Research Article 
 Creative Commons, CC-BY
Creative Commons, CC-BY
A Mild Case of COVID-19 Infection: An Observational Longitudinal Study 27 Days Post Symptom of Antigen, Antibodies (IgM & IgG), IL-6 and D-Dimer
*Corresponding author: John Bolodeoku, JB Consulting MDP Limited, 1 Bell Street, Maidenhead, SL6 1BU, UK.
Received: January 19, 2020; Published: February 04, 2021
DOI: 10.34297/AJBSR.2021.11.001686
Abstract
A 58-year-old male developed symptoms of COVID-19 infection, fever, aches and pains and general malaise. The next day the symptoms got worse and his breathing became laboured on climbing the stairs. He had a throat and nose swab which tested positive using a COVID-19 antigen test and the diagnosis was confirmed using PCR test. Swabs and blood samples were taken daily till day 27 post onset of symptoms and analysed for COVID-19 antigen, Covid19 antibodies, IL-6, D-dimer, CK-MB, Troponin and Myoglobin. His nasopharyngeal swabs remained positive for 15 days post onset of symptoms, his IgM and IgG seroconversion started on day 10 and 11 post onset of symptoms, respectively. His interleukin-6 levels were elevated on day 1 and was elevated most of the time until day 8 when it returned to normal. His D-dimer concentrations were elevated from day 1 and remained elevated fluctuating up and down till day 24 post onset of symptoms. Other measurements taken during the course included CK-MB, Troponin and myoglobin which were all normal throughout the period of the study. This is the first observation in a mild manifestation of COVID-19 infection in self isolation, showing an elevation of IL-6, and D-dimer levels.
Keywords: Covid 19, D-dimer, Interleukin-6, Antigen, Antibody
Introduction
The clinical manifestations of COVID-19 infection vary from asymptomatic individuals to critically ill individuals. Symptoms associated with the COVID-19 infection include fever, cough, sore throat, general weakness, general body aches and pains, anorexia, diarrhoea, nausea, vomiting and loss of taste and smell. The features of mild illness are fever, cough, or change in taste or small with no dyspnoea [1]. Serological COVID-19 antibody tests to understand seroconversion patterns and antigen methods to detect the presence of the virus in nasal, throat and mouth swabs are becoming important in the understanding of the disease. In addition, prognostic markers such as IL-6, D-dimers etc have also been useful in managing the disease [1]. A longitudinal study of the presence of virus in nasopharyngeal or oropharyngeal swabs using reverse transcription - polymerase chain reaction (RT-PCR) demonstrated dynamic viral shedding as the throat swabs were positive from day 2 to day 20 post onset of symptoms [2]. In another study on mild cases of COVID-19 infection, the earliest time point that the virus was detected in pharyngeal swabs, using RT-PCR, was day 1 of symptoms and the latest time point was 28 days after the onset of symptoms [3]. Longitudinal studies of seroconversion after exposure to COVID-19, show that 81.8% of patients achieve seroconversion within a week [2]. In, another study, IgM seroconversion started in the first week and IgG seroconversion started after week 1 [4]. Increased levels of Interleukin-6 (IL-6) have been observed in 67.9% of COVID-19 infected patients on admission, excessive increased cytokines such as IL-6 lead to cytokine storms, which is an acute severe systemic inflammatory response. In addition, IL-6 concentration has been shown to be an independent risk factor for disease severity and in hospital mortality, its dynamic changes may serve as a potential predictor for lung injury. Functionally, IL-6 has been described as producing a significant dysregulation in serum levels of coagulation factors with increased levels of antifibrinolytic components [5-7]. Increased D-dimer levels have been observed in 74.6% of ill patients on admission and D-dimer levels have been shown to be increased in patients needing critical care support and have a higher incidence of mortality [8-10]. In this observational study, COVID-19 antigen, antibodies (IgM & IgG), IL-6 and D-dimer concentrations were followed for 27 days post symptoms in a mildly COVID-19 infected subject.
Materials and Methods
A 58-year-old male developed symptoms of COVID-19 infection, fever, aches and pains and general malaise on day 0. The next day the symptoms persisted and got worse and his breathing became laboured on walking up the stairs. He had a throat and nose swab which tested positive using a COVID-19 antigen, the diagnosis was confirmed using real time PCR test. For the next 26 days, the following study investigation schedule below was adhered to throughout the self-isolation period at home by the subject:
Investigation schedule of PCR, antigen, antibody (IgG & IgM), IL-6, D-dimer, CK-MB, Troponin and Myoglobin tests
A. COVID-19 antigen - on Day 1,2,3,4,5,6,7,8,9,10,11,12,13,1 4,15,16,17,18,19,20,21,22 and 23 post onsets of symptoms.
B. COVID-19 IgM and IgG antibodies-on Day 1,2,3,4,5,6,7,8 ,10,11,12,13,14,15,16,17,18, 19,20,21,22,23,24,25,26 and 27 post onsets of symptoms.
C. IL-6 - on Day 1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18, 19 and 20 post onsets of symptoms.
D. D-dimer - on Day 1,2,3,4,5,6,7,8,10,11,12,13,14,15,16,17, 18,19,20,21,22,23,24,25,26 and 27 post onsets of symptoms.
E. CK-MB - on Day 3,4,10,11,12,18, 19, 20, 22, 25, 26 and 27 post onsets of symptoms.
F. Troponin - on Day 3,4,10,11,12,18, 19, 20, 22, 25, 26 and 27 post onsets of symptoms.
G. Myoglobin - on Day 3,4,10,11,12,18, 19, 20, 22, 25, 26 and 27 post onsets of symptoms.
Nasopharyneal Swabs
A nasopharyngeal swab was taken from both nostrils and treated as per manufacturers instruction on Day 1,2,3,4,5,6,7,8,9,1 0,11,12,13,14,15,16,17,18, 19 and 20 post onsets of symptoms.
Whole blood samples
Capillary blood (100-150uL) samples were collected on days of analysis (Day 1,2,3,4,5,6,7,8,10,11,12,13,14,15,16,17,18, 19 and 20 post onset of symptoms) using finger prick collected into lithium heparin tube (microvette).
COVID-19 antigen, COVID 19 IgM and IgG antibodies, IL-6 and D-dimer concentrations
Were estimated using the Boditech iCHROMA device and reagents.
iCHROMA™ Method Principle
The test uses a sandwich immunodetection method; fluorescence labelled conjugates in a dried detection buffer binds to antibody in sample (human whole blood/serum/plasma), forming antibody-antigen complexes, and migrates onto nitrocellulose matrix to be captured by the other immobilised anti-human (target) test strip [5]. The more antigen-antibody complexes lead to stronger fluorescence signal by the detector which is processed by the iCHROMA™.
Results
Symptoms
The symptoms started on day 0, fever, aches and pains and general malaise, increasing in intensity over the following days (1,2,3,4,5,6 and 7) during which there was loss of taste and shortness of breath. However, a lingering cough, occasional runny nose and mild shortness of breath persist to day 20 post onset of symptoms. Average oxygen saturation throughout study remained at 97%.
Medication
The subject took paracetamol on days 1 – 7 post onset of symptoms and stopped once the symptoms improved.
COVID-19 Antigen
The nasopharyngeal swabs were positive on Days 1,2,3,4,5,6,7, 8,9,10,11,12,13,14,15, and became negative on day 16.
COVID-19 IgM Antibody
The cut off indices of the blood samples on Days 1,2,3,4,5,6,7 and 8 were <0.1 and were interpreted as negative for the IgM antibodies [6]. On day 10 the samples cut off index rose to 0.4, rising to 0.8 on day 11, it finally got to 1.3 on day 12 and was interpreted as positive. The cut off index continued to rise to 3.4 on day 20 post onset of symptoms and plateaued (Figure 1a).
COVID-19 IgG Antibody
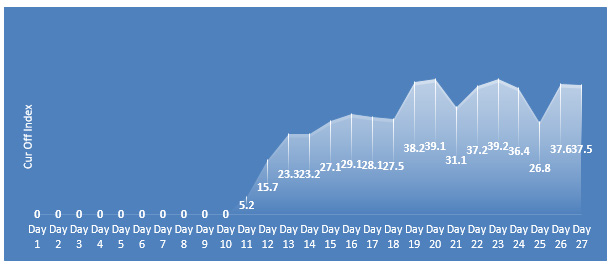
The cut off indices of the blood samples on Days 1,2,3,4,5,6,7, 8 and 10 were 0 and were interpreted as negative for the IgG antibodies. On day 11 the samples cut off index rose to 5.2 and interpreted as positive, rising to 39.1 on day 20 post onset of symptoms and plateaued (Figure 1b).
IL-6 Concentrations
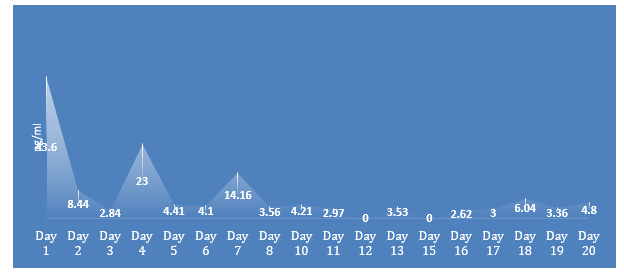
The normal reference range for IL-6 is < 7.0 pg/ml, the IL-6 concentration was elevated on day 1,2,4 and day 7 post symptoms, with normal levels seen on days 3, 5 and 6. On day 8, the IL-6 levels returned to normal (Figure 2a).
D-dimer Concentrations
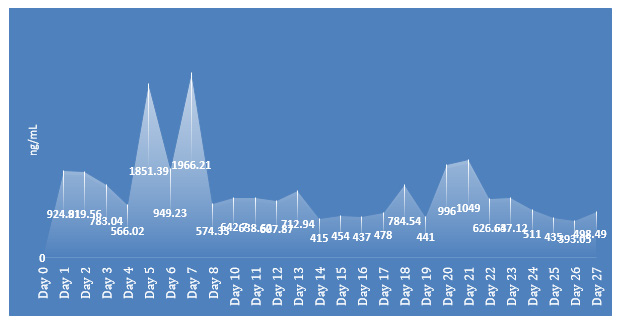
The normal reference range for the D-dimer is < 500 ng/mL, the D-dimer concentrations were elevated on day 1,2,3, 4, 5, 6, 7, 8, 10, 11,12 and 13, returning to normal levels on days 14 – 17 and 19 then going up on day 18 and 20 and finally dropping on day 24 and back to normal levels on Day 25 (Figure 2b).

Figure 1a: Showing Covid 19 antibody (IgM), longitudinal data in subject with mild Covid 19 infection.
Discussion
In this report of a case of a mildly infected COVID-19 subject that had sequential measurements of COVID-19 antigen in nasopharyngeal swabs, COVID-19 IgM & IgG antibodies with IL-6 and D-dimer concentrations measurement in blood [7]. The subject’s respiratory swabs remained positive for 15 days post onset of symptoms. The subject’s IgM seroconversion started on day 10 post onset of symptoms, still rising by day 20. The subject’s IgG seroconversion started on day 11 post onset of symptoms, still rising by day 20 quite consistent with what is seen in the literature. The subject’s IL-6 was elevated on day 1 and was elevated most of the time up to day 8 when it returned to normal. The period it was elevated coincides with the early phase when the subject was having chills and rigors. Interestingly as these symptoms subsided the IL-6 also dropped to normal levels [8]. The subject’s D-Dimer concentrations were elevated from day 1 and remained elevated fluctuating up till day 20 post onset of symptoms. Indicating a hypercoagulable state that has been described [9]. This could be an explanation for the increased incidence of strokes and sudden deaths seen in mild infected COVID-19 patients asked to selfisolate. Other measurements taken during the course included CKMB, Troponin and myoglobin which were all normal throughout the period of the study. This is the first observational study in a subject with mild symptoms of COVID-19 infection showing a development of the “cytokine storm” characterised with the elevation of the pro inflammatory interleukin, IL-6 [9]. In addition, the increase in D-dimers levels indicating the presence of a hypercoagulable environment. It would be important for a larger study to be conducted in the mildly infected COVID-19 individuals measuring cytokines such as IL-6 and D-dimers or other coagulation markers to understand the significance of this observation. In addition, there might be a need to consider initiating anticoagulants in some patients with mild to moderate COVID-19 infection.
Ethical Statement
Consent was provided by the subject to use the data provided.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. JB is a Consultant to Boditech Med Inc, the manufacturers of the iCHROMA device.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
- Gandhi RT, Lynch JB, Rio C (2020) Mild or Moderate Covid-19. N Engl J Med 383(18): 1757-1766.
- Yongchen Z, Shen H, Wang X, Shi X, Li Y, et al. (2020) Different longitudinal patterns of nucleic acid and serology testing results based on disease severity of COVID-19 patients. Emerg Microbes Infect 9(1): 833-836.
- Wölfel R, Corman VM, Guggemos W, Seilmaier M, Zange S, et al. (2020) Virological assessment of hospitalized patients with COVID-2019. Nature 581(7809): 465-469.
- Hou H, Wang T, Zhang B, Luo Y, Mao L, et al. (2020) Detection of IgM and IgG antiboides in patients with coronavirus disease 2019. ClinTransl Immunology 9(5): e1136.
- Liu F, Li L, Xu M, Wu J, Luo D, et al. (2020) Prognostic value of interleukin-6, C-reactive protein, and procalcitonin in patients with COVID-19. J Clin Virol 127: 104370.
- Liu Z, Li J, Chen D, Gao R, Zeng W, et al. (2020) Dynamic Interleukin-6 Level Changes as a Prognostic Indicator in Patients With COVID-19. Front Pharmacol 11:1093.
- Liu B, Li M, Zhou Z, Guan X, Xiang Y (2020) Can we use Interleukin-6 (IL-6) blockade for coronavirus disease 2019 (COVID-19) induced cytokine release syndrome (CRS)? J Autoimmu 111: 102452.
- Yu HH, Qin C, Chen M, Wang W, Tian DS (2020) D-dimer level is associated with the severity of COVID-19. Thromb Res 195: 219-225.
- Zhang L, Yan X, Fan Q, Liu H, Liu X, et al. (2020) D-dimer levels on admission to predict in-hospital mortality in patients with Covid-19. J Thromb Haemost 18(6): 1324-1329.
- Yao Y, Cao J, Wang Q, Shi Q, Liu K, et al. (2020) D-dimer as a biomarker for disease severity and mortality in COVID-19 patients: a case control study. j intensive care 8: 49.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.