Short Communication 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Use of Osmolytes For Improving Abiotic Stress Tolerance in Brassicaceae Plants: The Case of Proline
*Corresponding author: Walid Saibi, Biotechnology and plant Improvement Laboratory, Centre of Biotechnology of Sfax, University of Sfax, Tunisia.
Received: July 15, 2020; Published: September 25, 2020
DOI: 10.34297/AJBSR.2020.10.001521
Abstract
Osmolytes are low-molecular-weight compounds like amino acids, tertiary sulphonium and quaternary ammonium compounds, sugars and polyhydric alcohols. They are accumulated in the plant cytoplasm so as to balance the osmotic potential of the Na+ and Cl− accumulated in the vacuole. The osmolytes accumulation advantages are that they keep the main physiological functions of the cell active, the induction of their anabolism is controlled by environmental cues, and they can be synthesized at all developmental stages. In addition, osmolytes have crucial functions in protecting subcellular structures and in scavenging reactive oxygen species. This mini-review discusses the use of osmolytes in Brassicaceae plants and their role in osmoregulation and osmoprotection, using the proline as a model. Increasing the osmolyte content in plants is an interesting strategy to improve the growth and yield of crops upon exposure to salinity.
Keywords: Abiotic stress, Brassicaceae plants, Osmolytes, Osmoregulation, Osmoprotection, Proline, Salt tolerance
Introduction
At the beginning, the plants undergo several abiotic stresses such as salinity that is known among the most limiting one [1,2]. Indeed, it is basic to indicate that as much as one-half of the irrigated areas of the world are affected by high level salinity [2,3]. Hence, the one distinctive feature of most plants growing under this type of environmental stress is the accumulation in their cells increased amounts of low-molecular-weight water-soluble solutes. The last described process was called osmotic adjustment. It has been repeatedly inferred, but never proven, that there might be a relationship between salt tolerance and osmotic adjustment [2-7].
Osmoprotectants are small organic molecules with neutral charge and low toxicity at high concentrations that act as osmolytes and help organisms survive extreme osmotic stress [2,8]. They can be classified in three chemical classes such as: (i) betaines and associated molecules, (ii) sugars and polyols and (iii) amino acids. These molecules accumulate in cells and balance the osmotic difference between the cell’s surroundings and the cytosol. In plants, their accumulation can increase survival during various stresses [5].
Intracellular osmoprotectant concentrations are regulated in response to environmental conditions such as osmolarity and temperature via regulation of specific transcription factors and transporters. They have been shown to play a protective role by maintaining enzyme activity through freeze-thaw cycles and at higher temperatures. It is currently believed that they function by stabilizing protein structures by promoting preferential exclusion from the water layers on the surface of hydrated proteins. This favors the native conformation and displaces inorganic salts that would otherwise cause misfolding.
Compatible solutes have a functional role in agriculture. In high stress conditions, such as drought and high salinity, plants that naturally create or take up osmoprotectants show increased survival rates [5]. By inducing expression or uptake of these molecules in crops in which they are naturally not present, there is an increase in the areas in which they are able to be grown. One documented reason for increased growth is regulation of toxic reactive oxygen species (ROS). In high salinity, ROS production is stimulated by the photosystems of the plant. Osmoprotectants can prevent the photosystem-salt interactions, reducing ROS production [1,4,9]. For these reasons, introduction of biosynthetic pathways which result in the creation of osmoprotectants in crops is a current area of research, but inducing expression at significant amounts is currently posing a barrier in this area of research. They are also important for the maintenance of top soil bacteria populations. Desiccation of top soils results in increased salinity. In these situations, the soil microbes increase the concentration of these molecules in their cytoplasm into the molar range allowing them to persist until conditions approve.
Osmolytes and osmoprotectants dynamic: novel strategy for plant tolerance to Abiotic Stress
Osmolytes and osmoprotectants have long been identified as ways to combat abiotic stress [10], as they enable plants to overcome extremely difficult environmental conditions through constant cellular homeostatic monitoring [2,8,9,11]. They mainly consist of sugars, polyols, amino acids, and betaines. Together, they shield plants by exercising a number of physiological responses such as membrane integrity strengthening, enzymatic/antioxidant activity balancing, and water adjustments under various abiotic stresses such as water deficit and salinity. Crop plant cultivation across the globe needs such comprehensive protection strategies against a vast array of ever-changing environmental conditions by developing tolerance process [10].
Proline: The Most Excellent Osmolyte
Proline: definition and structure
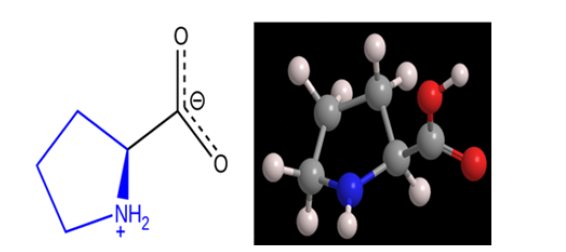
Proline (Pro) is one of the 20 DNA-encoded amino acids [2,8]. This atypical residue is defined, in genetic code, by fore codons such as CCU, CCC, CCA, and CCG [2]. Proline is classified as a non-essential amino acid because of the ability of the human body to synthesize it. It is the unique one among the 20 protein-forming amino acids in that the amine nitrogen is bound to two alkyl groups, thus making it a secondary amine as indicated in Figure 1[2]. Moreover, Table 1 summarizes the most important properties of this atypical amino acid like the systematic IUPAC name, chemical formula, Molar Mass, Melting Point, Solubility and also Acidity.
Proline: a typical example of osmolyte in plants
Osmolytes and osmoprotectants have long been identified as pivotal abiotic stress busters because they help plants overcome extremely harsh environmental conditions by constant cellular homeostatic monitoring [10]. In this case, the best characterized biochemical response of plant cells to osmotic stress is the accumulation of proline [1,4,6]. It is a peculiar amino acid playing an osmoprotectant role in plants subjected to hyperosmotic stresses such as soil salinity [8]. The accumulation of this shrewd amino acid in leaves was observed for the first time by [12] and since then its cardinal function as an osmoprotectant effector under various stress conditions, especially salinity, has been described [8]. The effect of salt stress on plants depends on the salt concentration, the duration of exposure and also the plant genotype [12,13]. The presence of salt in the environment induces water deficit in plants because of the lowered external water potential, while ion toxicity and nutritional alterations disturb ion transport systems [11,13]. The cell proline amount in organs and plant tissues is directed by the interplay of metabolism (anabolism and catabolism) and intra as well as intercellular translocation processes [1,3,8]. We assist in the case of plants, to two different pathways for Proline anabolism [2,8]. The most important one uses the glutamic acid that is transformed to proline by two successive reductions and catalyzed by the pyrroline-5-carboxylate reductase (P5CR) and the Δ1-pyrroline-5-carboxylate synthetase (P5CS), respectively. P5CS is considered as the rate limiting enzyme in proline anabolism [14]. Several findings reported the fact that the feedback regulation of P5CS plays crucial role in controlling the level of the proline under both normal and stress conditions [3,14-16].
Proline metabolism behaviour’s affects plant abiotic stress tolerance
According to our investigations on the proline metabolism behaviour under some of the abiotic stresses such as salinity, we report in this part of mini-review some response elements that can give rise clarification related to the plausible duality between proline metabolism and the salt tolerance acquisition process. Furthermore, some physiological, biochemical and metabolic assessments were monitored. Indeed, we disposed of some mutant and transgenic Arabidopsis thaliana lines named DH2, DH4 and P5CS1-4, as described in [1,3,4].
At this stage, the chlorophyll and the free proline amounts are considered the two crucial related physiological effectors to be monitored in leaves of the described biological matrix (the wild type plant, the durum wheat dehydrin transgenic lines and also the proline metabolic mutant. This choice is not random but is encoded by the fact that the both metabolites (proline and chlorophyll) reflect the physiological state in plants and also their metabolisms have the same anabolic precursor (glutamic acid) [3]. So, we witness a kind of balance in the leaves between the both metabolisms, in which the stimulation of the proline metabolism could modulate the chlorophyll one. Indeed, the proline accumulation in leaves increases from MS (conventional medium), MS-Proline to MS-NaCl- Proline in the case of wild type plant, the durum wheat dehydrin transgenic lines. This fact can be explained by the internalization of the exogenous proline from the culture medium to the plant leaves. However, in the case of the Proline metabolic mutant, very little amount of proline is accumulated in conventional medium and which improves very weakly in MS-Proline, MS-NaCl and MSNaCl- Proline [4]. Those investigations and other’s emphasize the development of tolerance followed through the presence of the durum wheat dehydrin (DHN-5), explained by the better growth of transgenic lines and activation of Proline anabolism in the latter (proline accumulation in plant leaves).
Exogenous proline and salt stress direct the key proline metabolism enzyme
To understand the proline implication in the improvement and/or the acquisition of salt tolerance ability, we performed the monitoring of P5CS activity. The last one is considered as the key enzyme during the proline anabolism process. Indeed, as reported in [3,4], in conventional culture medium (MS), the P5CS activity upgrades in the Durum wheat transgenic context comparing with the wild type plant and this fact seems to be explained by the effect of the presence of the dehydrin (DHN-5) context conferring tolerance acquisition process under salt stress conditions. The improvement of the proline anabolic activity for the two transgenic lines is clear in presence of both salt and exogenous proline. Nevertheless, in the case of the metabolic mutant, it is also clear that the enzymatic activity is too low in the MS medium and is practically absent in MS-Proline, MS-NaCl-Proline and also in MSNaCl medium. According to those findings, we can signal the basic relation-ship existing between the transgenic and the proline metabolic contexts. Based on those investigations and studies, we guess that salt tolerance capability seems to be judged through the functional duality encoded by the proline metabolism under mutational and transgenic contexts.
Causal enzymology, physiological aspects and cell homeostasis supplier salt tolerance in plants
It is known that apart from its significance in the protection against stress conditions, the cell membranes are essential for proper development of the diverse surface structures formed on aerial plant organs. In another hand, membrane remodeling and integrity are function of some of causal enzymological and some of physiological aspects such as the proline, the MDA, the ion leakage and also the monitoring of some phytozymes involved in lipid and cellulose metabolisms.
In this part of mini-review, we report that various studies and investigations deal with the identification of the roles of some physiological parameter’s and also causal enzymological behaviors that may be explain the membrane integrity and also remodeling in case of response of Arabidopsis thaliana lines to salt stress treatment. Hence, we report the potential contribution of phytozymes involved in membrane integrity such as lipases and cellulases and also the impact of the tolerance level on the MDA and also the ion leakage and especially on proline metabolism. Schematic illustration followed in Figure 2 indicates the interaction between the roles played by some of crucial phytozymes, some of physiological parameters and also the cell homeostasis to confer tolerance in plant submitted to salt stress.
The Proline accumulation and the plausible toxicity
As indicated previously, proline accumulation is the most common response of plants against stress. However, external supply of proline in control conditions may be toxic. In addition, there is a debate whether proline or its derived product P5C seem to be the cause of toxicity. Indeed, application of both proline and P5C cause cell death in plants. Various studies support the fact that proline toxicity is mediated by GSA/P5C accumulation. Exogenously applied P5C increases ROS production, reduces growth, and induces a number of stress responsive genes [17,18]. Furthermore, p5cdh overexpression decreased sensitivity to externally supplied proline while p5cdh knockout mutant was hypersensitive to proline [2,8].
At the same case, it is crucial to note that various studies using mutants with impaired proline catabolism support clearly that the proline toxicity is not, or not only, mediated by P5C/GSA. Moreover, proline external concentrations are toxic to Arabidopsis wild-type plants. In pdh-antisense plants that displayed a lower proline catabolism. Proline toxicity was observed at lower proline concentrations than in wild type [19]. pdh-sense transgenic plants, with a higher proline catabolism capacity, displayed wild-type proline sensitivity. Pdh knockout mutants were even more sensitive to proline than PDH-antisense plants, most probably because the mutation results in a more severe inhibition of proline catabolism [2,20-22].
Proline: plausible supplementary roles
In short, the proline accumulation is not only a physiological response to various stresses but is also part of the developmental program in generative tissues like pollen. However, consensus was not achieved on the exact roles of proline accumulation. Classical gain or loss of function strategies could not bring clear answers, probably because proline also displays the essential role of being a protein component. Stresses like drought or salt stress have multiple targets and proline is also believed to play different roles.
Role of proline in regulation of pluripotent cells: The development of cell therapeutics from embryonic stem cells will request technologies which direct cell differentiation to specific somatic cell lineages, in response to defined factors. The first step in formation of the somatic lineages from embryonic stem cells, differentiation to an intermediate, pluripotent primitive ectodermlike cell, can be achieved in vitro by formation of early primitive ectoderm-like cells in response to a biological activity contained within the conditioned medium. The last one fractionation has identified two activities requested for primitive ectoderm-like cell formation, an activity with a molecular weight of 3 kDa and a second, much larger species. Then, the identification step follows that the low-molecular-weight activity seems to be L-proline. An inhibitor of L-proline uptake, glycine, prevented the differentiation of embryonic stem cells in response to the conditioned medium [2,23].
Proline governs the protein self-organization: In this case, it is crucial to note that elastin provides extensible tissues, including arteries and skin, with the propensity for elastic recoil, whereas amyloid fibrils are associated with tissue-degenerative diseases, such as Alzheimer’s. Although both elastin-like and amyloidlike materials result from the self-organization of proteins into fibrils, the molecular basis of their differing physical properties is poorly understood. They prove that elastin-like and amyloidlike peptides are separable based on the backbone hydration and peptide-peptide hydrogen linkage. The analytical studies of diverse sequences, including those of elastin, amyloids, wheat gluten, insect resilin, and spider silks, follow a sill in proline and glycine composition above which amyloid formation is impeded and elastomeric properties become apparent. The predictive ability of this threshold is confirmed by the self-assembly of recombinant peptides in either amyloid fibrils or elastin like. These results support a unified model of protein aggregation in which hydration and conformational disorder are fundamental requirements for elastomeric function [2,8,14,24].
Proline in catalysis: novel mechanistic investigations on the proline-catalyzed aldol reaction: The mechanism of the prolinecatalyzed aldol reaction has stimulated considerable debate and discussion; at least more than five different mechanisms have been discussed [14,20,25]. Discovered 45 years ago (since 1970), the Hajos-Parrish-Eder-Sauer-Wiechert reaction, a prolinecatalyzed intramolecular aldol reaction, represents not only the first asymmetric aldol reaction invented by chemists but also the first highly enantioselective organocatalytic transformation. Inspired by nature’s phenomenal enzymes, which catalyze direct asymmetric aldolizations of unmodified carbonyl compounds, they have recently extended to the first intermolecular variant and to several other reactions including proline-catalyzed asymmetric, α-amination, and intramolecular aldolization reactions [2,20,25].
As similarly as the aldolases, proline catalyzes direct asymmetric aldol reactions between two different carbonyl compounds to provide aldol products with excellent yields and enantioselectivities. In the enzyme classification, there are two classes of aldolase (class I and class II). Early on, it has been speculated that in addition to operating on related substrates, both class I aldolases and Proline may also share a similar enamine mechanism. However, there has been some debate over several mechanistic aspects of the reaction, and a number of alternative models have been proposed. For example, Hajos suggested a mechanism that involves the “activation” of one of the enantiotopic acceptor carbonyl groups as a carbinol amine. At least, the stereochemistry of this model was questioned by Jung soon after its initial proposal. An enamine mechanism was suggested by various groups already since 1970 and 1980. Nonlinearity studies have led to the proposal of a sidechain enamine mechanism that involves two Proline molecules in the C-C-bond-forming transition state, one involved in enamine formation and the other as a proton transfer mediator [2,20].
Conclusion
Although proline has long been considered as the most known osmolyte, recent results follow its multifunctionality in stress adaptation, recovery, and signaling. The compartmentalization of proline metabolism adds to their functional diversification complexity and may explain their importance in various domains. Stabilization of proteins in chloroplast and cytosol protects efficiently the photosynthetic apparatus and also enzymes involved in detoxification of ROS. The enhanced rate of proline biosynthesis in chloroplasts can contribute to the redox balance stabilization and to the cellular homeostasis maintenance. Proline catabolism in the mitochondria is connected to oxidative respiration and administers energy to resumed growth after stress. As well as modulating responses to abiotic and biotic stresses, proline appears to function as a metabolic signal that regulates metabolite pools and redox balance controlling the expression of numerous genes and influencing plant growth and development. The amelioration of plant salt tolerance via engineering proline metabolism is considered now a plausible opportunity that will be explored more extensively. Eventually, the fact that proline can act as a signaling effector and influence defense pathways and regulates metabolic and developmental processes offers additional opportunities for plant improvement.
References
- Saibi W, Zouari N, Masmoudi K, Brini F (2016) Role of the durum wheat dehydrin in the function of proteases conferring salinity tolerance in Arabidopsis thaliana transgenic lines. International journal of biological macromolecules 85: 311-316.
- Saibi W, Feki K, Yacoubi I, Brini F (2015) Bridging between proline structure, functions, metabolism, and involvement in organism physiology. Applied biochemistry and biotechnology 176(8): 2107-2119.
- Saibi W, Feki K, Rihem, Ben M, Brini F (2015) Durum wheat dehydrin (DHN-5) confers salinity tolerance to transgenic Arabidopsis plants through the regulation of proline metabolism and ROS scavenging system. Planta 242(5): 1187-1194.
- Bouazzi H, Feki K, Brini F, Saibi W (2019) Is duality between proline metabolic mutation (p5cs 1-4) and durum wheat dehydrin transgenic contexts a “pacemaker” for salt tolerance process in Arabidopsis thaliana? Acta Physiologiae Plantarum 41(3): 36.
- Bouazzi H, Feki K, Zouari N, Sahnoun M, Brini F, et al. (2019) Causal Enzymology and Physiological Aspects May Be Accountable to Membrane Integrity in Response to Salt Stress in Arabidopsis thaliana Lines. BioMed Research International.
- Di Martino C, Pizzuto R, Pallotta ML, De Santis A, Passarella S (2006) Mitochondrial transport in proline catabolism in plants: the existence of two separate translocators in mitochondria isolated from durum wheat seedlings. Planta 223(6): 1123-1133.
- Ghoulam C, Foursy A, Fares K (2002) Effects of salt stress on growth, inorganic ions and proline accumulation in relation to osmotic adjustment in five sugar beet cultivars. Environmental and experimental Botany 47(1): 39-50.
- Szabados L, Savoure A (2010) Proline: a multifunctional amino acid. Trends in plant science 15(2): 89-97.
- Othman AB, Ellouzi H, Planchais S, De Vos D, Faiyue B, et al. (2017) Phospholipases Dζ1 and Dζ2 have distinct roles in growth and antioxidant systems in Arabidopsis thaliana responding to salt stress. Planta 246(4): 721-735.
- Resham S, Renu B, Ashwani T, Neha H, Ravdeep K, et al. (2014) Osmolyte dynamics: new strategies for crop tolerance to abiotic stress signals. Emerging Technologies and Management of Crop Stress Tolerance 2: 405-430.
- Kaouthar F, Ameny FK, Yosra K, Saibi W, Ali G, et al. (2016) Responses of transgenic Arabidopsis plants and recombinant yeast cells expressing a novel durum wheat manganese superoxide dismutase TdMnSOD to various abiotic stresses. Journal of plant physiology 198: 56-68.
- Kemble A, Macpherson HT (1954) Liberation of amino acids in perennial rye grass during wilting. Biochemical Journal 58(1): 46-49.
- Feki K, Quintero FJ, Khoudi H, Leidi EO, Masmoudi K, et al. (2014) A constitutively active form of a durum wheat Na+/H+ antiporter SOS1 confers high salt tolerance to transgenic Arabidopsis. Plant Cell Reports 33(2): 277-288.
- Rejeb KB, Abdelly C, Savoure A (2014) How reactive oxygen species and proline face stress together. Plant Physiology and Biochemistry 80: 278-284.
- Alonso JM, Anna NS, Liesse TJ, Kim CJ, Chen H, et al. (2003) Genome-wide insertional mutagenesis of Arabidopsis thaliana. Science 301(5633): 653-657.
- Wei C, Cui Q, Zhang XQ, Zhao YQ, Jia GX (2016) Three P5CS genes including a novel one from Lilium regale play distinct roles in osmotic, drought and salt stress tolerance. Journal of Plant Biology 59(5): 456-466.
- Sharma A, Shahzad B, Kumar V, Kohli SK, Gagan Preet SS, et al. (2019) Phytohormones regulate accumulation of osmolytes under abiotic stress. Biomolecules 9(7): 285.
- Liang X, Lu Z, Natarajan SK, Becker DF (2013) Proline mechanisms of stress survival. Antioxidants & redox signaling 19(9): 998-1011.
- Cecchini NM, Monteoliva MI, Alvarez ME (2011) Proline dehydrogenase contributes to pathogen defense in Arabidopsis. Plant Physiology 155(4): 1947-1959.
- Saibi W, Salma A, Khaled M, Ali G (2012) Biocatalysts: beautiful creatures. Biochemical and biophysical research communications 426(3): 289-293.
- Verbruggen N, Hermans C (2008) Proline accumulation in plants: a review. Amino acids 35(4): 753-759.
- Stein H, Zilberstein A, Miller G, Kapulnik Y (2008) Plants tolerant of environmental stress conditions, methods of generating same and novel polynucleotide sequence utilized thereby. Google Patents.
- Washington JM, Joy R, Felquer F, Ana L, Bettess M, et al. (2010) L-Proline induces differentiation of ES cells: a novel role for an amino acid in the regulation of pluripotent cells in culture. American Journal of Physiology-Cell Physiology 298(5): C982-C992.
- Rauscher S, Baud S, Miao M, Keeley FW, Pomès R (2006) Proline and glycine control protein self-organization into elastomeric or amyloid fibrils. Structure 14(11): 1667-1676.
- List B, Hoang L, Martin HJ (2004) New mechanistic studies on the proline-catalyzed aldol reaction. Proceedings of the National Academy of Sciences 101(16): p. 5839-5842.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.