Research Article 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Migration of OxLDL+ Macrophage Phenotypes from Pericoronary Adipose Tissue to Coronary Intima: A Possible Mechanism for Human Coronary Atherosclerosis
*Corresponding author: Yasumi Uchida, Japan Foundation for Cardiovascular Research, 2-30-17, Narashinodai, Funabashi, 274-0063, Tel: +81-47-462-2159, Japan
Received: March 20, 2020; Published: March 30, 2021
DOI: 10.34297/AJBSR.2021.12.001750
Abstract
Background: We previously found that oxidized low-density lipoprotein (oxLDL), a core atherogenic protein, is stored in human pericoronary adipose tissue (PCAT) and macrophages residing in PCAT accumulate oxLDL. Here, we aimed to identify whether and how the macrophage phenotypes transport oxLDL from PCAT to the coronary intima.
Methods and results: Coronary arteries and their surrounding PCAT were excised from human autopsy subjects and examined using immunohistochemical techniques to investigate macrophage-mediated oxLDL transport from PCAT to the coronary intima. OxLDL+CD68+ and oxLDL+CD206+ macrophages were observed in PCAT of both normal coronary segments (normal group) and adjacent coronary segments with plaques (plaque group). External elastic lamina (EEL) was loosened, or fragmented and internal elastic lamina (IEL) was disrupted in the plaque group, and oxLDL+CD68+ and oxLDL+C206+ macrophages extending pseudopod forward were frequently observed to pass through these portions into the intima. OxLDL+CD11c+ macrophages were not found in PCAT, EEL and the media but were found in disrupted IEL and plaques.
Conclusions: The results suggest that CD206+ or CD68+ macrophages transport oxLDL from PCAT into the media and they and CD11c+ macrophages transformed at the site of IEE into plaque via loosened or fragmented EEL and disrupted IEL and could participate in initiation and acceleration of human coronary atherosclerosis.
Keywords: Human coronary plaques, Immunohistochemical staining, Macrophage phenotypes, Oxidized low-density lipoprotein, Pericoronary adipose tissue
Introduction
It is a general belief that monocytes and low-density lipoprotein (LDL) enter from the lumen into the arterial wall, and while the former become macrophages, the latter become oxidized- LDL (oxLDL),a core pro-atherogenic protein; subsequently macrophages accumulate oxLDL in themselves and play an important role in the initiation and progression of atherosclerosis [1-4]. However, this process is largely dependent on animal experiments and it is not known whether it is the sole mechanism underlying atherosclerosis development in humans. Previously, we found that oxLDL is stored in human pericoronary adipose tissue (PCAT) and accumulated in macrophages residing in the PCAT [5]. However, whether macrophage acquire oxLDL from PCAT and through what route(s) are oxLDL transported to the intima remained unclear in our previous study. Thus, the present ex vivo study aimed to examine which of the macrophages namely those expressing CD68 (a nonspecific marker of macrophage), CD11c (a marker of atherogenic M1-macrophage) [6,7] or CD206 (a marker of anti-inflammatory and antiatherogenic M2-macrophage) [6], participate in this transport process and the routes they follow using immunohistochemical techniques and by excising the normal coronary segments (normal group) or segments containing plaques (plaque group) together with their surrounding PCAT from human autopsy subjects who had suffered from ischemic heart disease.
Methods
Immunohistochemical Studies of Excised Human Pericoronary Adipose Tissue (PCAT) and Its Adjacent Coronary Artery
Ethics
This ex vivo study was conducted after obtaining approval from the Ethical Committees of the Japan Foundation for Cardiovascular Research, Funabashi-Futawa Hospital, Chiba-kensei Hospital and Toho University, and after obtaining written informed consent from the families involved regarding the use of excised coronary artery and its surrounding adipose tissue for histological studies to clarify the mechanisms underlying atherosclerosis [8-11].
Subjects
The proximal to middle segments of coronary arteries (7 left anterior descending arteries and 7 right coronary arteries) and the surrounding adipose tissue were carefully excised from 7 autopsy cases of patients with coronary artery disease who had died at Funabashi-Futawa Hospital or Toho University Medical Center Sakura Hospital and obtained informed consent from the concerned families between April 1, 2016 and March 31, 2018 [61.0 ± 3.1years (mean±SD); 2 females and 5 males]. Three had acute myocardial infarction, two had an old myocardial infarction, and the remaining two patients had angina pectoris. Cause of their death: pneumonia (2), congestive heart failure (1), diabetic nephropathy (1), cerebral infarction (1), visceral carcinoma (1), sudden death (1)] (Table 1).
Classification of coronary plaques and normal segments by histology
Percentage (%) stenosis was calculated by planimetry using the ring segments by the formula: % stenosis= (area within internal elastic lamina - lumen area/ area within internal elastic lamina) x 100.
Selection of plaques and normal segments
A 4 – 5 -mm long section of proximal or middle segments of the anterior descending coronary artery or right coronary artery and their surrounding PCAT were isolated by transecting its proximal and distal ends at the shorter axes. The normal segments and those with plaques were excised (one segment from each subject). Thus, 7 normal segments (normal group) and 7 segments with plaques (plaque group) were obtained and used for further examination (Table 1). They were embedded in O.C.T. Compound (Sakura Finetck USA Inc., Torrance, CA) before being stored at -20○ C. Before embedding these segments, a 0.5 mm section was obtained from each segment and fixed with 5% glutaraldehyde solution for electron microscopic study.
Immunohistochemical staining of oxldl and macrophage phenotypes
Single immunohistochemical staining: All plaques and normal segments with their surrounding PCAT, which had beenv stored at -20°C, were cut into successive 10 μm sections on a cryostat (Tissue Tec 3D, SakuraFinetec Japan, Tokyo). Such relatively thick and frozen sections were used to prevent leaking from the PCAT. Sections were fixed with 4 % paraformaldehyde for 7 min at 4°C, and incubated with a mixture of 1% hydrogen peroxide in methanol for 30min. Successive sections were processed at first by single immunohistochemical as follows: oxLDL, CD68 for nonspecific macrophages [12], CD11c for M1-inflammatory and atherogenic macrophages [13] and CD206 for M2- anti-inflammatory and antiatherogenic macrophages [14].
Double immunohistochemical staining: Then, double immunohistochemical staining was performed as follows: oxLDL with CD68 (oxLDL+CD68+) [12], oxLDL with CD11c (oxLDL+ CD11c+) and oxLDL with CD206 (oxLDL+ CD206+).
The antibodies used for immunohistochemical staining are as follows:
a. For oxLDL, anti-oxLDL-antibody orb 10973; rabbit polyclonal, which reacts with human oxLDL; Biorbyt Ltd, Cambridge, UK [12].
b. For CD68, anti-CD68 antibody mouse monoclonal NCL-CD 68- KP1, Leica Biosystems Newcastle Ltd, Newcastle, UK [12].
c. For CD11c, an anti-CD11c antibody (ab52632, rabbit monoclonal (EP1347Y), reacts with human CD11c, Abcam Ltd).
d. For CD206, a human MMR/CD206 antibody, source polyclonal goat IgG, immunogen mouse myeloma cell line NSO-derived recombinant human MMR/CD206, R&D Systems, Minneapolis, MN.
Sections were incubated with anti-CD68, anti-CD206 or anti- CD11c antibodies for 60 min. Subsequently, the sections were incubated with anti-mouse Alexa555 (Alexa Fluoro555 goat antimouse IgG, Code A21422, Molecular Probe Ltd, CA, USA) for 30 min to emit a red fluorescence for CD68, CD206 or CD11c. The same sections were incubated with the anti-oxLDL antibody for 60 min, and then with anti-rabbit FITC (FITC conjugated Affeini Pure goat anti-rabbit IgG, Code 111-095-003, Vector Laboratories Inc, Burlingame, CA, USA) for 30 min to emit a green fluorescence for oxLDL. Finally, the section was reacted with DAPI (4’,6-diamidino- 2-phenylindole; Life Technologies Carlsbad, Carsbad, CA, USA) to emit a blue fluorescence for cell nuclei [15].
Identification of macrophages by double immunohistochemical staining
Fluorescein and Alexa 555 stain elastin which is contained in EEL and IEL in human vascular wall. When fluorescein and oxLDL or Alexa 55 and oxLDL in combination are used for staining oxLDL+ macrophages, not only oxLDL+ macrophages and EEL and IEL are stained. So, the cells amoeboid configuration which contain nucleus were considered as oxLDL+ macrophages.
Stained sections were photographed separately or merged with one another using a microscope (IX70, Olympus Co, Tokyo, Japan) connected through an ICCD camera (DP 73, Olympus Co) to CellSens Standard (Olympus Co). For fluorescence imaging, a 460 nm bandpass filter (BPF) and a 510 nm band absorption filter (BAF) for the green fluorescence of oxLDL, a 555 nm BPF and a 575 nm BAF for the red fluorescence of macrophages, and a 345 nm BPF and 420 nm BAF for the blue fluorescence of cell nuclei as previously reported [16].
Microscopic observation of pcat and coronary artery after immunohistochemical staining
Density of dotted OxLDL deposits and macrophage phenotypes: Using single Immunohisto Chemical staining preparations, histological observations of oxLDL deposits, CD68+, CD206+ or CD11c+ macrophages was performed by a microscope (IX 70, Olympus Co., Tokyo, Japan). Densities of oxLDL depositions or macrophage phenotypes in PCAT, adventitia, media, and intima was compared between the normal and plaque groups.
Incidence of OxLDL-containing (OxLDL+) macrophage phenotypes in PCAT, media and intima: Using double immunohistochemically stained preparations, the frequency of oxLDL occurrence (%) in macrophage phenotypes was examined in PCAT, media or intima and compared between the normal segment and plaque groups.
Microscopic observation of EEL and IEL
EEL and IEL exhibit a strong fluorescence when stained with fluorescein. One section from each sample was stained with fluorescein, after which EEL and IEL were examined using fluorescent microscopy. Normal EEL is composed of tight continuous elastin fibers that form a band. In case any loosening or fragmentation was noted, EEL was defined as loosened or fragmented EEL, and the number of loosened or fragmented portions in the circumference of EEL was compared between the normal and plaque groups.
IEL is composed of elastin plates arranged in series connected side by side with thin filaments. In a normal coronary artery, the distance between the plates is approximately ≦5 m [8]. Therefore, widening of the distance by 10 μm, fractured or decaying portions of IEL were defined as disruption. The number of disrupted portions across the entire circumference of IEL was compared between the normal and plaque groups.
The Number of OxLDL+ Macrophage Phenotypes Passing through EEL or IEL:
The number of oxLDL-containing macrophage phenotypes passing through the entire circumference of EEL or IEL was counted and compared between the normal and plaque groups. Macrophages are amoeboid cells and extend pseudopod toward the direction of crawling. Therefore, pseudopod side was considered as the direction of their crawling.
Electron microscopic study
Because macrophages are ameboid in shape, cells with an amebic configuration and passing through IEL were examined using an electron microscope (InTouchScopeTM, JSM-IT200, Nihondenshi Co., Tokyo, Japan). Pseudopod was defined as a thin and low-density protrusion from the body.
Statistical analysis
Fisher’s exact test was used for statistical analysis of data. The data obtained were expressed as mean± standard deviation (SD), A p value of < 0.05 was considered to be statistically significant.
Because of a large number of comparisons, Bonferroni correction was used [17].
Results
OxLDL in PCAT
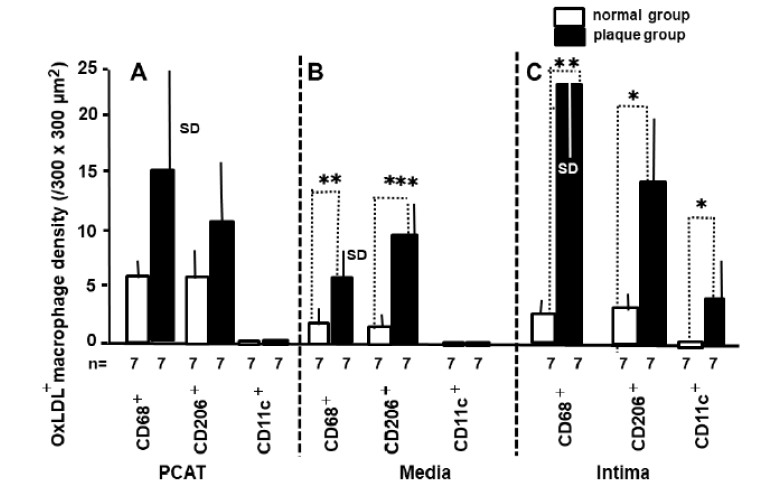
Figure 1: Oxidized Low-density Lipoprotein (OxLDL) and Macrophage Phenotypes in Pericoronary Adipose Tissue (PCAT), Media and Intima.
Figure 1a: OxLDL and Macrophage Phenotypes in PCAT OxLDL is contained in adipocyte cytoplasms (AC; arrow in A). Dotted oxLDL is observed in the interstitial space between the adipocytes (arrowhead in A). CD68+and CD206+ but not CD11c+ macrophages were observed in the interstitial space (arrowhead in B, C, D). The macrophages co-insides with the dotted oxLDL (arrowhead in E – E-2). Scale bars= 5 μm.
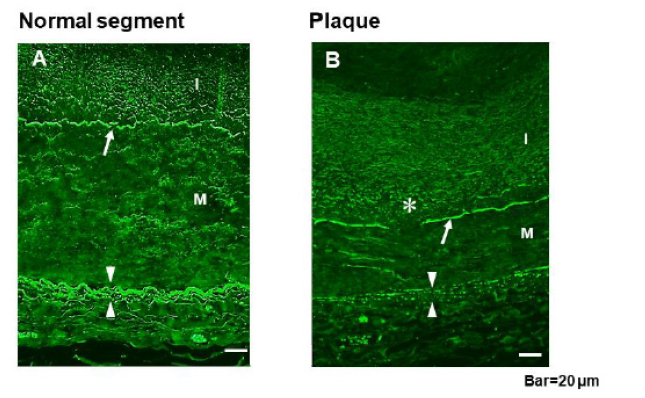
Figure 2: OxLDL+ CD68+ Macrophage in the Border Zone of PCAT and Adventitia OxLDL+CD206+macrophage phenotype in the border zone of PCAT and adventitia (arrows in A – A-2) and in the adventitia (Ad) (arrows in B–B-2). AC: adipocyte. EEL: external elastic lamina. Scale bar=5 μm.
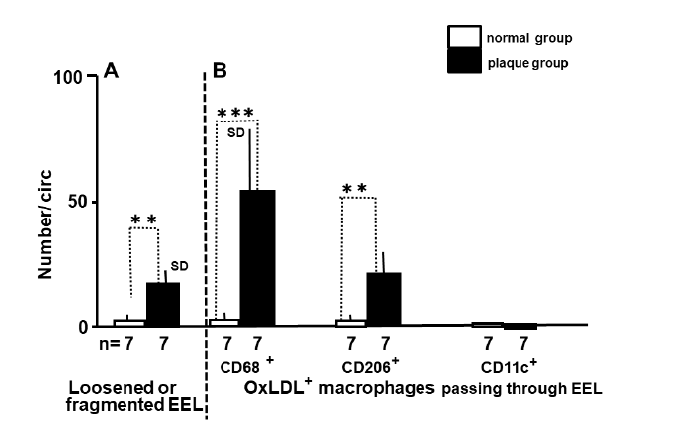
Figure 3: OxLDL+ Macrophage Phenotypes in the Media and Intima. An OxLDL+CD68+ macrophage in the media, showing eel-like configuration (arrows in A – A-2). Arrowheads: pseudopod. An oxLDL+CD11c+ macrophage in the intima (arrows in A – A-2). Arrowheads: OxLDL- CD11c+ macrophage. Scale bars= 5 μm.
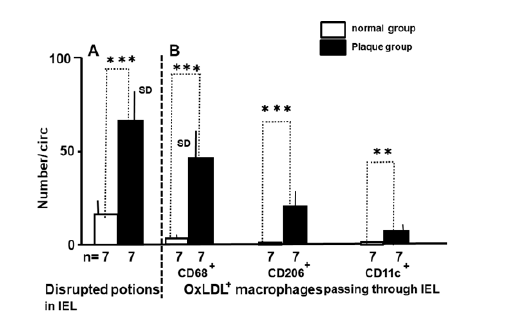
Figure 4: Macrophage Density in PCAT, Media and Intima n: number of samples examined. One normal and plaque samples respectively was obtained from each autopsy subject. A. No significant difference in density of oxLDL+ CD68+ and oxLDL+ CD206+ macrophages in PCAT is observed between normal group and plaque group. OxLDL+CD11c+ macrophage is not observed in PCAT in both the groups. B. CD68+ and CD206+ macrophages density in the media is significantly higher in the plaque group than in the normal group. OxLDL+CD11c+ macrophage is not observed in the media. C. The density of oxLDL+CD68+, oxLDL+ D206+ and oxLDL+ CD11c(+) macrophages in the intima (plaque) is significantly larger in plaque group than in normal group. D. SD: standard deviation. * p<0.05. ** p<0.01. *** p<0.001.
Discussion
OxLDL+ Macrophage Phenotypes in PCAT
Storage of oxLDL was observed in PCAT not only in the plaque group but also in the normal group, indicating that oxLDL storage begins before atherosclerosis development. Percentages of oxLDL+CD68+ and oxLDL+CD206+ macrophages in PCAT were not different between the normal and plaque groups, indicating that oxLDL containing capacity of these macrophages was not influenced by the presence or absence of plaques in the adjacent coronary intima.
OxLDL+ Macrophage Phenotypes in the Intima
In the plaque, the percentage of oxLDL+ macrophage phenotypes was significantly higher than that in the intima of normal group. Their accelerated migration through the EEL and IEL is considered to be the reason for this difference.
Mechanism(s) for Damages in EEL and IEL
Fragmented EEL and disrupted IEL were frequently observed in the coronary segments with plaques (plaque group). There are at least three possible mechanisms for such changes:
a) 1). Plaques induce distension of EEL or IEL and subsequently damage them. In a normal coronary artery, IEL is composed of elastin plates in series, which are connected side by side with thin filaments, and the space between the plates is within 5 μm [18]. Probably, the filaments connecting IEL plates were disrupted and the plates were separated owing to plaque distension.
b) 2). Because EEL and IEL are composed mainly of elastin, elastases that were diffused from the lumen or adventitia eventually damaged EEL and IEL [19].
c) Macrophages excreted cytokines during invasion and caused EEL and IEL damage [20,21].
Migration of oxLDL+ Macrophage Phenotypes through EEL and IEL
The incidence of invasion of oxLDL+ macrophage phenotypes through EEL and IEL increased in the plaque group, suggesting that their influx into the intima contributed to plaque formation. EEL and IEL lost their function as a mechanical barrier, and may have enhanced macrophage migration.
Direction of crawling of oxLDL+ macrophages
Ameba and amoeboid cells such as white blood cell and macrophages protrude a pseudopod forward, retracts the pseudopod, change body shape and crawls, indicating that the pseudopod shows the direction of crawling [22-25]. In this study, oxLDL+ macrophage phenotypes in the loosened EEL or disrupted IEL protruded their pseudopod toward the media and toward the intima, respectively, strongly suggesting that they crawled toward the media and then to the intima.
Macrophage transformation
There are a number of macrophage phenotypes. Among them, M1-macrophages(CD11c+ macrophages) are considered as atherogenic and M2-macrophages (CD206+ macrophages) are considered as anti-atherogenic, mostly based on the findings in animals or cultured cells [26]. In our study, CD11c+ macrophages were not found in PCAT, adventitia and media, but were found at the site of disrupted IEL, suggesting that CD68+ and/or CD206+ macrophages were switched over to CD11c+ macrophages while passing through IEL. In the present study, not only CD11c+ but also CD206+ macrophages frequently contained oxLDL. This finding suggested that CD11c+ and CD206+ macrophages (so-called M1- and M2-macrophages, respectively) are not well differentiated with respect to oxLDL carriage. Switching of M2 - to M1 -macrophages seen in animals,[27] might have occurred in human macrophages during migration; or because those macrophages may have possessed both M1 and M2 characters, and the potency of these characters changed while passing through IEL. Further studies are necessitated to clarify the role of individual macrophage phenotypes, including the subtypes of M1-macorphages and M2- macropahges [28], with regard to oxLDL carriage.
Possible mechanisms for underlying migration of OxLDL+ macrophages phenotypes from PCAT to the Intima

Figure 4: Schematic Representation of the Mechanism of OxLDL-containing (oxLDL+) Macrophage Migrating from the Pericoronary Adipose Tissue (PCAT) into the Intima Through External (EEL) and Internal Elastic Laminae (IEL). CD68+ or CD206+ macrophages in PCAT acquired oxLDL from adipocytes, moved to adventitia, passed though loosened or fragmented portions of EEL into the media, and subsequently passed through disrupted portions of IEL, whereas a portion of such a macrophage population transformed into CD11c+ macrophages, entered into the intima and secreted cytokines [4,7] to induce atherosclerosis [1]. EC: endothelial cell. MPO: myeloperoxidase. HOCL: hypochlorous acid. MMP: matrix metalloprotease.
Based on the findings of the present study and previously reported articles by other investigators [1,4], we propose a possible mechanism for human coronary atherosclerosis. The oxLDL stored in PCAT is transferred to CD68+ and/or CD206+ macrophages that reside in the interstitial space between adipocytes; oxLDL+ CD68+ and /or oxLDL+ CD206+ macrophages pass through the adventitia and loosened or fragmented EEL into the media; Subsequently, these macrophages pass through the disrupted portions of IEL, while a part of them transform into CD11c+ macrophages, and enter into the intima; these oxLDL+ macrophage phenotypes thus enter into the intima, release a number of cytokines [1,4,7] and cause atherosclerosis (Figure 5).
Study Limitations
1. Because a majority of the patients were admitted for a serious condition at terminal stage, lipid plasma levels and other substances examined at this stage were not considered to reflect the levels during stable conditions, therefore it was difficult to examine the association of plasma lipid levels and other substances with macrophages.
2. Although oxLDL storage in human PCAT was confirmed, the following mechanisms remain to be elucidated.
a) Despite the findings of the present study strongly suggesting that oxLDL is transported by macrophages, the real-time movement of oxLDL+ macrophages was not examined, and should be clarified by tracing labelled native oxLDL or macrophages in vivo.
b) The factor(s) that regulate oxLDL storage in PCAT as well as macrophage movement into the intima (plaque) remain to be elucidated.
c) It is still unknown whether oxLDL is synthetized in PCAT, or from where and how oxLDL is transported to the PCAT.
Clinical Perceptives
Therapies targeting PCAT and/or oxLDL+macrophage phenotypes could prevent human coronary atherosclerosis. Transthoracic or transcardiac administration via the right atrium [29] of anti- oxLDL substances could be used for this purpose.
Conclusion
OxLDL is stored in human PCAT. CD68+ and CD206+ macrophage acquire oxLDL from PCAT and transport it through the adventitia, loosened and fragmented EEL, media, and disrupted IEL into the intima, however a portion of these macrophages get converted to CD11c+ macrophages while passing through IEL before entering into the intima. Thus, our findings suggest that such an process would contribute to the initiation and progression of human coronary atherosclerosis and targeting therapy on oxLDL in PCAT or oxLDL+ macrophages could suppress coronary atherosclerosis.
Disclosure
The authors have no conflicts of interest to declare. Relationships with Industry: The authors have no relationships with industry.
Funding
No external funding was received for this study.
Author contributions
Yasumi Uchida and Yasuto Uchida conceived and designed the study, performed the in vitro study and wrote the manuscript; Ei Shimoyama and Nobuyuki Hiruta performed autopsy and immunohistochemical staining and conducted microscopic studies; and Tsuyoshi Tabata performed statistical analysis. All authors played a role in editing the manuscript.
References
- Libby P (2015) The vascular biology of atherosclerosis. In: Mann DL, Zipes DP, Libby P, Bonow RO, Braunwald E (Eds.) Heart disease (10th edn). Elsevier Saunders Ltd, Philadelphia 2: 873-890.
- Yoshida H, Kisugi R (2011) Mechanisms of LDL oxidation. Chimica Acta 411: 1875-1882.
- Takahashi K, Takeya M, Sakashita N (2002) Multifunctional roles of macrophages in the development and progression of atherosclerosis in human and experimental animals. Med Electron Microsc 35: 179-203.
- Sugiyama S, Kuriyama K, Aikawa M, Nakamura S, Ogawa H, et al. (2004) Hypochlorous acid, a macrophage product, induces m endothelial apoptosis and tissue factor expression: involvement of myeloperoxidase-mediated oxidant in plaque and thrombogenesis. Arterioscler Thromb Vasc Biol 24(7): 1309-1314.
- Uchida Y, Uchida Y, Shimoyama E, Hiruta K, Kishimoto T, et al. (2016) Pericoronary adipose tissue as storage and supply site for oxidized low-density lipoprotein in human coronary plaque. PloS One 11(3): e0150862.
- Lee CW, Hwang I, Park CS, Lee H, Park DW, et al. (2013) Macrophage heterogeneity of culprit coronary plaques in patients with acute myocardial infarction or stable angina. Am J Clin Pathol 139(3): 317-322.
- Cho KY, Miyoshi H, Kuroda S, Yasuda H, Kamiyama K, et al. (2013) The phenotype of infiltrating macrophages influences arteriosclerotic plaque vulnerability in carotid artery. J Stroke Cerebrovasc Dis 22(7): 910-918.
- Uchida Y (1995) Angioscopy system. In: Atlas for Cardioangioscopy. Uchida Y (Eds.), (Medical View Ltd, Tokyo 23-27.
- Uchida Y (2001) Angioscopes and its manipulation. In: Uchida Y (Eds.) Coronary angioscopy, Futura Publishing LTD, Armonk, NY: 11-24.
- Uchida Y (2001) Clinical classification of atherosclerotic coronary plaques. In: Coronary angioscopy, Uchida Y (ed), Futura Publishing Ltd, Armonk, NY: 71-81.
- Uchida Y, Uchida H, Kawai S, Shirai S, Tomaru T (2010) Detection of vulnerable coronary plaques by color fluorescent angioscopy. JACC Cardiovasc Imaging 3(4): 398-408.
- Holness CL, Simmons DL (1993) Molecular cloning of CD68, a human macrophage marker related to lysosomal glycoproteins. Blood 81(6): 1607-1613.
- McAlpine CS, Huang A, Emdin A, Banko NS, Beriault DR, et al. (2015) Deletion of myeloid GSK3 alpha attenuates atherosclerosis and promotes an M2 macrophage phenotype. Arterioscler Thromb Vasc Biol 35(3): 1113-1125.
- Park SJ, Lee KP, Kang S, Lee J, Sato K, Chung HY, et al. (2014) Shingosine 1- phosphate induced anti-atherogenic and atheroprotective M2 macrophage polarization through IL-4. Cell Signal 26(10): 2249-2258.
- Bonne D, Heusele C, Simon C, Pantaloni D (1985) 4’,6-Diamidino-2-phenylindole, a fluorescent probe for tubulin and microtubules. J Biol Chem 260: 2819-2825.
- Uchida Y, Uchida Y, Shimoyama E, Hiruta N, Kishimoto T, Watanabe S (2017) Human pericoronary adipose tissue as storage and possible supply site for oxidized low-density lipoprotein and high-density lipoprotein in coronary artery. J Cardiol 69(1): 236-244.
- Armstrong RA (2014) When to use the Bonferroni correction. Ophthalmoc Physiol Opt 34(5): 502-508.
- Uchida Y, Uchida Y, Matsuyama A, Koga A, Maezawa Y, et al. (2011) Functional medial thickening and folding of the internal elastic lamina in coronary spasm. Am J Physiol Circ Physiol 300(2): H423-430.
- Osbome-Pellegrin MJ, Farijanel J, Homebeck W (1990) Role of elastase and lysyl oxidase in spontaneous rupture of internal elastic lamina in rats. Atherosclerosis 10(6): 1136-1146.
- Denswil NP, van der Wal AC, Ritz K, de Boar OJ, Aronica E, et al. (2016) Atherosclerosis in the circle of Willis: Spacial differences in composition and in distribution of plaques. Atherosclerosis 251: 78-84.
- Shapouri-MA, Mohammadian S, Vazini H, Taghadosi M, Esmaeili SA, et al. (2018) Macrophage plasticity, polarization, and function in health and disease. J Cell Physiol 233(9): 6425-6440.
- Wessels DJ, Kuhl S, Soll DR (2001) Light microscopy to image and quantify cell movement. Methods Mol Biol 571: 455 - 471.
- Friedl P, Borgmann S, Broecker EB (2001) Amoeboid leukocyte creawling through extracellular matrix: lessons from the Dictyostelium paradigm of cell movement. J Leuko Biol 70(4): 491-509.
- Antrovic V, Marinovic M, Filic V, Weber I (2014) A simple optical configuration for cell tracking by dark-field microscopy. J Microbiol Methods 104: 9-11.
- Wolf K, Mueller R, Borgmann S, Broeker EB, Friedl P (2003) Amoeboid shape change and contact guidance: T-lymphocyte crawling through fibrillar collagen is independent of matrix remodeling by MMPs and other proteases. Blood 102(9): 3262-3269.
- DePaori F, Staeles B, Chinetti-Ghaguidi G (2014) Macrophage phenotypes and their modulation in atherosclerosis. Circ J 2014; 78(8): 1775-1781.
- Kallou-Lachest J, Varthuaman A, Fornasa G (2010) Macrophage plasticity in experimental atherosclerosis. PLoS One 5(1): e8852.
- Gambaro SE, Zubiria MG, Portales AE, Rey MA, Rumbo M, et al. (2018) M1 macrophages subtypes activation and adipocyte dysfunction worsen during prolonged consumption of a fructose-rich diet. J Nutr Biochem 61: 173-182.
- Uchida Y, Yanagisawa A, Nakamura F, Yamada K, Tomaru T, et al. (1995) Angiogenic therapy of acute myocardial infarction by intrapericardial injection of basic fibroblastic growth factor and heparin sulfate: An experimental study. Am Heart J 130(6): 1182-1188.




 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.