Research Article 
 Creative Commons, CC-BY
Creative Commons, CC-BY
The Mechanism of Radioprotective Effect and Immunoreactive Effect on Vitamin c
*Corresponding author: Yeun-Hwa Gu, Department of Radiological Science, Faculty of Health Science, Junshin Gakuen University, Fukuoka, Japan.
Received: August 25, 2020; Published: September 16, 2020
DOI: 10.34297/AJBSR.2020.10.001505
Abstract
Background and Objective: Vitamin c preparations have a fairly high antioxidant effect, demonstrating increased efficiency for the immune system. Also, since Vitamin c is sensitive to heat, it is less safe to heat.
Methods: Radiation protection from death and stimulating leukocyte recovery by oral administrations consecutively of Vitamin c, 200 mg/kg and 400mg/kg b.w., once a day, before whole-body x-rays irradiation was confirmed by tests with C3H mice, meanwhile, its radioprotective effecs compared to immunological enhancement. Based on the studies of survival, behavior of hematograms, and numbers of lymphocytes, whole body following irradiation, it was demonstrated that Vitamin c was an effective radioprotector. The survival of irradiated mice protected by Vitamin c was significantly increased and statistically higher than that of mice pre-treated with oral administration. After administration of Vitamin c, enhanced CD4 and CD8 cells, numbers of NK cells and CD4 and CD8 cells in mice were found and lymphocytes numbers was higher than in irradiated control group.
Results: Stimulated recovery of leukocyte, lymphocytes, and NK cells counts were observed in mice pre-treated with Vitamin c. All abovementioned results were similar to those in mice protected by Vitamin c, but the protecting actions of Vitamin c on promoting recovery of nucleated cells and leukocyte counts were significantly higher than those of Vitamin c. It could be deduced that the uncertainly radioprotective action against death is induced by a possible process of enhanced regeneration of the leukocyte stem cells due to not only strengthened radioresistance and increased numbers of remained leukocyte cells, but also enhanced post-irradiation repair or promoted proliferation of the leukocyte stem cells.
Conclusion: This effect of Vitamin c may have some therapeutic implications for radiation-induced injuries. We can analyze a result of this study than this thing as follows. We think that CD 4 and CD 8 did immunological enhancement of Vitamin c than helper T cells and suppressor T cell activation from there having been a rise. In addition, we think that indicating the activation of cell-mediated immune responses.
Keywords: Vitamin c; Lymphocyte; NK Cell; Radiation Protection; Immune Activity
Introduction
Hematopoietic cells and peripheral blood cells are known to be sensitive to radiation, and low doses of radiation can cause damage. Radioprotectors are given prior to exposure to ionizing radiation to reduce harmful effects, including radiation-induced lethality [1]. In recent years, natural substances such as SH groups and cysteine have been investigated for their effectiveness in protecting from radiation damage [2]. Among radioprotective compounds, vitamins have been extensively studied. Vitamin c contained in natural substances exerted radioprotection against radiation sickness in experimental animals such as improved survival rate and accelerated recovery of hematopoiesis [3-6]. Moreover, estrogens also ameliorated hematopoietic suppression induced by caner radiotherapy or chemotherapy in the clinic [7,8]. However, the inherent toxicity of these agents in radioprotectors calls for further studies of safer and more effective radioprotectors [9,10]. Vitamin c, a naturally occurring Vitamin c found in nature products [11]. Previous studies have shown that one of the most important natural substances, vitamin C, is not toxic to human health at pharmacological concentrations, acts as an antioxidant and immunity enhancer, and has the potential to block radicals. Is shown. That is, it may improve the secondary immune system [12,13]. As a result, it is gaining more and more attention due to its association with beneficial effects for people with lifestyle-related diseases [14-16]. In addition, Vitamin c is an effective antioxidant, capable of scavenging free radicals and increasing the activity of antioxidant enzymes. Therefore, application to the skin of nude mice prior to radiation exposure may provide protection against UV and radiation [17]. Vitamin c also reduced the frequency of micronucleated reticulocytes and provided cell protection in the reproductive system, thus increasing the survival rate of subletically irradiated mice [18,19]. The purpose of the test reported here was to study the in vivo radioprotection of vitamin C against hematopoietic recovery, which contributes to increased survival of sublethal irradiated mice [20].
Methods
Animals
Male C3H/Hej mice purchased from Japan SLC (Shizuoka, Japan) were used at 7 weeks of age. Mice were housed with controlled lightning (12L: 12D) and food and water were given ad libitum. All mice were acclimated to laboratory conditions for 1 week before experimentation.
Test Material
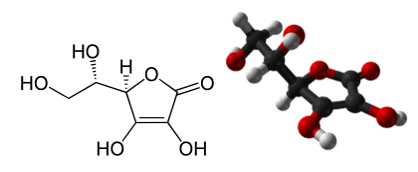
Vitamin c as an active ingredient was contained at the ratio of 6.5mg/g of the naturel product. Vitamin c used in this study is one of the water-soluble vitamins and is chemically L-ascorbic acid (Figure 1).
Radio-Protective Effect
Mice were oral medicated with Vitamin c suspended in physiological saline at a dose of 200mg/kg/day for two weeks at one day intervals. The vehicle-control mice received an equivalent volume of physiological saline. After the final injection, mice were exposed to X-ray radiation. Whole body radiation exposure was carried out at a dose of 2Gy and 8Gy (a dose rate of 1.12 Gy/min) using an X-ray irradiation device (MG226/4.5, Phillips, Inc. Tokyo). Body weight and the number of surviving animals were daily monitored.
Leukocyte and Lymphocyte Counts
Mice were oral medicated with Vitamin c suspended in physiological saline at a dose of 200 mg/kg and 400mg/kg. The vehicle- control mice received an equivalent volume of physiological saline. After the injection, blood samples were obtained from caudal vein into heparinized tubes at given time points for measuring leukocyte and lymphocyte counts using an automated hematology analyzer (Celltac-αMEK-6318, Nihonkouden Co., Ltd. Tokyo).
NK Activity
Mice were oral medicated with Vitamin c suspended in physiological saline at a dose of 200mg/kg for two weeks at one day intervals. The vehicle-control mice received an equivalent volume of physiological saline. Twenty-four hours after the final injection, spleen cells were prepared for measuring NK cell-mediated cytotoxicity by 51C-release from labeled YAC-1 cells. Briefly, 51Cr-labelled YAC-1 cells (2x104 cells) were added to various dilutions of spleen cell suspension in flat-bottomed microplates. The mixtures were incubated at 37℃ for 4 hr in a CO2-incubator. The radioactivity released into the supernatant was counted by a γ-counter, and the magnitude of cytolysis calculated based on the average radioactivity of the control group was defined as NK activity.
Measurement of CD3-, CD4-, and CD8-Positive T Lymphocytes in Peripheral Blood
Lymphocytes were separated by the gravity centrifugation method. Lymphocyte separating solution (5 mL; sodium hypaque, Ficoll 400; specific gravity, 1.0875 ± 0.0005 at 25 °C) was added into a 15-ml sample tube, on to which 5 ml of cell suspension was carefully loaded. After centrifugation at room temperature (15-20 °C) at 500 g for 20 min, plasma in the supernatant was collected to extract lymphocyte subsets. After addition of PBS (pH 7.2, without Ca2+ or Mg2+) supplemented with 10% inactivated FBS (heat-inactivated at 56 °C for 30 min), and red blood cell lysing solution, the mixture was centrifuged at room temperature at 400 g for 10 min. The supernatant was collected and the cells were resuspended and washed twice in PBS containing FBS. Lymphocytes were resuspended in PBS prior to analysis.
Flow cytometry reagents for lymphocyte subset measurement were added into the lymphocyte suspension in PBS, and the mixture was stained for immunofluorescence for about 30 min at 4 °C in a dark room. After the reaction, the solution was rinsed three times with PBS, and CD3, CD4, and CD8 subsets were analyzed by a FACS Caliber flow cytometer (Becton Dickinson). To analyze T lymphocyte subsets, Multicolor Flowcytometry (FCS) System (Santa Cruz Biotechnology Inc.) was employed and CD3-, CD4-, and CD8-positive T lymphocytes in the peripheral blood were counted by three-color flow cytometry using anti CD3-PE-Cy5.5, anti CD4- FITC, and anti CD8-PE.
Statistical Analysis
Significance of the difference in each parameter among groups was assessed by t-test and the Dunnett comparison test following analysis of variance. Values of P<0.05 were considered significant.
Results
Survival Rate of Mice After Irradiation
It followed from results that mortality increased markedly in all irradiated groups and most mice were dead within the 7-14 days following irradiation. As illustrated in (Figure 2), at day 30 following irradiation, survival of irradiated groups, by group, were 8Gy group, approximately 58%; 200mg/kg+8gy group, approximately 77%; 400mg/kg+8Gy group, approximately 86%. It showed that, after Vitamin c pre-treatment, there was a significant enhancement in 8-day survival and about approximately 81% higher than that of 400mg/kg+8Gy group. The survival curve illustrated that, compared with the 8Gy group data, the time to death was significantly shifted to the right for mice pre-treated with Vitamin c after irradiation. Those results demonstrated that Vitamin c possessed highly radioprotective efficacy on prevention of mortality in sublethally irradiated mice and its protecting actions were superior to that of Vitamin c.
Leukocyte Counts
The number of blood leukocytes in normal mice is summarized in (Figure 3). The number of leukocytes increased with time at least up to 24 hr after each repeated dose of Vitamin c in a dose-dependent manner. Statistically significantly higher and increase of leukocyte counts in 200mg/kg group were observed in comparison with control group. In addition, at time of irradiation until day 12, statistically significantly higher and more rapidly recovery of leukocyte counts in 2Gy+200mg/kg group were observed in comparison with 8Gy group.
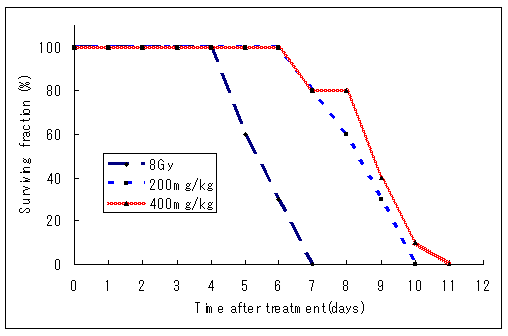
Figure 2: 11 days survival of mice in different groups. Mice received Vitamin c after irradiation exhibited a significant increase in survival 4days after irradiation and the radioprotective action of Vitamin c was more significantly higher than that of vVitamin c. * Statistically significant (p<0.05) from the control group.
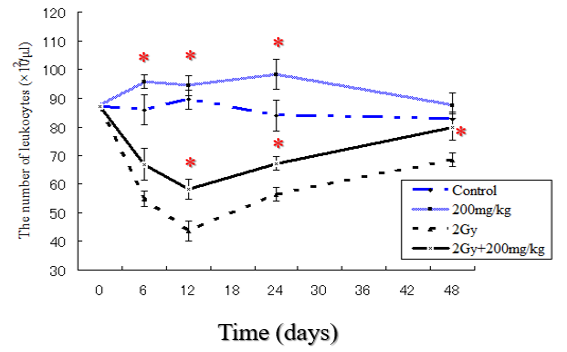
Figure 3: Leukocyte counts on different days after irradiation in mice of different groups. The number of leukocytes was calculated from the preirradiation values taken as 100%. The bars represent standard deviation. * Statistically significant (p<0.05) from the control group.
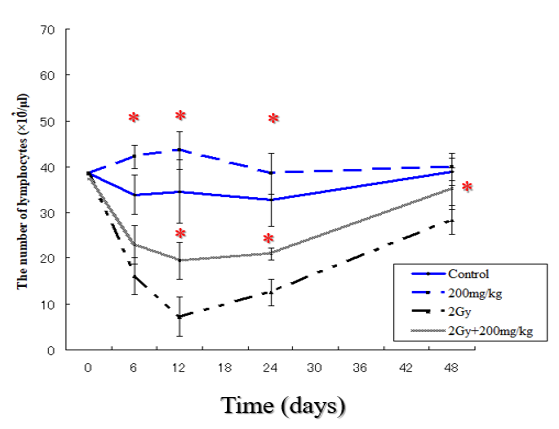
Figure 4: Lymphocyte counts on different days after irradiation in mice of different groups. The number of lymphocytes was calculated from the pre-irradiation values taken as 100%. The bars represent standard deviation. * Statistically significant (p<0.05) from the control group.
Lymphocyte Counts
The number of blood lymphocytes in normal mice is summarized in (Figure 4). The number of lymphocytes increased with time at least up to 24 hr after each repeated dose of Vitamin c in a dose-dependent manner. The lymphocyte counts also showed a similar tendency as in the leukocyte counts. Statistically significantly higher and increase of lymphocytes counts in 200mg/kg group were observed in comparison with control group. In addition, at time of irradiation until day 12, statistically significantly higher and more rapidly recovery of lymphocytes counts in 2Gy+200mg/kg group were observed in comparison with 8Gy group.
NK Activity
NK activity in mice is shown in (Figure 5). Both of the NK activity increased significantly about twofold to threefold after each repeated dose of Vitamin c (200 and 400 mg/kg). Mice were administrated with Vitamin c suspended in physiological saline at a dose of 200 or 400mg/kg for two weeks at one day intervals. The vehicle-control mice received an equivalent volume of physiological saline. Twenty-four hours after the final administration, spleen cells were prepared for measuring NK cell-mediated cytotoxicity by 51C-release from labeled YAC-1 cells. Briefly, 51Cr-labelled YAC- 1 cells (2x104 cells) were added to various dilutions of spleen cell suspension in flat-bottomed microplates. The mixtures were incubated at 37℃ for 4 hr in a CO2-incubator. The radioactivity released into the supernatant was counted by a γ-counter, and the magnitude of cytolysis calculated based on the average radioactivity of the control group was defined as NK activity.
Analysis of T lymphocyte Subsets in Mouse Peripheral Blood
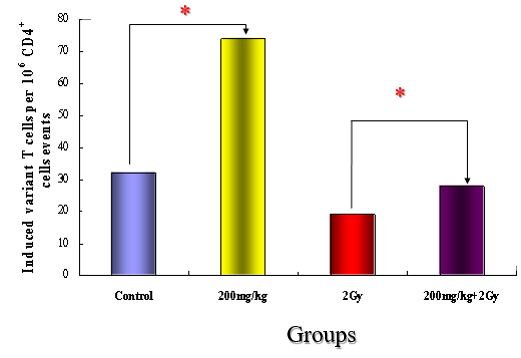
On the cytogram, the lymphocyte fraction was gated, so that CD4-positive (CD3+CD4+) and CD8-positive (CD3+CD8+) cells were counted. First, CD4-positive cells were counted by flow cytometry for comparison. As shown in (Figure 6), the number of CD4-positive cells was increased (by approximately 130%) in only the vitamin C group. Likewise, the number CD4-positive cells increased by approximately 50% in the Vitamin c with irradiation group compared with the irradiation alone group. This indicates that Vitamin c administration increased CD4-postive cells, i.e., helper T cells in the peripheral blood.
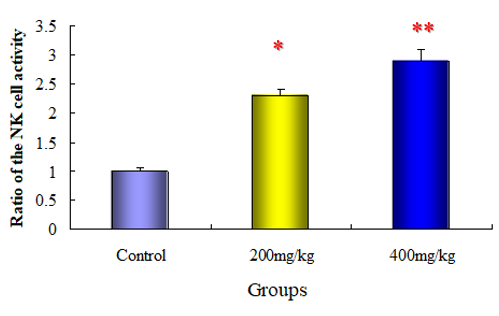
Figure 5: Repeated dose effect of Vitamin c on the NK activity in mice. Groups of ten mice each were subjected to each treatment. Results represent means±S.D. * Statistically significant (p<0.05) from the control group.
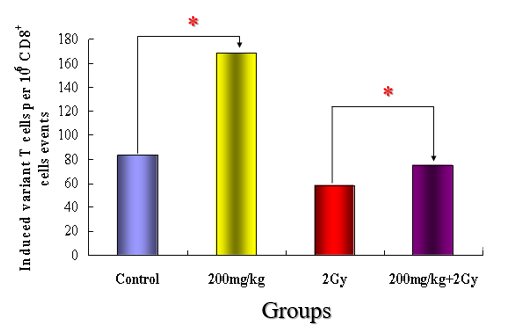
CD8-positive cells were also counted by flow cytometry. As shown in (Figure 7), CD8-positive cells were increased by approximately 100% in the Vitamin c group compared with the control group. Compared with the irradiation alone group, CD8-positive cells were decreased by approximately 30% in the Vitamin c with irradiation group. This indicated that Vitamin c administration increased CD8-positve cells, i.e., suppresser T cells and killer T cells in the peripheral blood. However, in the Vitamin c with irradiation group, irradiation decreased the number of CD8-positive cells.
CD4+ T cells from C3H/Hej mice were immunomagnetically purified and then cell sorted for low CD3 expression. Purity was assessed by FACS analysis. Naive status of cells was confirmed by staining for CD4, CD3, Phenotype of CD4+ T cells after transfer.
Discussion
Vitamin c is well known to exert radioprotective effect and anti- tumor effect in vivo [21-22], and these effects were reproduced in this study. To confirm the elucidative mechanisms by which vitamin C these effects, the number of leukocyte and lymphocyte was monitored as a hemopoietic action. Furthermore, NK activity was measured as immunological parameters. The results of these parameters demonstrated that the radioprotective effect of vitamin C is probably mediated at least in part by a hemopoietic action in irradiated mice since the leukocyte and lymphocyte number was increased by a single dose of Vitamin c. In addition, augmented immunological activity as seen in increased NK activity by Vitamin c seems to play a role in preventing secondary infections associated with irradiation. Natural killer (NK) cells are well known to be associated with cytotoxic effect on various kinds of tumor cells [23-27]. Therefore, increased activity of NK by Vitamin c contributes probably to attenuated tumor growth in tumor-bearing mice. From these, Vitamin c is expected to be promising for the treatment of cancer patients receiving radiotherapy.
One of the major syndromes of hematopoietic system by high dose total-body exposure to ionizing radiation is bone marrow aplasia. It is generally agreed that radiation death in the midlethal dose range is due to impairment of bone marrow hematopoietic function such as leucopenia which will ultimately lead to whole body infection, hemorrhage and even death. Survival brought about by radioprotectors after potentially lethal irradiation is thought to be due primarily to their effect on hematopoietic cells [28]. Accordingly, we used peripheral blood cell counts as indicators of bone marrow function in order to assess the radioprotection of normal tissue, which is critical for survival in this study. The data from our experiments showed that prior oral administrations of Vitamin c to mice with 200mg/kg/day for consecutive 7 days rendered 80% survival in irradiated mice and its survival was significantly higher than that of irradiated control group as well as that of Vitamin c administration. Meanwhile, the prescription Vitamin c ameliorated not only leukocytes but also lymphocytes, CD4 and CD8 decreased by radiation. Stimulating recovery of peripheral hematocytes were also observed in mice pre-treated with Vitamin c, but protecting actions of Vitamin c on leukocytes and nucleated cells were more stronger than those of Vitamin c, although its protection against the decrease of lymphocytes counts was lower than that of Vitamin c. We inferred that Vitamin c, was an effective radioprotector against radiation-induced death by stimulating the rehabilitation of hematopoiesis. These properties have been associated preciously with radioprotection [29-32]. In summary, the results of the current study demonstrated that pre-treated with Vitamin c have some effects on promoting survival and accelerating the rehabilitation of hematopoiesis by protecting bone marrow stem cells and peripheral hematocytes against radiation-induced regression and stimulating proliferation and differentiation of hematopoietic cells. Although our preliminary investigations might Vitamin c information basis for the possibility of Vitamin c to be as a selective radioprotector of hematopoietic system, the evidence was not enough to apply yet and its active constitutions in radioprotection should be further examined individually.
Conclusion
The effects of Vitamin c may act to protect normal cells in the treatment of some radiation cancers against radiation-induced damage. With this study, the results of this study can be analyzed as follows. Radical protection can be expected due to the radical blocking ability of Vitamin c. Furthermore, regarding the protective effects on CD 4 and CD 8, it is considered that the immunological enhancement of Vitamin c was carried out rather than the activation of helper T cells and suppressor T cells. We also believe that it suggests activation of the cell-mediated immune response.
References
- Gu YH, Yamashita T, Yamamoto H, Matsuo T, Washino T, et al. (2019) Plant Enzymes Decrease Prostate Cancer Cell Numbers and Increase TNF-α In Vivo: A Possible Role in Immunostimulatory Activity. International Journal of Food Science.
- Shama M, Kumar M (2007) Radioprotection of Swiss Albino Mice by Myristica fragrans houtt. J Radiat Res 48(2): 135-141.
- Nair CKK, Uma Devi P, Shimanskaya R, Kunugita N, Murase H, et al. (2003) Water soluble vitamin E (TMG) as a radioprotector. In J Exp Bio 41(12): 1365-1371.
- Yamashita T, Kato T, Isogai T, Gu Y, Ma N (2019) Protective Effects of Taurine on the Radiation Exposure Induced Cellular Damages in the Mouse Intestine. Adv Exp Med Biol 1115: 443-450.
- Ma N, Kato T, Isogai T, Gu Y, Yamashita T (2019) The Potential Effects of Taurine in Mitigation of Radiation Nephropathy. Adv Exp Med Biol 1155:497-505.
- Gu YH, Yamasita T, Inoue T, Song JH and Kang KM (2019) Cellular and Molecular Level Mechanisms against Electrochemical Cancer Therapy. Journal of Pathogens.
- Gu YH, Yamasita T, Choi DJ, Yamamoto H, Matsuo T, et al. (2018) The anticancer effect of plant enzymes on mouse breast cancer model. Adv Medi Plant Res 6(4): 70-77.
- Nakamura T, Itokawa Y, Tajima M, Ukawa Y, Cho KH, et al. (2007) Radioprotectve effect of lyophyllum decastes and the effect on immunological functions in irradiated mice. J Tradi Chi Med 27(1): 70-75.
- Gu YH, Park SR, Hasegawa T, Koike M (2000) Antihypertensive effect of Lyophyllum decastes Sing in spontaneously hypertensive rats. Int J Med Mus 3(1): 103-110.
- Gu YH, Hasegawa T, Suzuki I, Hayashi I, Ahn KS, et al. (2000) A study of the radioprotection effect of macro-glucan (β-1-3 glucan) on fetuses of ICR mice. J Ori Med 5(1): 63-70.
- Gu YH, Yamasita T, Kang KM (2018) Subchronic oral dose toxicity study of Enterococcus Faecalis 2001(EF 2001) in mice. Toxi Res 34(1): 55-63.
- Yamashita T, Kato T, Tunekawa M, Gu YH, Wang S, et al. (2017) Effect of radiation on the expression of Taurine transporter in the intestine of mouse. Adv Exp Med Biol 975: 729-740.
- Jin UH, Song KH, Motomura M, Suzuki I, Gu YH, et al. (2007) Caffeic acid phenethyl ester induces mitochondria-mediated apoptosis in human myeloid leukemia U937 cells. Mol Cell Biochem 310(1-2): 43-48.
- Gu YH, Yamashita T, Maenaka T, Itokawa Y, Nakamura T, et al. (2005) Anti-tumor Immunity and Radioprotection effect of Fuscoporia oblique. Med Bio 149(9): 304-318.
- Gu YH, Yamashita T, Oshima M, Takagi Y (2004) Immunological enhancement effect in PASSION FRUIT (MARACUJYA). Med Bio 148(1): 20-25.
- Gu YH, Yamashita T, Oshima M, Takagi Y (2004) The blood estradiol-17β, Testosterone and progesterone of male and female mice change by Lepidium meyenii Walp (MAX-180). Med Bio 148(1): 14-73.
- Gu YH, Choi HJ, Yamashita T, Kang KM, Iwasa M, et al. (2017) Pharmaceutical production of anti-tumor and immune-potentiating enterococcus faecalis-2001 β-glucans: enhanced activity of macrophage and lymphocytes in tumor-implanted mice. Curr Pharm Biotechnol 18(8): 653-661.
- Gu YH, Takebe M (2009) Immunological enhancement effect and radiation protection effect of Fuscoporia oblique. Med Bio 153: 165-175.
- Gu YH, Iwasa M, Iwasa H, Kobayashi K, Itokawa Y, et al. (2007) Radiation protection effect for EF 2001 (Enterococcus Faecalis 2001). Med Bio 151: 289-294.
- Jin UH, Song KH, Motomura M, Gu YH, Kang YJ, et al. (2007) Caffeic acid phenethyl ester induces mitochondria-mediated apoptosis in human myeloid leukemia U937 cells. Mol Cell Biochem 310(1-2): 43-48.
- Gu YH, Itokawa Y, Maenaka T, Yamashita T, Oshima M, et al. (2006) The Blood Estradiol-17β, Testosterone and Progesterone of Male and Female Mice Change by Lepidium meyenii Walp (MAX-180) and Muira Puama. Med Bio 150(4):159-168.
- Kang YN, Maenaka T, Itokawa Y, Yamashita T, Nakamura T, et al. (2006) Embryonic Death Effects of Hyperthermia Induced by RF (Radiofrequency) Waves. Med Bio 150(3): 97-103.
- Gu YH, Fujimiya Y, Itokwa Y, Oshima M, J Choi JS, et al. (2008) Tumoricidal effects of beta-glucans: mechanisms include both antioxidant activity plus enhanced systemic and topical immunity. Nutr Cancer 60(5): 685-691.
- Gu YH, Takagi Y, Nakamura T, Hasegawa T, Suzuki I, et al. (2005) Enhancement of radioprotection and anti-tumor immunity by yeast-derived β-glucan in Mice. J Med Food 8(2): 154-158.
- Lebedev VG, Moroz BB, Vorotnikova TV (1994) Mechanism of radio resistance of the hematopoietic system after treatment with diethylstilbestrol. Radiat Biol Radioecol 34(4-5): 565-571.
- Thompson JS, Simmons EL, Crawford MK, Severson CD (1969) Studies on the mechanisms of estradiol-induced radioprotection. Radiat Res 40(1): 70-84.
- Jenkins VK, Upton AC, Odell TT Jr (1969) Effect of estradiol on splenic repopulation by endogenous and exogenous hemopoietic cells in irradiated mice. J Cell Physiol 73(2): 149-157.
- Rooks WH, Dorman RI (1961) Estrogen radioprotection in mice. Endocrinology 68: 838-843.
- Landauer MR, Davis HD, Kumar KS, Weiss JF (1992) Behavioral toxicity of selected radioprotectors. Adv Space Res 12: 273-283.
- Landauer MR, Castro CA, Benson KA, Hogan JB, Weiss JF (2001) Radioprotective and locomotor responses of mice treated with nimodipine alone and in combination with WR-151327. J Appl Toxicol 21(1): 25-31.
- Patt HM, Straube RL, Tyree EB, Swift MN, Smith DE (1949) Influence of estrogens on the acute X-irradiation syndrome. Am J Physiol 159(2): 269-280.
- Uckun FM, Tuel-Ahlgren L, Song CW, Waddick K, Myers DE, et al. (2000) Radioprotection by antioxidants. Ann NY Acad Sci 899: 44-60.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.