Case Report 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Recurrent Fever Diagnosis: A Case Disguised as PFAPA Syndrome
*Corresponding author: Cristina Naharro Fernández, Department of Dermatology, Hospital Universitario Marqués de Valdecilla, Santander, Avenida Valdecilla s/n, 39008 Santander. Cantabria. Spain.
Received: September 01, 2020; Published: November 02, 2020
DOI: 10.34297/AJBSR.2020.10.001558
Abstract
Recurrent fever is common in childhood. Differential diagnosis should be conducted in some patients. Autoinflammatory diseases could be responsible in some circumstances but diagnosis is not easy, as different syndromes could appear overlapped. We present a clinical case of a boy with recurrent fever, aphthous stomatitis and growing delay.
Keywords Recurrent fever; Aphthae; Aphthous stomatitis PFAPA syndrome; Familiar Mediterranean Fever; FMF; MEVF
Abbreviations: FMF: Familiar Mediterranean Fever; MEVF: Familiar Mediterranean Fever Gene; PFAPA: Periodic Fever; Aphthous Stomatitis; Pharyngitis; Adenitis
Introduction
Fever is a prevalent symptom in childhood. In the first years of life, a healthy child could have annually up to twelve fever episodes, especially during the cold season. In most cases, these episodes correspond to viral infections. However, in other circumstances, infectious etiology is not responsible, and diagnosis becomes a challenge for the paediatrician.
Differential diagnosis of recurrent fever encompasses neoplastic processes, autoinflammatory diseases and immunodeficiencies.
Clinical case
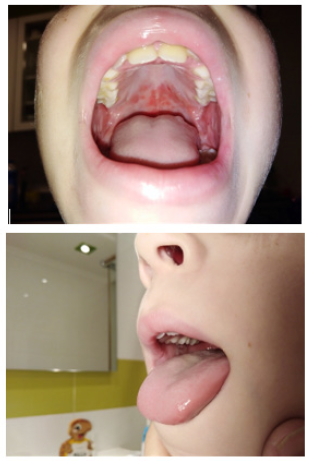
We present the case of a 10-year-old boy with recurrent fever and aphthous stomatitis. He was asthmatic and he received treatment with inhaled corticosteroids. The child had short stature with a delay in bone age of nearly three years. Parents referred previous episodes of fever and aphthae. At the age of nine, he was admitted at the hospital with fever, erosive lesion in mouth and decreased intake that started three days before.
Physical examination highlighted multiple aphthae on oral mucosa, hard palate and soft palate and cervical bilateral lymphade nopathies. Blood count revealed leukocytosis and RCP of 2,9 mg/dl with a maximum during the hospital admission of 8,3 mg/dl. Chest radiograph showed a left lower lobe infiltrate suggesting pneumonia. He received amoxicillin-clavulanic acid and azithromycin. After 72 hours fever disappeared, and oral lesion improved. Serology for herpes simplex virus and bacteria were negative. Mycoplasma pneumoniae IgM and IgG were positives. The patient was discharged with diagnosis of mucositis cause by Mycoplasma pneumoniae.
The patient continued to present additional episodes of fever and oral lesions. A rheumatology study was conducted, showing negative immunodeficiency and autoimmunity. Even though the patient fulfilled the diagnosis criteria for PFAPA, a genetic analysis was requested and the potentially pathogenic mutation p. Lys 695Arg was detected in the gene MEFV. We finally made the diagnosis of FMF in our case. Colchicine was prescribed, and the boy became asymptomatic in a few weeks.
Discussion
For paediatricians, knowing which patients affected of recurrent fever could benefit of the study becomes essential, given the possibility of improving prognosis in some diseases when they are diagnosed and treated correctly. Our patient was initially diagnosed as a mucositis in the context of pneumonia. It seems to be an extrapulmonary manifestation of Mycoplasma pneumoniae and it is characterized for affection limited to the mucous membranes with a good prognosis, especially when antibiotics against Mycoplasma are administered. The real causes are unknown but autoimmune mechanisms have been proposed. This situation is considered as an atypical presentation of Steven Johnson Syndrome (SJS) by some authors, where patients have severe oral mucositis, but they lack typical skin manifestations. This entity was described for the first time in 1967 [1]. And after that, more cases have been reported in literature [2-4].
In our case, successive episodes of fever and aphthae in a patient with growing delay led us to investigate further differential diagnosis. In recurrent fever, clinical features can help paediatricians to guide the diagnosis. Thus, tumoral etiology was discarded because of chronicity of the symptoms. On the other hand, immunodeficiency related syndromes may be suspected in cases of recurrent and severe infections, uncommon microorganisms, dimorphic features, growing delay, bronchiectasis… which not correspond with our patient except for short stature and also analysis was negative. Autoinflammatory syndromes are another possibility in recurrent fever. It includes genetic disorders secondary to mutations in genes involved in the innate immune response: Familial Mediterranean fever (FMF), Tumor necrosis Factor Receptor-associated Periodic fever Syndrome (TRAPS), Haploinsufficiency a20 and Mevalonate Kinase Deficiency. The most common syndrome in relation with recurrent fever is a non-genetic syndrome that includes periodic fever, aphthosis, pharyngitis and adenitis. It is named as PFAPA syndrome and it is a multifactorial disorder.
Marshall et al. [5] described it in 1987. Fever attacks occur typically in intervals of 3 to 8 weeks and are usually accompanied by pharyngitis, aphthae and cervical lymphadenopathy. In most cases, symptoms start in children under five but cases in adults are also described. Fever quickly disappears with one dose of prednisone. This effect is considered as diagnosis criteria. It has a good prognosis tending to resolution in a few years. In children tonsillectomy is considered curative [6]. Diagnosis criteria were described by Thomas et al. [7] and Padeh et al. [8]. Their low specificity is a problem, because these criteria could also be positive in other autoinflammatory monogenic disorders, as clinical manifestation are frequently overlapped.
It is important, to take in account, to identify which patients with positive criteria of PFAPA could be at high risk of having a monogenic autoinflammatory disease. The Gaslini score was created by Gattorno et al. [9] and is used to assess the risk of having a genetic disease in patients with diagnosed PFAPA.
Moreover, a few recent studies have reported that heterozygous variants of the MEFV gene and polymorphisms of MEFV, TNFRSF1A and MVK were found in patients with PFAPA syndrome. Incidence rate of 27-65% MEFV mutations have been reported in some studies [10]. In our case, although the patient met criteria of PFAPA, the growing retard led us to investigate autoinflammatory diseases, and a genetic test was ordered.
FMF represents the most common genetic autoinflammatory disease and even though it is globally present, it is more frequent in Mediterranean countries. Responsible gene named as MEFV (Mediterranean Fever) was discovered in 1997 [11]. It is located on chromosome 16p13.3, and it is comprised of ten exons. Traditionally FMF was considered as a recessive disorder. However, in the last years this hypothesis has been discussed, as some patients with clinical diagnosis of FMF show in genetic analysis heterozygous mutations presenting a dominant behavior [12,13]. FMV is linked to different mutations which can be consulted in INFIVER database.
Treatment with colchicine is recommended in order to reduce the progression to renal failure caused by secondary amyloidosis [14].
It is worth to mention a new disease recently described, the Haploinsufficiency a20. It looks like Beçhet disease, but first symptoms occur in early childhood instead of adulthood. In this case, recurrent fever is common, unlike classical Beçhet. Genetic investigations should target the TNFAIP3 gene, which in our case did not show any abnormality [15]. However, our genetical study revealed a heterozygous MEFV mutations affecting to exon 10 showing that Familial Mediterranean fever should be considered in differential diagnosis of PFAPA. In some patients, genetic screening could help to make diagnosis of autoinflammatory disease with a PFAPA-like presentation. In this hypothesis underlying MEFV gene mutations possibly lead to PFAPA-like clinical presentation in FMF patients.
Furthermore, frequency differences of clinical features between groups suggest that some of the MEFV gene mutations may modify phenotype of PFAPA syndrome. Some studies have tried to find clinical differences related to fever duration, free intervals of symptoms between PFAPA syndromes associated or not with MEFV mutations (Figures 1 and 2).
Conclusion
Advances in genetic testing enable to identify genetic diseases with higher sensibility. Clinical presentation of autoinflammatory syndromes are often overlapped, especially in PFAFA syndrome, where an important rate of mutation in MEFV gene has been detected. We should not forget that FMF could have a PFAPA-like presentation in order to not misdiagnose this entity. Genetic test in selected patients could allow us to make the correct diagnosis in order to improve prognosis of our patients and families.
Conflict of Interest
Authors do not declare any conflict of interest.
References
- Sieber OF, John J, Fulginiti VA, Overholt EC (1967) Stevens Johnson syndrome associated with Mycoplasma pneumoniae infection. JAMA 200: 79-81.
- Schalock PC, Dinulos JGH (2003) Mycoplasma pneumoniae-induced Stevens-Johnson syndrome without skin lesions: Fact or fiction? J Am Acad Dermatol 52(2): 312–315.
- Ravin KA, Rappaport LD, Zuckerbraun NS, Wadowsky RM, Wald ER, et al. (2007) Mycoplasma pneumoniae and atypical Stevens- Johnson syndrome: A case series. Pediatrics 119(4): e1002-e1005.
- Fearon D, Hesketh EL, Mitchell AE, Grimwood K (2007) Mycoplasma pneumoniae infection complicated by pneumomediastinum and severe mucositis. J Peadiatr Child Health 43(5): 403-405.
- Marshall GS, Edward KM, Butter J, Lawton AR (1987) Syndrome of periodic fever. Pharyngitis and aphtous stomatitis. J Pediatr 110: 43-46.
- Vanoni F, Theodoropoulou K, Hofer M (2016) PFAPA syndrome: a review on treatment and outcome. Pediatr Rheumatol Online J 14(1): 38.
- Thomas KT (1999) Periodic fever syndrome in children. J Pediatr 135: 15-21.
- Padeh S, N Brezniak, D Zemer, E Pras, A Livneh, et al. (1999) Periodic fever, aphtous stomatitis, pharingitis and adenophaty syndrome: clinical caracteristics and outcome. J Pediatric 135(1): 98-101.
- Gattorno M, Caorsi R, Meini A, Marco Cattalini, Silvia Federici, et al. (2009) Differentiating PFAPA syndrome from monogenic periodic fevers. Pediatrics 124(4): e721-e728.
- Celiksoy MH, Ogur G, Yaman E, Abur U, Fazla S, et al. (2016) Could familial Mediterranean fever gene mutations be related to PFAPA syndrome? Pediatr Allergy Immunol 27(1): 78-82.
- (1997) Ancient missense mutations in a new member of the RoRet gene family are likely to cause familial Mediterranean fever. The International FMF Consortium. Cell 90(4): 797-807.
- Dode C, Pecheux C, Cazeneuve C, Cattan D, Dervichian M, et al. (2000) Mutations in the MEFV Gene in a Large Series of Patients with a Clinical Diagnosis of Familial Mediterranean Fever. Am J Med Genet 92(4): 241-246.
- Aróstegui Jl (2011) Enfermedades autoinflamatorias sistémicas hereditarias. Reumatol Clin 7(1): 45-50.
- Lachmann HJ, Goodman HJB, Gilbertson JA, Galimore JR, Sabin CA, et al. (2007) Natural History and outcome in systemic AA amyloidosis. N Engl J Med 356(23): 2361-2371.
- Berteau F, Rouviere B, Delluc A, Alice Nau, Rozenn Le Berre, et al. (2018) Autosomic dominant familial Behçet disease and haploinsufficiency A20: A review of the literature Autoimmun Rev 17(8): 809-815.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.