inflammatory mediators, which are generated during tissue injury [42]. TRP channels are emerging as sensory transducers that may participate in the generation of pain sensations evoked by chemical, thermal and mechanical stimuli. In this currently results, TRPV1 was down-regulated in TRP channels. Some studies suggest that TRPV1 may play a role in the development and maintenance of chronic pain [43,44]. The target genes of differentially LncRNAs were enriched in Wnt signal by panther pathway analysis, including Ppard gene. Ppard is a ligand-activated transcription factor. Prior studies suggested Ppard play a role for this protein in lipid metabolism, differentiation, and epidermal cell proliferation [45]. These results suggests that lncRNAs may play an important role in their corresponding networks or signalling, and therefore may contribute to the molecular regulation of MDD. LncRNAs participate in the regulation of gene expression by targeting transcription factors, initiating chromatin remodelling, directing methylation complexes, and blocking nearby transcription [46].
Transcription factors (TFs) are key regulators of the transcriptional expression of multiple genes in diverse biological processes. The results were shown that most of these potential trans-regulatory lncRNAs participate in pathways, which regulated by top5 TFs: FOXJ2, Pax-6, 1-oct, HNF-4 and COMP1. There are several limitations in our study. Firstly, like other studies based on gene microarray analysis , the altered genes that were revealed were limited to the RNA level, and not all of these RNA-level changes led to changes at the protein level. Secondly, we only compared the gene expression profiles of kidney tissues exposed to 6mg/kg cadmium with control group for 8 weeks, and therefore did not evaluate the changes of genes that occur during the different exposure times and different doses. Nevertheless, in this study, the genetic changes of rats after renal injury caused by exposure cadmium were investigated for the first time by gene microarray.
Acknowledgment
The authors wish to acknowledge professor CHUN-HONG WANG of Wuhan university for her help in interpreting the significance of the results in this study.
Funding
The authors acknowledge funding from National Natural Science Foundation of China (NO. 81660535) and Natural Science Foundation of Jiangxi ( 20181BAB205067).
Availability of Data and Materials
All data generated or analyzed during this study were included in this published article.
Authors’ Contributions
Shaoxin Huang conceived and designed the study. Hui Liu and Shaoxin Huang wrote the paper. Hui Liu, Xichen Wan, Qihan Zhao, Jin Ouyang performed the experiments. Jun Liu analyzed the results. All authors reviewed the manuscript.
Highlights
- The animal model of renal injury induced by cadmium poisoning in rats was established by intragastric administration, which provided the method basis for the establishment of animal model (including dose and intervention time) for related research.
- The possible molecular effects and mechanisms of cadmium-induced renal damage were explained by LncRNA and mRNA gene chip combined with bioinformatics analysis.
- Renal damage induced by cadmium may be related to LncRNA-mediated "The inflammatory mediator regulation of TRP channels" pathway.
Ethics Approval and Consent to Participate
Not applicable.
Patient Consent for Publication
Not applicable.
Competing Interests
The authors declare that they have no competing interests.
References
- Kawada T (2018) Cadmium intake and chronic kidney disease. Clin Nutr 37(5): 1779.
- Shi Z, Taylor AW, Riley M, Byles J, Liu J, et al. (2018) Association between dietary patterns, cadmium intake and chronic kidney disease among adults. Clin Nutr 37(1): 276-284.
- Jain RB (2019) Cadmium and kidney function: Concentrations, variabilities, and associations across various stages of glomerular function. Environ Pollut 256: 113361.
- Godt J, Scheidig F, Grosse Siestrup C, Esche V, Brandenburg P, et al. (2006) The toxicity of cadmium and resulting hazards for human health. Journal of Occupational Medicine and Toxicology 1(1): 22.
- Wang Q, Wei S (2018) Cadmium affects blood pressure and negatively interacts with obesity: Findings from NHANES 1999-2014. Sci Total Environ 643(1): 270-276.
- Le TH, Alassane Kpembi I, Oswald IP, Pinton P (2018) Analysis of the interactions between environmental and food contaminants, cadmium and deoxynivalenol, in different target organs. Sci Total Environ (1): 622-623, 841-848.
- Zhang L, Song L, Liu B, Wu M, Wang L, et al. (2019) Prenatal cadmium exposure is associated with shorter leukocyte telomere length in Chinese newborns. BMC Med 17(1): 27.
- Madrigal JM, Ricardo AC, Persky V, Turyk M (2010) Associations between blood cadmium concentration and kidney function in the U.S. population: Impact of sex, diabetes and hypertension. Environ Res 169: 180-188.
- Bernhoft RA (2013) Cadmium toxicity and treatment. Scientific World Journal 2013.
- Godt J, Scheidig F, Grosse Siestrup C, Esche V, Brandenburg P, et al. (2006) The toxicity of cadmium and resulting hazards for human health. Journal of Occupational Medicine and Toxicology 1(1): 22.
- Thomas LD, Elinder CG, Tiselius HG, Wolk A, Akesson A (2013) Dietary cadmium exposure and kidney stone incidence: a population-based prospective cohort study of men & women. Environ Int 59: 148-151.
- Kostecka Sochon P, Onopiuk BM, Dabrowska E (2018) Protective Effect of Increased Zinc Supply against Oxidative Damage of Sublingual Gland in Chronic Exposure to Cadmium: Experimental Study on Rats. Oxid Med Cell Longev.
- Ge J, Zhang C, Sun YC, Zhang Q, Lv MW, et al. (2019) Cadmium exposure triggers mitochondrial dysfunction and oxidative stress in chicken (Gallus gallus) kidney via mitochondrial UPR inhibition and Nrf2-mediated antioxidant defense activation. Sci Total Environ 689(1): 1160-1171.
- Gobe GC, Johnson DW (2007) Distal tubular epithelial cells of the kidney: Potential support for proximal tubular cell survival after renal injury. The International Journal of Biochemistry & Cell Biology 39(9): 1551-1561.
- McRae NK, Gaw S, Glover CN (2018) Effects of waterborne cadmium on metabolic rate, oxidative stress, and ion regulation in the freshwater fish, inanga (Galaxias maculatus). Aquat Toxicol 194: 1-9.
- Luo B, Lin Y, Jiang S, Huang L, Yao H, et al. (2016) Endoplasmic reticulum stress eIF2alpha-ATF4 pathway-mediated cyclooxygenase-2 induction regulates cadmium-induced autophagy in kidney. Cell Death Dis 7(6): e2251.
- Shi L, Cao H, Luo J, Liu P, Wang T, et al. (2017) Effects of molybdenum and cadmium on the oxidative damage and kidney apoptosis in Duck. Ecotoxicol Environ Saf 145: (24-31).
- Kawada T (2018) Cadmium intake and chronic kidney disease. Clin Nutr.
- Wang KC, Chang HY. (2011) Molecular Mechanisms of Long Noncoding RNAs. Molecular Cell. 43(6): 904-914.
- Gao M, Li C, Xu M, Liu Y, Cong M, et al. (2018) LncRNA MT1DP Aggravates Cadmium-Induced Oxidative Stress by Repressing the Function of Nrf2 and is Dependent on Interaction with miR-365. Adv Sci (Weinh) 5(7).
- Huang Q, Lu Q, Chen B, Shen H, Liu Q, et al. (2017) LncRNA-MALAT1 as a novel biomarker of cadmium toxicity regulates cell proliferation and apoptosis. Toxicol Res (Camb) 6(3): 361-371.
- Hirao Suzuki M, Takeda S, Kobayashi T, Kino K, Miyazawa H, et al. (2018) Cadmium down-regulates apolipoprotein E (ApoE) expression during malignant transformation of rat liver cells: direct evidence for DNA hypermethylation in the promoter region of ApoE. The Journal of Toxicological Sciences 43(9): 537-543.
- Cao H, Xia B, Zhang M, Liao Y, Yang Z, et al. (2016) Changes of Antioxidant Function and the mRNA Expression Levels of Apoptosis Genes in Duck Ovaries Caused by Molybdenum or/and Cadmium. Biological Trace Element Research 171(2): 410-418.
- Mi H, Muruganujan A, Thomas PD (2013) PANTHER in 2013: modeling the evolution of gene function, and other gene attributes, in the context of phylogenetic trees. Nucleic Acids Res 41(Database issue) pp. D377-D386.
- Dong R, Jia D, Xue P, Cui X, Li K, et al. (2014) Genome-Wide Analysis of Long Noncoding RNA (lncRNA) Expression in Hepatoblastoma Tissues. PLOS ONE 9(1): e85599.
- Jia H, Osak M, Bogu GK, Stanton LW, Johnson R, et al. (2010) Genome-wide computational identification and manual annotation of human long noncoding RNA genes. RNA 16(8): 1478-1487.
- Guttman M, Amit I, Garber M, French C, Lin MF, et al. (2009)Chromatin signature reveals over a thousand highly conserved large non-coding RNAs in mammals. Nature 458(7235): 223-227.
- Zhang Z, Yang P, Yao P, Dai D, Yu Y, et al. (2016) Identification of transcription factors and gene clusters in rabbit smooth muscle cells during high flow-induced vascular remodeling via microarray. Gene 575(2, Part 2): 407-414.
- Gabr SA, Alghadir AH, Ghoniem GA (2017) Biological activities of ginger against cadmium-induced renal toxicity. Saudi Journal of Biological Sciences 26(2): 382-389.
- Rana K, Verma Y, Rani V, Rana SVS (2018) Renal toxicity of nanoparticles of cadmium sulphide in rat. Chemosphere. 193:142-150.
- Gao JR, Qin X, Jiang H, Wang T, Song J, et al. (2016) Screening and functional analysis of differentially expressed genes in chronic glomerulonephritis by whole genome microarray. Gene 589(1): 72-80.
- Rani A, Kumar A, Lal A, Pant M (2014) Cellular mechanisms of cadmium-induced toxicity: a review. International Journal of Environmental Health Research 24(4): 378-399.
- Johri N, Jacquillet G, Unwin R (2010) Heavy metal poisoning: the effects of cadmium on the kidney. BioMetals 23(5): 783-792.
- Zheng J, Yuan S, Wu C, Lv Z, Zhu A (2017) Circadian time-dependent antioxidant and inflammatory responses to acute cadmium exposure in the brain of zebrafish. Aquatic Toxicology 182: 113-119.
- Wu C, Zhang Y, Chai L, Wang H (2017) Histological changes, lipid metabolism and oxidative stress in the liver of Bufo gargarizans exposed to cadmium concentrations. Chemosphere 179: 337-346.
- Erboga M, Kanter M, Aktas C, Sener U, Fidanol Erboga Z, et al. (2016) Thymoquinone Ameliorates Cadmium-Induced Nephrotoxicity, Apoptosis, and Oxidative Stress in Rats is Based on its Anti-Apoptotic and Anti-Oxidant Properties. Biological Trace Element Research 170(1): 165-172.
- Gawel S, Wardas M, Niedworok E, Wardas P (2004) [Malondialdehyde (MDA) as a lipid peroxidation marker]. Wiad Lek 57(9-10): 453-455.
- Liu R, Xu Y, Li B, Qin G, Tian S (2015) The role of 1-methylcyclopropene in lipid peroxidation, anti-oxidant enzyme activities, and ethylene biosynthesis in ‘Laiyang’ pear (Pyrus bretschneideri Rehd.) during fruit ripening. The Journal of Horticultural Science and Biotechnology 90(2): 210-216.
- Liu L, Yang B, Cheng Y, Lin H (2015) Ameliorative Effects of Selenium on Cadmium-Induced Oxidative Stress and Endoplasmic Reticulum Stress in the Chicken Kidney. Biological Trace Element Research 167(2): 308-319.
- Liu Z, Li X, Sun N, Xu Y, Meng Y, et al. (2014) Microarray Profiling and Co-Expression Network Analysis of Circulating lncRNAs and mRNAs Associated with Major Depressive Disorder. PLOS ONE 9(3): e93388.
- Clapham DE (2003) TRP channels as cellular sensors. Nature 426(6966): 517-524.
- Levine JD, Alessandri-Haber N (2007) TRP channels: Targets for the relief of pain. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease 1772(8): 989-1003.
- Gold MS (2000) Spinal nerve ligation: what to blame for the pain and why. PAIN® 84(2): 117-120.
- Davis JB, Gray J, Gunthorpe MJ, Hatcher JP, Davey PT, et al. (2000) Vanilloid receptor-1 is essential for inflammatory thermal hyperalgesia. Nature 405(6783): 183-187.
- Elinder CG, Lind B, Kjellstrom T, Linnman L, Friberg L (1976) Cadmium in kidney cortex, liver, and pancreas from Swedish autopsies. Estimation of biological half time in kidney cortex, considering calorie intake and smoking habits. Arch Environ Health 31(6):292-302.
- Ponting CP, Oliver PL, Reik W (2009) Evolution and Functions of Long Noncoding RNAs. Cell 136(4): 629-641.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.