Opinion 
 Creative Commons, CC-BY
Creative Commons, CC-BY
3D Volume Rendering Pulmonary Reconstructions in Covid-19 pneumonia at Computed Tomography
*Corresponding author: Francesco Messina, Radiology Unit-Riuniti Hospital, Azienda Ospedaliera Grande Ospedale Metropolitano “Bianchi-Melacrino-Morelli”, Reggio Calabria, Italy.
Received: May 10, 2021; Published: May 19, 2021
DOI: 10.34297/AJBSR.2021.12.001814
Abstract
Covid-19, acronym of the English Coronavirus Disease 19, also known as SARS-CoV-2 acute respiratory disease or coronavirus disease 2019, is an infectious respiratory disease caused by the virus SARS-CoV-2 belonging to the coronavirus family. The Covid-19 Coronavirus infectious pandemic started since December 2019. It caused mainly bilateral interstitial pneumonia in the patients. The heterogeneity of the course and clinical frameworks of the disease represents an important problem both for the acute phase and for the possible complications. Imaging Computed Tomography (CT) had a key-role in the diagnosis and follow-up, allowing an optimal management for the patients [1,2]. Many typical imaging features of the pneumonia were described: bilateral multilobar ground-glass opacity (GGO), with a peripheral or posterior distribution, mainly in the lower lobes; consolidative areas of the lungs; septal thickenings; pleural thickenings; subpleural involvements and pleural effusion [3,4]. The 3D-Volume Rendering Reconstructions at the CT of the lungs represent an important and superior model to evaluate the pulmonary envolvement by Covid-19 and is currently being used in support of “classic” CT reconstruction (MPR, MIP) as a guidance for the evaluation in their complexity of the patients affected by Covid-19 pneumonia (Figure 1-3).
Discussion
Covid-19 pneumonia had an unpredictable clinical course, and varies from patient to patient, until getting to severe disease, with development of acute respiratory failure and organ failure. The use of 3D Volume Rendering pulmonary CT reconstructions had spread during the Covid-19 pandemic. Thanks to their main characteristics (real-time imaging, execution speed, safety and availability), they can be used for the monitoring of the patients with Covid-19 pneumonia, as well as for a fast stratification and triage of symptomatic patients. Therefore, we have introduced in our manuscript a model of reconstructions that can be applied individually for each case of patients with Covid-19 pneumonia, in order to examine them in the shortest possible time and with the best diagnostic accuracy that each CT multislices offers us.
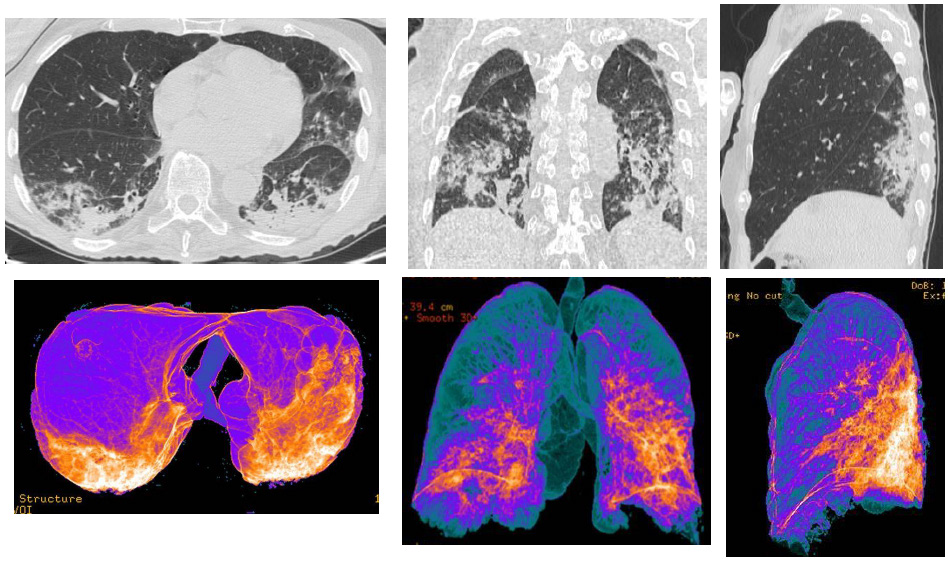
Figure 1: CT patterns of bilateral Covid-19 pneumonia of a 75-year-old male patient. In the upper line, MPR (axial, coronal, sagittal) reconstructions, that showed bilateral typical “ground-glass” areas, with interstitial thickenings. In the line above, Volume Rendering (VRT) “colored” reconstructions, that indicated as blue the areas representing the normal lung parenchyma, while orange/white areas indicate the inflammatory pulmonary involvements.
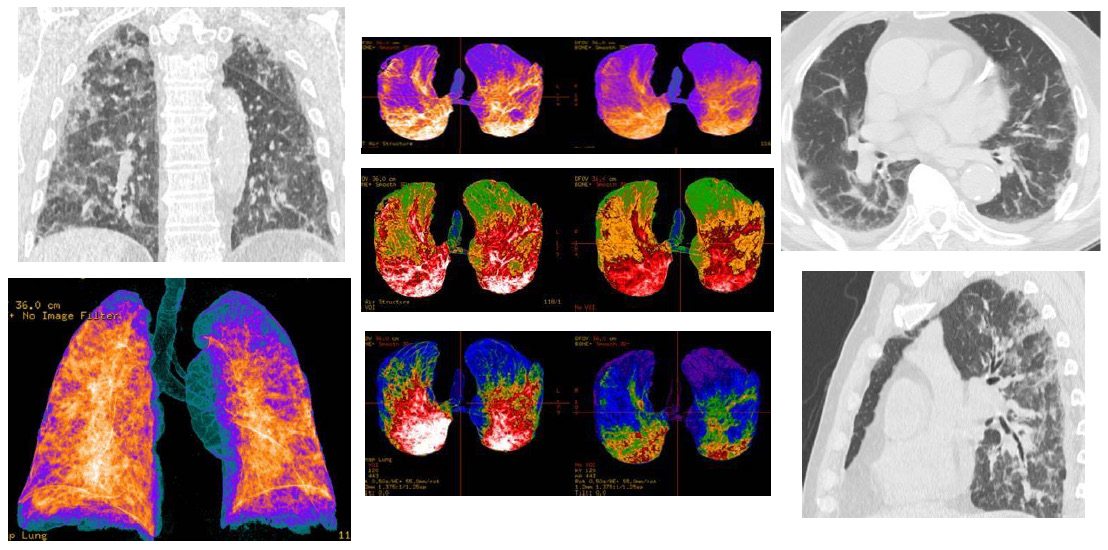
Figure 2: CT patterns of extended bilateral Covid-19 pneumonia of a 80-year-old male patient. MPR (axial, coronal, sagittal) reconstructions showed: bilateral and multifocal typical “ground-glass” areas; bilateral interstitial thickenings, with some consolidations areas; bi-basal pleural effusion. In the Volume Rendering (VRT) “colored” reconstructions, the blue areas represented the normal lung parenchyma (also in the ventral and dorsal areas), while orange/white and red areas indicate the inflammatory pulmonary involvements, with consolidations.
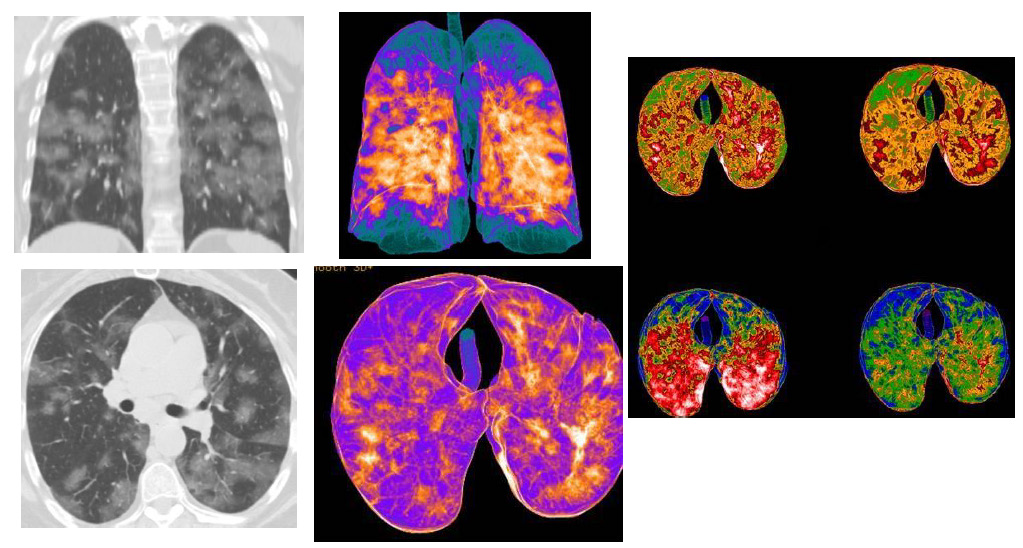
Figure 3: CT patterns of bilateral Covid-19 pneumonia of a 60-year-old female patient. MPR (coronal and axial) reconstructions showed: multiple bilateral consolidations areas, with bilateral interstitial thickenings, and few “ground-glass” areas. In the Volume Rendering (VRT) “colored” reconstructions (three images), the blue/green areas represented the normal lung parenchyma (also in the ventral and dorsal areas), while orange/ white and red areas indicate the inflammatory pulmonary involvements, with consolidations.
Conclusion
Computed Tomography had an important role for the diagnosis, the severity assessment, monitoring and management of Covid-19 infection, thanks to a high sensitivity and rapid execution, thus orienting towards the best management/clinical outcome for the patients.
Acknowledgement
None.
Conflicts of Interest
No conflicts of interest.
References
- Kligman A (2005) The future of cosmeceuticals: An interview with Albert Kligman, MD, Ph.D. Dermatologic Surgery 31: 890-981.
- Schagen SK, Zampeli VA, Makrantonaki E, Zouboulis CC (2012) Discovering the link between nutrition and skin aging. Dermatoendocrinol 4(3): 298-307.
- Di Cerbo A, Laurino C, Palmieri B, Iannitti TA (2015) Dietary supplement improves facial photoaging and skin sebum, hydration and tonicity modulating serum fibronectin, neutrophil elastase 2, hyaluronic acid and carbonylated proteins. J Photochem Photobiol B Biology 144: 94-103.
- Farris P, Krutmann J Li, Y H, Mc Daniel D, Krol Y, et al. (2013) Resveratrol: a unique antioxidant offering a multi-mechanistic approach for treating aging skin. J Drugs Dermatol 12(12): 1389-1394.
- Khavkin J, Ellis DA (2011) Aging skin: histology, physiology, and pathology. Facial Plast Surg Clin North Am 19(2): 229-234.
- Branchet MC, Boisnic S, Frances C, Robert AM (1990) Skin thickness changes in normal aging skin. Gerontology 36(1): 28-35.
- Cadet J, Douki T, Ravanat JL (2015) Oxidatively generated damage to cellular DNA by UVB and UVA radiation. Photochem Photobiol 91(1): 140-155.
- Baek B, Lee SH, Kim K, Lim HW, Lim CJ, et al. (2016) Ellagic acid plays a protective role against UV-B-induced oxidative stress by up-regulating antioxidant components in human dermal fibroblasts. Korean J Physiol Pharmacol 20(3): 269-277.
- Soleymani S, Iranpanah A, Najafi F, Belwal T, Ramola S, et al. (2019) Implications of grape extract and its nanoformulated bioactive agent resveratrol against skin disorders. Arch Dermatol Res 311(8): 577-588.
- Zhuang Y, Wu H, Wang X, He J, He S, et al. (2019) Resveratrol Attenuates Oxidative Stress-Induced Intestinal Barrier Injury through PI3K/Akt-Mediated Nrf2 Signaling Pathway. Oxid Med Cell Longev 7591840.
- Ma Q (2013) Role of nrf2 in oxidative stress and toxicity. Annu Rev Pharmacol Toxicol 53: 401-426.
- Hu Q, Zhang T, Yi L, Zhou X, Mi M, et al. (2018) Dihydromyricetin inhibits NLRP3 inflammasome-dependent pyroptosis by activating the Nrf2 signaling pathway in vascular endothelial cells. Biofactors 44(2): 123-136.
- Saw CL, Yang AY, Guo Y, Kong AN (2013) Astaxanthin and omega-3 fatty acids individually and in combination protect against oxidative stress via the Nrf2-ARE pathway. Food Chem Toxicol 62: 869-875.
- Fisher G J Varani J, Voorhees JJ (2008) Looking older: fibroblast collapse and therapeutic implications. Arch Dermatol 144(5): 666-672.
- Varani J, Dame MK, Rittie L, Fligiel SE, Kang S, et al. (2006) Decreased collagen production in chronologically aged skin: roles of age-dependent alteration in fibroblast function and defective mechanical stimulation. Am J Pathol 168(6): 1861-1868.
- Watanabe-Kamiyama M, Shimizu M, Kamiyama S, Taguchi Y, Sone H, et al. (2010) Absorption and effectiveness of orally administered low molecular weight collagen hydrolysate in rats. J Agric Food Chem 58(2): 835-841.
- Sontakke SB, Jung JH, Piao Z, Chung HJ (2016) Orally Available Collagen Tripeptide: Enzymatic Stability, Intestinal Permeability, and Absorption of Gly-Pro-Hyp and Pro-Hyp. J Agric Food Chem 64(38): 7127-7133.
- Postlethwaite AE, Seyer JM, Kang AH (1978) Chemotactic attraction of human fibroblasts to type I, II, and III collagens and collagen-derived peptides. Proc Natl Acad Sci USA75(2): 871-875.
- Papakonstantinou E, Roth M, Karakiulakis G (2012) Hyaluronic acid: A key molecule in skin aging. Dermatoendocrinology 4(3): 253-258.
- Fu H, You S, Zhao D, Quan An, Zhang J, et al. (2021) Tremella fuciformis polysaccharides inhibit UVA-induced photodamage of human dermal fibroblast cells by activating up-regulating Nrf2/Keap1 pathways. J Cosmet Dermatol.
- Bollag WB, Aitken L, White J, Hyndman KA (2020) Aquaporin-3 in the epidermis: more than skin deep. Am J Physiol Cell Physiol 318(6): C1144-C1153.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.