Case Report 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Primary Typical Carcinoid of the Pleura: Case Report
*Corresponding author: Lamyae Nouiakh, Department of Medical Oncology, Hassan II University Hospital, Morocco, Tel: +212 641510692
Received: September 01, 2020; Published: November 09, 2020
DOI: 10.34297/AJBSR.2020.10.001566
Abstract
Carcinoid tumors are rare, slow-growing neuroendocrine tumors. The most common location of these tumors is in the gastrointestinal tract and broncho-pulmonary system. Primitive pleural localization is very rare. The majority of cases found in the literature are metastatic forms or relapses. We report the case of a 56-year-old patient with a typical carcinoid tumor of the pleura.
Keywords: Pleura; Typical carcinoid tumor; Multidisciplinary approach
Background
Carcinoid tumors are a very rare variety of neuroendocrine tumors, most commonly found in the digestive tract and in the lungs [1]. Primary pleural localization is rarely found. Typical carcinoid tumors pose a diagnostic problem. Because of the scarcity of these tumors, this diagnosis is sometimes difficult. Histologically, it is a neuroendocrine tumor whose morphology often joins that in other locations.
The prognosis of these tumors are generally pejorative given the late diagnosis, the absence of an effective therapeutic approach, even for localized stages where surgery remains the standard treatment as it is impossible to perform carcinological surgery due to the localization of the tumor. We report the case of a 56-year-old patient with a typical carcinoid tumor of the pleura.
Case Presentation
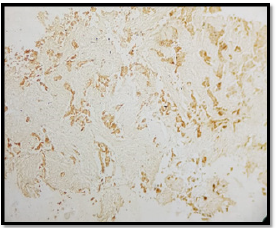
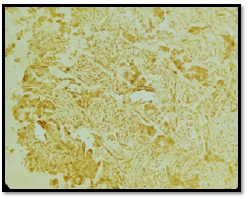
A 56-year-old patient, 30 years of chronic smoking, weaned 6 years ago, who consulted for grade 2 dyspnea according to NYHA classification evolving for more than a year, becoming grade 4 one month before your admission without any other associated signs. The clinical examination found a patient in moderate performance status (PS2) with a left pleural effusion syndrome at the pulmonary examination. The rest of the clinical examination was normal. As a result of symptomatology, the patient benefited from a standard X-ray and then a chest scan that showed a left pleural nodular thickening reaching of 18 mm in maximum thickness associated with a pleural effusion of great abundance responsible for the pulmonary collapse and discharge from the medieval elements to the right (Figure 1). A pleural puncture biopsy was performed for diagnostic and evacuating purposes. Histological analysis showed infiltration of the pleural membrane by malignant tumor proliferation consisting of epitheliomatous cell flows, cord, and trabecular structure, sometimes laying out glanduliform structures or acinous formations which are arranged in an endocrine mode in palisades and rosettes, dissociated by a desmoplastic stroma of variable abundance. The tumor cells were rounded, polyhedral, or fusiform with eosinophilic cytoplasm and ovoid nuclei, sometimes voluminous, nucleoli with rare mitosis figures: less than 2M/10CFG (Figures 2 & 3). At immunohistochemistry, the tumor cells expressed antibody anti CK7, anti- SYNAPTOPHYSINE, but instead, they were negative for anti-chromogranin, anti-CD56, anti-CK20, and anti-CDX2 antibodies (Figures 4 & 5). The pathological study concluded a typical carcinoid tumor of pleural localization. The patient completed the extension assessment with an abdominal-pelvic computed tomography which did not show any secondary localization. The case was discussed in a multidisciplinary consultation meeting, the tumor was found to be unresectable and the therapeutic decision was to start the first-line chemotherapy. Unfortunately, the patient died 15 days later due to respiratory failure.
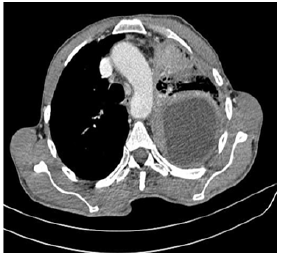
Figure 1: Axial scangraphic section showing a left pleural nodular thickening reaching of 18 mm in maximum thickness associated with a pleural effusion of great abundance responsible for the pulmonary collapse.
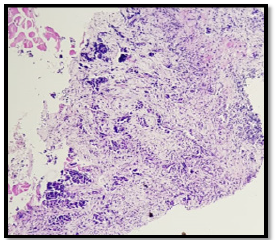
Figure 2: Histological study: HESx200: infiltration of the pleural membrane by malignant tumor proliferation consisting of epitheliomatous cell flows, cord and trabecular structure, sometimes laying out glanduliform structures or acinous formations which are arranged in an endocrine mode in palisades and rosettes, dissociated by a desmoplastic stroma of variable abundance.

Figure 3: Histological study: HESx400: The tumor cells were rounded, polyhedral or fusiform with eosinophilic cytoplasm and ovoid nuclei, sometimes voluminous, nucleoli with rare mitosis figures: less than 2M/10CFG.
Data were analyzed from 10 patients admitted to the infection department identified to be nucleic acid-positive for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in Beijing Ditan hospital on February 27, 2020. The inclusion procedures and criteria were in accordance with the Guidelines for COVID-19 Diagnosis and Treatment (Trial version 7) published by the National Health Commission of the People’s Republic of China. All had a definite history of exposure to the same COVID-19 patient. After admission, the patients would receive empirical antiviral treatment and supportive care followed by consecutive PCR testing and chest CT scan according to a standard protocol. Patients would be discharged from the hospital if they met all of the following criteria: nucleic acid test negative twice in a row; chest CT showed no aggravation; improved symptoms; and normal body temperature for three consecutive days. These patients had returned to the hospital for follow-up visits 2 and 4 weeks after discharge.
Data Collection
Basic information (age, gender, smoking history, and comorbidities) was collected for each patient. Clinical manifestations were recorded, along with disease condition changes. Laboratory test results were compiled, including standard blood counts (absolute white blood cells and lymphocytes), blood biochemistry (alanine transaminase, aspartate transaminase, creatine kinase, creatinine, fasting glucose, Electrolyte), coagulation function, procalcitonin, C-reactive protein, erythrocyte sedimentation rate, and myocardial enzyme spectrum. Additional data collected included medical imaging, treatment regimens.
Data Analysis
Continuous data are expressed as medians and ranges, and categorical data are presented as counts and percentages. The association between two categorical variables was tested with T- Test. All tests were 2-sided with P < 0.05 as significance threshold. The analysis was performed with SPSS 19.
Discussion
Carcinoid tumors are rare neuroendocrine tumors. The most frequent localization is at the digestive tract in 70% of cases, then at the broncho-pulmonary in 10-30% of cases [1]. In the literature, cases at the gallbladder, breast, thymus, larynx, ovary, prostate, and kidney have been described [2-6]. Primary pleural localization is unusual. Few cases have been reported in the literature [4]. The majority of reported cases were related to either metastatic forms or relapses at the pleural level of another primitive [7-9]. Only one retrospective study conducted by Moss and colleagues including 50 patients with carcinoid syndrome. In this study, pleural thickening was found in nine patients, four of whom were primary [10]. There are two types of carcinoid tumors; typical and atypical. Typical carcinoid tumors are good prognosis compared to atypical tumors and rarely metastasize.
The diagnostic of a carcinoid tumor remains the pathological examination, based on histological examination with immunohistochemical complement. In a histological analysis, the diagnosis of the neuroendocrine nature of these tumors can be suspected to morphology; tumor cells contain secretion granules attached to the membrane and to the cytoplasm level and will be confirmed by immunohistochemical analysis in case of the positivity the tumor cells by one or more endocrine markers (Synaptophysin, chromogranin A, CD56) [7].
The treatment of choice of typical carcinoid tumors is based on surgery because it is the only therapeutic modality that seems reasonable to improve the prognosis of patients with these tumors. However, the pleural location of these tumors makes this treatment difficult as it is almost impossible to do carcinological surgery. In cases where surgical excision is not feasible and in metastatic forms, there is no consensus on the best treatment to use in the first line due to the scarcity of these tumors. However, chemotherapy seems to be beneficial in these situations to avoid the rapid progression of the disease and relieve symptoms related to the disease.
Conclusion
Through this work, a very rare case of a typical carcinoid tumor of pleural localization has been reported. Few cases have been found in the literature. These tumors pose a diagnostic problem, hence the interest of a multidisciplinary diagnostic approach to avoid missing out on metastatic disease.
Acknowledgement
None.
Competing Interests
The authors declare that they have no competing interests.
References
- Iglesias M, Belda J, Baldó X, Gimferrer JM, Catalan M, et al. (2004) Bronchial carcinoid tumor: a retrospective analysis of 62 surgically treated cases. Archivos De Bronconeumologia 40(5): 218-221.
- Ogawa H, Nishio A, Satake H, Naganawa S, Imai T, et al. (2008) Neuroendcorine tumor in the breast. Radiation Medicine 26(1): 28-32.
- Anjaneyulu V, Shankar-Swarnalatha G, Rao SC (2007) Carcinoid tumor of the gallbladder. Annals of Diagnostic Pathology 11(2): 113-116.
- Chan E, Sharma A, Strollo D, Lawrence C, Luketich J, et al. (2018) Primary typical carcinoid of the pleura: a rare incidental finding. Chest 2018 154(4): 516-517.
- Piura B, Dgani R, Zalel Y, Nemet D, Yanai-Inbar I, et al. (1995) Malignant germ cell tumors of the ovary: a study of 20 cases. Journal of Surgical Oncology 59(3): 155-161.
- Murali R, Kneale K, Lalak N, Delprado W (2006) Carcinoid tumors of the urinary tract and prostate. Archives of Pathology & Laboratory Medicine 130(11): 1693-1706.
- Liu C, Crawford JM (2005) The Gastrointestinal Tract. Chapter 17. Kumar V, Abbas AK, et al. Eds., (2005) Robbins and Cotran Pathologic Basis of Disease-7th Elsevier Saunders, Philadelphia, USA.
- Townshend AP, Lakshminarayanan B, Ucar AE, Chaudry ZR, Duffy JP (2007) Rare pleural recurrence of typical pulmonary carcinoid tumor 30 years after lobectomy. The Annals of Thoracic Surgey 83(4): 1523-1524.
- Klee H, Vestring T, Bittmann I (2008) Pleural metastases of a typical bronchial carcinoid 7 years after lobectomy. Pneumologie; 62(10): 607-610.
- Moss SF, Lehner PJ, Gilbey SG, Kennedy A, Hughes JMB, et al. (1993) Pleural involvement in the carcinoid syndrome. QJM: An International Journal of Medicine 86(1): 49-53.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.