Review Article 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Viral Infection and SIDS: A Multidisciplinary Approach for the Forensic Pathologist.
*Corresponding author: Mazzanti Roberta, Dip. di Scienze Biomediche e Sanità Pubblica, Sez. di Medicina Legale, Università Politecnica delle Marche, Italy
Received: February 20, 2021; Published: March 22, 2021
DOI: 10.34297/AJBSR.2021.12.001744
Abstract
Sudden infant death syndrome (SIDS) is the sudden unexpected death of an apparent healthy infant under 1-year-old whose cause of death remains unknown despite a thorough death scene investigation, a review of the clinical history and autopsy. The risks factors for sudden infant death syndrome parallel those associated with susceptibility to or severity of infectious disease. This review assesses SIDS research in the context of clinicopathological features and focuses on the association of SIDS with a recent viral infection, most commonly a mild upper respiratory tract infection. The autoptic investigation of cases of sudden and unexpected death of the child must be carried out following standardized protocols and a multidisciplinary approach to establish the exact cause of death and in particular the role of respiratory tract infections as well as other exogenous triggers.
Keywords: Sudden infant death syndrome (SIDS), Sudden unexpected deaths in infancy (SUDI), Diagnosis, Classification, Viral infection, Human coronavirus HKU1
Introduction
Last century the scientist John Emery said that sudden infant death syndrome (SIDS) was in danger of becoming a “diagnostic dustbin [1]”. The term SIDS was introduced in the mid-1960s [2], partly for humanitarian reasons, as it provided a recognised category of natural death for the sudden demise of an infant without an identified cause, shifting the focus from parental blame onto research aimed at prevention [3]. Since 2004 there has been widespread adoption of the 2004 SIDS definition of San Diego: “the sudden unexpected death of an infant < 1 year of age, with onset of the fatal episode apparently occurring during sleep, that remains unexplained after a thorough investigation, including performance of a complete autopsy and review of the circumstances of death and the clinical history [4,5]”, emphasizing the importance of thoroughly considering the circumstances of death in formulating the diagnosis. The San Diego definition introduced three subcategories of SIDS to enable researchers to more accurately specify the likelihood of a case being attributable to “SIDS”. However, the more expansive sudden unexpected deaths in infancy (SUDI) grouping frequently appears to be erroneously used interchangeably with SIDS. Nearly 16 years on it appears that the San Diego definition is not working as well as it could as a definition/classification tool given that it does not appear to be applied rigorously or consistently [6]. There are now even proposals to use SUDI as the diagnostic term for unexplained sudden infant deaths, effectively becoming indistinct between unclassified sudden infant death (USID) and SIDS, which should only be recorded if all other possible causes of death have been excluded; this despite a quite robust framework being proposed for the use of SUDI by the Confidential Enquiries of Stillbirths and Deaths in Infancy (CESDI) group in 2000 [7]. Deaths due to SIDS account for about 1% of deaths under the year of life and is estimated that 250 SIDS cases occur in Italy every year [8].
SIDS affects families of all social, economic, and ethnic backgrounds. However, it is more likely to occur in babies born to mothers with limited or inadequate prenatal care, mothers who smoke during pregnancy, male infants (male-to-female ratio 3:2), prone and side-lying position during sleep [9-12], preterm or low birth weight infants, and in Native American/Alaska Native and non-Hispanic black infants [13]. In recent years, the rate of SIDS remained stationary despite major public health efforts aimed at improving infant’s sleep environment and focusing on high-risk groups [14].
Distinguishing between various causes of SUDI is complex, with classification issues long recognised as a “persistent and pervasive problem [15]”. A number of international initiatives have been undertaken to try to establish appropriate definitions, with death scene and autopsy protocols being formulated and published [16-28]. There are some international protocols that have been validated by several studies [29-31], in accordance with European guidelines for medico-legal autopsies [32], very similar to the International Standardised Autopsy Protocol [33] of the Global Strategy Task Force of SIDS International. The autopsy consists of an external examination, a complete internal examination, an X-ray investigation, histology, full toxicology, microbiological and genetic exams. The major changes in the last years regard the importance of the investigation of infant deaths, in particular the examinations of the circumstances of death [34].
Many USA [35] and other international [36,37] states have laws related to SIDS/SUID. In Italy, according to the Law 31/2006 [38] and the Decree of the Ministry of Health 7th October 2014 [39], child who died suddenly within the first year must undergo an autopsy in reference centres specialized. Information regarding the familial and environmental situations, must be accurately recorded and verified, for the diagnostic completion and purpose, by the obstetric gynaecologist, the neonatologist, the personal pediatrician and by the pathologist.
The Triple risk theory
According to the triple risk theory, SIDS occurs when (1) there is a vulnerable infant (2) in a critical but unstable period of development of homeostatic control (the highest risk period is at ages 2 to 4 months, with 90% of instances occurring before age 6 months) and (3) with previous exogenous stressor (e.g., prone or side resting position, soft bedding, or in utero or environmental tobacco exposure). Based on the model, all three factors must be present for a death to occur [40]. The combination of these factors leads toward a terminal respiratory pathway, and death occurs when protective mechanisms fail in the face of a life-threatening event [41]. This model has evolved suggesting that the mechanisms in SIDS is multifactorial [42]. Please see (Figure 1).
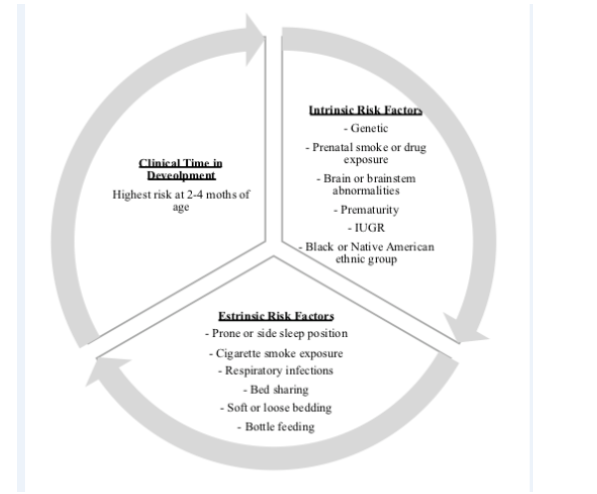
Figure 1: Triple-risk model for sudden infant death syndrome. IUGR, intrauterine growth restricted; SIDS, sudden infant death syndrome [37].
Infection and SIDS
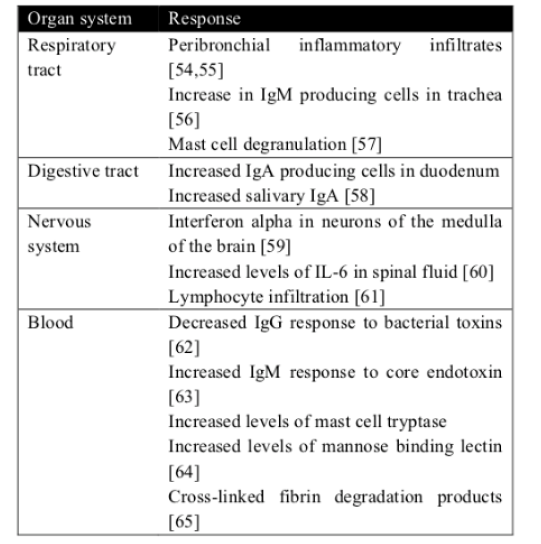
The idea that inflammation might be involved in these infant deaths in not new. In an article published in 1956, 126 nontraumatic sudden (“unexplained”) infant deaths were investigated, 106 (84%) revealed microscopic inflammatory changes in one or more sites of the respiratory tract, and there was histologic evidence of inflammatory disease in other organs in many cases [43]. Blood-Siegfried underlined the role of infection and inflammation in SIDS [44]. Also, Blackwell et al [45] found serum findings of inflammatory markers. These reviews confirm the existence of a link between infection/inflammation and SIDS. There is in fact evidence of an increase in interferon-γ [46] and interleukin [47] in infants who died of SIDS. Some recent studies show changes in the gut microbiome of SIDS babies and healthy babies [48,49]. Such a mechanism could operate in SIDS wherein an inflamed gut mucosa could promote microbial translocation into the bloodstream and cause overwhelming sepsis [50]. Respiratory infections are the leading causes of infant morbidity and mortality worldwide and new studies stressed the link of a recent viral infection and SIDS [51-53]. Please see (Table 1) [54-65].
A Standardized infant autopsy in Italy
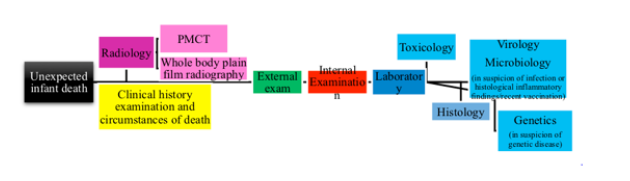
According to Decree of the Ministry of Health 7th October 2014 “Diagnostic protocols in infant victim of SIDS deceased suddenly within the first year of age…omitted” we propose the following steps. Clinical history examination and circumstances of death. The following information were collected: investigation data, witness interview, infant’s medical history, infant’s dietary history, pregnancy history, incident scene investigation. The first step is the knowledge of the case history and death scene investigation, following specific guidelines [66].
Complete autopsy. The term “complete” autopsy is used when a thorough external and internal examination has been performed. The concept was discussed by Matshes et al. [67], who said that a complete autopsy is when “every conceivable test has been performed”.
Photography
It starts with the documentation of the initial lividity pattern, as early as possible after death. Then, during the autopsy, are collected photos of all external body surfaces, all major soft tissue dissection planes and a photograph of the thoraco-abdominal organs.
External examination
Major body measurements (unclothed body weight with medical treatment removed, crown-heel length, crown-rump length, occipito-frontal circumference, chest circumference at nipples, abdominal circumference at umbilicus) are recorded. A full external examination of all body surfaces including the anogenital region, nose, and ears should be undertaken as soon as possible after death. The major reasons for this are to check for any unexplained injuries or lesions that may raise suspicions of accidental or inflicted injury. Internal examination Complete internal examination with evisceration and dissection of all organs and extensive histology; some organs are harvested as heart-lung bloc, tongue-hypopharynx bloc and the whole brain. With the proper use of radiographic examination, is possible to find traumatic injuries [68].
Ancillary techniques
Radiological screening
X-rays were performed to highlight major anatomic abnormalities or traumatic injuries. Is recommended to use three views (anteroposterior, lateral, and Towne’s) for the skull. When available, it is recommended the use of PMCT [69].
Toxicology
According to the German SIDS Study Group (GeSID) body fluids, tissue samples and stomach contents were taken and toxicological analysis was performed for general drug screens as sedativehypnotics.
Microbiology/Virology
Where histopathology confirmed infection, virology screening was done for the detection of defined viruses by PCR methods in tissues (e.g., lung tissue: influenza A, influenza A H1N1, influenza B, respiratory syncytial virus A, respiratory syncytial virus B, human parainfluenza 1, human parainfluenza 2, human parainfluenza 3, human parainfluenza 4, human coronavirus 229E, human coronavirus NL63, human coronavirus HKU1, human coronavirus OC43, human rhinovirus, human metapneumoviruses A/B, human bocavirus, human parechoviruses, entoroviruses, adenoviruses).
Genetics
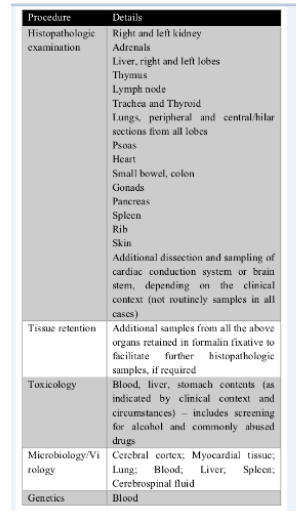
Molecular analysis of post-mortem blood samples has been shown to be beneficial in the identification of cardiac channelopathies in otherwise negative autopsy [70-78]. In case of suspect, is necessary to distinguish between lethal mutations leading to diseases such as medium-chainacyl-CoA dehydrogenase (MCAD) and long QT syndrome (LQTS), and polymorphisms (for instance, in the IL-10 gene and mtDNA) that are normal gene variants but might be suboptimal in critical situations and thus predispose infants to sudden infant death [79]. Please see (Table 2). We propose a standardized infant autopsy workflow to the autopsy of all unexpected infant deaths. Please see (Figure 2).
An Exemplary Case
Relevant clinical and scene data
At birth, in doubt of a LQTS at the first ECG control the infant had undergone a cardiological examination with a negative opinion for the presence of the canalopathy. There was an upper respiratory tract infection in the clinical history and diarrhoea two weeks before death.
Post-mortem findings
Well-developed and well-nourished infant with no apparent physical anomalies. Dark purple post-mortem hypostasis involved ventral surfaces. Petechial haemorrhages were on the epicardial surfaces of the heart, the lungs and thymus. Lungs were markedly congested and oedematous, with larger haemorrhages. Brain was slightly swollen but normal in size.
Histology
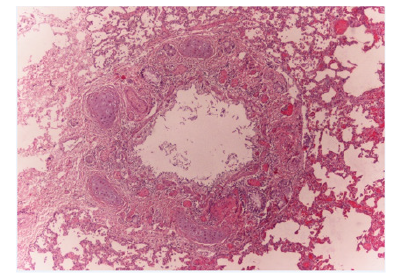
Vacuolar degeneration of hepatocytes. Lungs had oedema, hyperaemia, intra-alveolar haemorrhages and a small number of hystiocites with diffuse panbronchitis and eosinophilic and lymphoplasmacellular elements as shown in the image below. Please see (Figure 3). The other organs showed intense hyperaemia.
Ancillary techniques
Radiological screening was performed with negative results. Microbiological analysis was performed with real time PCR and Human Coronavirus HKU1 and Human Rhinovirus were demonstrated in lung tissue.
Conclusion
Acute Respiratory Failure in Pan bronchitis In Viral Hku1 Coronavirus and Rhinovirus Infection.
Discussion
f cases of sudden and unexpected deaths in all age groups is one of the major areas of interest of forensic medicine. The investigation of infant deaths requires the application of special diagnostic methods and includes forensic pathology as well as other fields of forensic medicine, such as forensic toxicology, and medicine in general, e.g. radiology, microbiology, virology, neuropathology, pediatrics, epidemiology and genetics. Autopsy, although often considered the gold standard in the determination of the causes of death, in often inadequate and allowed a definitive diagnosis in only 30% of the case in paediatric death [80]. Characteristic internal findings are exemplified by the triad of intrathoracic petechial haemorrhages involving the thymus, visceral pleura and epicardium. In most studies, ≥ 90% have the petechial triad, with significative differences from asphyxia [81,82]. Mainstream researchers attribute intrathoracic petechiae to changes in pulmonary pressure; however, this hypothesis does not stand up to scrutiny as argued in previous publications [83-85].
Krous et al. [86,87] disproved an association between intrathoracic petechiae and prone position, indicating that hypothetical upper airway obstruction attributable to face-down position is not causally related to development of intrathoracic petechiae. The lungs are congested and heavy [88] indicating pulmonary oedema/early shock lung as may occur in septic shock. Histopathology findings in these cases could provide important additional information which can be of help to establish the correct pathological diagnosis [89]. Histopathology shows very often mild inflammatory changes in the lungs [90,91]. Inflammatory alterations in the upper and lower respiratory tract are a common finding in sudden and unexpected death in infancy and up to 50-80% cases that can be explained after autopsy has detected to respiratory tract infections [92,93]. This is why most interest has focused on lung alterations, and especially on evidence of pulmonary inflammation. The typical hystological features in the lungs which can be seen in cases of suspected SIDS include pulmonary oedema, emphysema, atelectasias, increased number of alveolar macrophages, acute inflammation of the trachea and bronchi, pneumonia and prominent, hyperplastic bronchus-associated lymphoid tissue.
The significance of detecting viruses largely remains undetermined in the context of SIDS [94,95] and controversy exists same viruses are circulating at similar or higher frequencies in babies that died from other causes suchs as traumatic injuries and are also similar to those viruses circulating in the general population causing mild infections [96,97]. The presence of viral pathogens in lung tissue from SIDS cases in not always indicative of cause of deaths, for example dormant CMV has been found in numerous infants and babies without any pathological manifestation.
Fodha et al. [98] found that high nasopharyngeal RSV vital loads were shown to be a strong predictor of disease severity in infants, but a specific viral load that is required for respiratory infections to be deemed a significant contributing factor in causing death is still undefined [99]. Weber & Sebire [100] said to investigate the systemic responses to pathogens to find the role in disease progression [101]. Rambaud et al. [102] found that 62% of the sudden unexpected death in infancy cases had clinical symptoms of rhinitis and coughing prior to death. The pathogenicity of respiratory viruses can be low in immunocompetent individuals and is often associated with a good prognosis, but infants succumbing to SIDS have been suggested to be neither completely healthy, nor have normal defence mechanisms. Some may have underlying genetic and biological vulnerabilities which can predispose them to SIDS [103]. Virus infections and bacterial toxins induce cytokine activity, and it has been suggested that uncontrolled inflammatory mediators could be involved in some cases of SIDS [104-106]. In our case microbiological examination showed a viral infection. The aetiology of the inflammation was confirmed by microbiological investigations and Coronavirus HKU1 and Human Rhinovirus were demonstrated in lung tissue. Coronavirus HKU1 is an important pathogen in children under 3 years old with acute lower respiratory tract infection and the peak of its prevalence is spring and winter [107,108]. Coinfections show a more severe clinical outcome in comparison to single infections [109] and this could explain the fatal event in our case.
There is a big variation in cause of death determination and investigations practices for SUID among medical examiners. A recent study [110] highlights the lack of a uniformly applied and systematic approach to cause-of-death determination despite repeated calls for such [111]. Death certifiers need to develop mutually acceptable criteria and definitions to make causeof- death determinations reliably and accurately, especially to differentiate suffocation and asphyxia from other SUID cases [112-114]. The National Academy of Sciences (NAS) [115] report highlights several factors that negatively impact progress toward developing and disseminating standardized best practices related to medico-legal investigation, including variation in medico-legal death investigation systems, unequal levels of expertise, and lack of resources for medico-legal professionals (e.g., facilities, equipment, staff, and training). Until we address these limitations, accurate and reliable causes-of-death determination for SUID will remain a challenge.
Conclusion
The SIDS autopsy should be undertaken according to well established protocols. The most likely environmental or exogenous trigger is a viral infection, and the proposed underlying vulnerabilities include immune deficiencies and poor control or over-expression of inflammatory mediators. Viral infections may act in combination with the immune response of the host to infection and the initiation of bacterial toxin production.
This review highlights important virological aspects regarding investigations into the infectious nature of SIDS, including the importance to follow standardized guidelines for appropriate specimen collection at autopsy and subsequent laboratory analysis.
Key Points
1. For the forensic pathologist tasked with paediatric SIDS autopsies, significant training and experience in paediatric pathology is ideal.
2. Respiratory tract infection in the clinical history seems to be a common feature prior to death.
3. Our case underlines the necessity of a multidisciplinary approach to researching SIDS, involving both clinicians and biologists, to determine the causes of these deaths.
4. Implementing autopsy protocol would appear to be an essential prerequisite to gaining a better understanding of SIDS.
References
- Emery JL (1989) Is sudden infant death syndrome a diagnosis? Or is it just a diagnostic dustbin? Br Med J 299: 1240.
- Beckwith JB, Bergman AB, Beckwith JB, Ray CG (1970) Discussion of terminology and definition of the sudden infant death syndrome. In: Sudden infant death syndrome: proceedings of the second international conference on the causes of sudden death in infants. Seattle: University of Washington Press 14-22.
- Hunt C, Darnall R, McEntire B, Hyma B (2015) Assigning cause for sudden unexpected infant death. Forensic Sci Med Pathol 11(2): 283-288.
- Krous HF, Beckwith JB, Byard RW (2004) Sudden infant death syndrome and unclassified sudden infant deaths: a definitional and diagnostic approach. Pediatrics 114(1): 234-238.
- Willinger M, James LS, Catz C (1991) Defining the sudden infant death syndrome (SIDS): deliberations of an expert panel convened by the National Institute of Child Health and Human Development. Pediatr Patho 11(5): 677-684.
- Byard RW, Shipstone RA, Young J (2019) Continuing major inconsistencies in the classification of unexpected infant deaths. J For Leg Med 64: 20-22.
- Fleming P, Bacon C, Blair P, Berry PJ (2000) Sudden Unexpected Deaths in Infancy. The CESDI SUDI Studies 1993-1996. London: The Stationary Office.
- (1887-2015) Istat Years 1887-2015. The mortality of children yesterday and today in Italy.
- (1992) American Academy of Pediatrics AAP Task Force on Infant Positioning and SIDS: Positioning and SIDS. Pediatrics 89(6): 1120-1126.
- Willinger M (1995) SIDS prevention. Pediatric Ann 24(7): 258-364.
- Shapiro-Mendoza CK, Tomashelk KM, Anderson R, Wingo J (2006) Recent national trends in sudden, unexpected infant deaths: more evidence supporting a chance in classification or reporting. Am J Epidemiol 163(8): 762-769.
- Goldstein RD, Tachtenberg FL, Sens MA (2016) Overall postneonatal mortality and rates of SIDS Pediatrics 137(1).
- Heron M (2016) Deaths: leading causes for 2014. Natl Vital Stat Rep 65(5): 1-96.
- Goldberg N, Rodriguez-Prado Y, Tillery R, Chua C (2018) Sudden Infant Death Syndrome: A Review. Pediatric Ann 47(3): e118-e123.
- Byard RW (2001) Inaccurate classification of infant deaths in Australia: a persistent and pervasive problem. Med J Aust 175: 5-7.
- Willinger M, James LS, Catz C (1991) Defining the sudden infant death syndrome (SIDS): deliberations of an expert panel convened by the National Institute of Child Health and Human Development. Pediatr Pathol 11: 677-684.
- Gibson AAM (1994) The use of a standard SIDS postmortem protocol in Scotland: two years’ audit. Third SIDS International Conference, Stavanger. Norway. Oslo: Norwegian SIDS Society.
- Mitchell E, Krous HF, Donald T, Byard RW (2000) An analysis of the usefulness of specific stages in the pathological investigation of sudden infant death. Am J Forensic Med Pathol 21(4): 395-400.
- Krous HF, Byard RW (2001) International standardized autopsy protocol for sudden unexpected infant death. In: Byard RW, Krous HF, eds. Sudden Infant Death Syndrome. Problems, Progress and Possibilities. London: Arnold: 319-333.
- Fleming PJ, Blair PS, Sidebotham P, Hayler T (2004) Investigating sudden unexpected deaths in infancy and childhood and caring for bereaved families: an integrated multiagency approach. Br Med J 328: 331-334.
- Krous HF, Beckwith JB, Byard RW, Rognum TO, Bajanowski T, et al. (2004) Sudden infant death syndrome and unclassified sudden infant deaths: a definitional and diagnostic approach. Pediatrics 114(1): 234-238.
- Bajanowski T, Brinkmann B, Vennemann M (2006) The San Diego definition of SIDS: practical application and comparison with the GeSID classification. Int J Legal Med 120(6): 331-336.
- (2007) Centers for Disease Control and Prevention. The Sudden Unexplained Infant Death Investigation Report Form (SUIDIRF).
- Hanzlick R, Jentzen J (2007) Centers for Disease Control and Prevention. Sudden unexplained infant death investigation: Guidelines for the scene investigator.
- Blair PS, Byard RW, Fleming PJ (2012) Sudden unexpected death in infancy (SUDI): suggested classification and applications to facilitate research activity. Forensic Sci Med Pathol 8(3): 312-315.
- Shapiro-MCK, Camperlengo L, Ludvigsen R, Cottengim C, Anderson RN, et al. (2014) Classification system for the Sudden Unexpected Infant Death Case Registry and its application. Pediatrics 134(1): e210-e219.
- Jensen LL, Rohde MC, Banner J, Byard RW (2016) Reclassification of SIDS cases - a need for adjustment of the San Diego classification? Int J Legal Med 126(2): 271-277.
- Kennedy H (2016) Sudden Unexpected Death in Infancy and Childhood. Multi-agency guidelines for care and investigation. second ed. The Royal College of Pathologists: 12.
- Mitchell EA, Scragg R, Stewart AW, Becroft DM, Taylor BJ, Ford et al. (1991) Results from the first year of the New Zealand cot death study. N Z Med J 104: 71-76.
- L’Hoir MP, Engelberts AC, van Well GThJ, Bajanowski T, Helweg-Larsen K (1998) Sudden unexpected death in infancy; epidemiology determinated risk factors related to a pathologhy classification. Acta Paediatr 87(12): 1279-1287.
- Findeisen M, Vennemann M, Brinkmann B, Ortmann C, Röse I (2003) German study on sudden infant death (GeSID): design, epidemiological and pathological profile. Int J Legal Med 118(3): 163-169.
- Brinkmann B (1999) Harmonisation of medico-legal autopsy rules. Int J Legal Med 113(1): 1-14.
- Krous HF (1996) Instruction and reference manual for the International Standardise Autopsy Protocol for sudden unexpected infant death, J. SIDS Infant Mortal 1: 203-246.
- Byard RW, Beal SM (1995) Has changing diagnostic preference been responsible for the recent fall in incidence of sudden infant death syndrome in South Australia? J Paediatr Child Health 31(3): 197-199.
- (2016) National Conference of State Legislatures. Sudden unexpected infant death legislation.
- (2017) Inquests Act 58 of 1959 (South Africa).
- (2003) National Health Act 2003 (South Africa) reg 636. Regulations Regarding the rendering of Forensic Pathology Service.
- Italian Law 31/2006 “Regulations for Diagnostic PostMortem Investigation in Victims of Sudden Infant Death Syndrome (SIDS) and Unexpected Fetal Death” (Health Ministry - G.U. no. 662/02-02-2006).
- (2014) Decree of the Ministry of Health 7th October 2014 “Protocolli diagnostici nei casi della morte improvvisa infantile e della morte inaspettata del feto” (Healh Ministry - G.U. no. 272/22-11-2014).
- Filiano JJ, Kinney HC (1994) A perspective on neuropathologic findings in victims of the sudden infant death syndrome: the triple-risk model. Biol Neonate 65(3-4): 194-197.
- Kinney HC, Thach BC (2009) The sudden infant death syndrome. N Engl J Med 361(8): 795-805.
- Spinelli J, Collins-Prain L, Van Den Heuvel C, Byard RW (2017) Evolution and significance of the triple risk model in sudden infant death syndrome. J Paediatr Child Health 53(2): 112-115.
- Adelson L, Kinney ER (1956) Sudden and unexpected death in infancy and childhood. Pediatrics 17(5): 663-699.
- Blood-Siegfried J (2009) The role of infection and inflammation in sudden infant death syndrome. Immunotoxicol 31(4): 516-523.
- Blackwell C, Moscovis S, Hall S, Burns C, Scott RJ (2015) Exploring the risk factors for sudden infant deaths and their role in inflammatory responses to infection. Front Immunol 6: 44.
- Moscovis SM, Gordon AE, Al Madani OM (2015) Genetic and environmental factors affecting TNF-α responses in relation to sudden infant death syndrome. Front Immunol 6: 374.
- Vege A, Rognum TO, Scott H, Aasen AO, Saugstad OD (1995) SIDS cases have increased levels of interleukin-6 in cerebrospinal fluid. Acta Paediatr 84(2): 193-196.
- Highet AR, Goldwater PN (2009) Staphylococcal enterotoxin genes are common in Staphylococcus aureus intestinal flora in sudden infant death syndrome (SIDS) and live comparison infants. FEMS Immunol Med Microbiol 57(2): 151-155.
- Leong LHX, Choo JM, Goldwater PN (2016) Gut microbiota composition in sudden infant death syndrome (SIDS) infants and healthy age-matched controls. Abstract International Symposium on Microbial Ecology Montreal 2016.
- Dickson RP, Erb-Downward JR, Martinez FJ, Huffnagle GB (2016) The Microbiome and the Respiratory Tract. Annu Rev Physiol 78: 481-504.
- Samuels M (2003) Viruses and sudden infant death. Pediatr Respir Rev 4(3): 178-183.
- Weber MA, Hartley JC, Ashworth MT, Malone M, Sebire Nj (2010) Virological investigations in sudden unexpected deaths in infancy (SUDI). Forensic Sci Med Pathol 6(4): 261-267.
- Limelette A, Boulagnon C, Terrade C, N’guyen Y, Guillard T, et al. (2013) Investigation of the sudden infant death syndrome: a multidisciplinary approach is required. Ann Biol Clin 71(3): 299-304.
- Howat WJ, Moore IE, Judd M, Roche WR (1994) Pulmonary immunopathology of sudden infant death syndrome. Lancet 343 (8910): 1390-1392.
- Baxendine JA, Moore IE (1995) Pulmonary eosinophilia in sudden infant death syndrome. J Pathol 177(4): 415-421.
- Stoltenberg I, Vege A, Opdal S, Saugstad O, Rognum TO (1995) Does immunostimulation play a role in SIDS? In: Rognum T, editor. Sudden Infant Death Syndrome, New Trends in the Nineties. Oslo: Scandinavian University. Press 179-181.
- Holgate ST, Walters C, Walls AF, Lawrence S, Shell DJ, et al. (1994) The anaphylaxis hypothesis of sudden infant death syndrome (SIDS): mast cell degranulation in cot death revealed by elevated concentrations of tryptase in serum. Clin Exp Allergy 24(12): 1115- 1122.
- Gleeson M, Clancy RL, Cripps AW (1993) Mucosal immune response in a case of sudden infant death syndrome. Pediatr Res 33(6): 554-556.
- Howatson AG (1992) Viral infection and alpha interferon in SIDS. J Clin Pathol 45(11): 25-28.
- Vege Å, Rognum TO, Scott H, Aasen AO, Saugstad OD (1995) SIDS cases have increased levels of interleukin-6 in cerebrospinal fluid. Acta Paediatr 84(2): 193-196.
- Morris JA, Harrison LM, Telford DR (2012) Postmortem cerebrospinal fluid pleocytosis: a marker of inflammation or postmortem artifact? Int J Pediatr: 964074.
- Siarakas S, Brown AJ, Murrell WG (1999) Immunological evidence for a bacterial toxin aetiology in sudden infant death syndrome. FEMS Immunol Med Microbiol 25(1-2): 37-50.
- Oppenheim BA, Barclay GR, Morris J, Knox F, Barson A, et al. (1994) Antibodies to endotoxin core in sudden infant death syndrome. Arch Dis Child 70(2): 95-98.
- Kilpatrick DC, James VS, Blackwell CC, Weir DM, Hallam NF, et al. (1998) Mannan binding lectin and the sudden infant death syndrome. Forensic Sci Int 97(2-3): 135-138.
- Goldwater PN, Williams V, Bourne AJ, Byard RW (1990) Sudden infant death syndrome: a possible clue to causation. Med J Aust 153(1): 59-60.
- (2006) Sudden unexplained infant death investigation: a systematic training program for the professional infant death investigation specialist. Atlanta: United States Department of Health & Human Services (Centers for Disease Control and Prevention): 250.
- Matshes E, Milroy C, Paraj (2011) What is a complete autopsy? Acad Forensic Pathol 1(1): 2-7.
- Pinneri K, Matshes EW (2017) Recommendations for the Autopsy of an Infant who has Died Suddenly and Unexpectedly. Acad Forensic Pathol 7(2): 171-181.
- Matshes E, Snyder V, Andrews S (2015) Utilitarian aspects of post-mortem computed tomography. Acad Forensic Pathol 5(4): 603-613.
- Wilson SJ, Wilson ML, Reller LB (1993) Diagnostic utility of postmortem blood cultures. Arch Pathol Lab Med 117(10): 986-988.
- Tester DJ, Ackerman MJ (2009) Cardiomyopathic and channelopathic causes of sudden unexplained death in infants and children. Annu Rev Med 60: 69-84.
- Tang Y, Siegel D, Sampson B (2011) Molecular investigations of sudden unexplained deaths. Acad Forensic Pathol 1(2): 194-201.
- Gill J, Lange R, Azar O (2011) What is sudden cardiac death. Acad Forensic Pathol 1(2): 176-186.
- Middleton O, Baxter S, Demo E, Honeywell C, Jentzen J, et al. (2013) National Association of Medical Examiners postition paper: retaining post-mortem samples for genetic testing. Acad Forensic Pathol 3(2): 191-194.
- Severson E, Timm K (2014) The value of molecular autopsy: genetic testing reveals long QT mutations in an autopsy-negative, postpartum sudden unexpected death. Acad Forensic Pathol 4(4): 552-556.
- Cunningham K, Pollanen M (2015) Evolution of a molecular autopsy program from within a death investigation system. Acad Forensic Pathol 5(2): 211-220.
- Duncanson E, Lynch K, Baker A, Abdelhadi RH, Mackey-Bojack SM (2015) Establising a multidisciplinary network for the workup of sudden cardiac death. Acad Forensic Pathol 5(2): 221-232.
- Krehbiel K, Pinckard J (2015) The toolbox approach to forensic pathology. Acad Forensic Pathol 5(4): 534-547.
- Opdal SH, Rognum TO (2004) The Sudden Infant Death Syndrome Gene: Does It Exist? Pediatrics 114(4): e506-512.
- Delteil C, Tuchtan L, Torrents J, Capuani C, Piercecchi-Marti MD (2018) Pediatric medicolegal autopsy in France: A forensic histopatological approach. J of Forensic and Legal Medicina 53: 106-111.
- Becroft DM, Thompson JM, Mitchell EA (1998) Epidemiology of intrathoracic petechial hemorrhages in sudden infant death syndrome. Pediatr Dev Path 1(3): 200-209.
- Goldwater PN (2008) Intrathoracic Petechial Haemorrhages in sudden infant death syndrome and other infant deaths: time for re-examination? Pediatr Dev Pathol 11(6): 450-455.
- Goldwater PN (2003) Sudden infant death syndrome: a critical review of approaches to research. Arch Dis Child 88: 1095-1100.
- Goldwater PN (2011) A perspective on SIDS pathogenesis. The hypotheses: plausibility and evidence. BMC Med 9: 64.
- Goldwater PN, Bettelheim KA (2013) SIDS risk factors: time for new interpretations the role of bacteria. Pediatr Res Int J 867520.
- Krous HF, Nadeau JM, Silva PD, Blackbourne BD (2001) Intrathoracic petechiae in sudden infant death syndrome: relationship to face position when found. Pediatr Dev Pathol 4(2): 160-166.
- Krous HF, Haas EA, Chadwick AE, Masoumi H, Stanley C (2008) Intrathoracic petechiae in SIDS: a retrospective population-based 15-year study. Forensic Sci Med Pathol 4(4): 234-239.
- Berry PJ (1992) Pathological findings in SIDS. J Clin Pathol 45(11): 11-16.
- Bokor J, Danics K, Bencze E, Keller E, Szollosi Z (2017) A single-centre review of suspected sudden infant death cases. Med Sci Law 57(2): 84-90.
- Kelmanson IA (1992) Differences in somatic and organ growth rates in infants who died of sudden infant death syndrome. J Perinat Med 20(3): 183-188.
- Rambaud C, Guibert M, Briand E, Grangeot-Keros L, Coulomb-L’Herminé A, et al (1999) Microbiology in sudden infant death syndrome (SIDS) and other childhood deaths. FEMS Immunol Med Microbiol. 25(1-2): 59-66.
- Bajanowski T, Brinkmann B (1995) Pulmonary viral infection in SIDS, in: T.O. Rognum (Eds.) Sudden Infant Death Syndrome. New Trends in the Nineties, Scandinavian University Press, Stockholm 199-202.
- Bajanowski T, Rolf B, Jorch G, Brinkmann B (2003) Detection of RNA viruses and pulmonary viral infection in 424 sudden infant death (SID). Int J Legal Med 117: 237-240.
- Dettmeyer R, Baasner A, Schlamann M (2004) Role of virus-induced myocardial affections in sudden infant death syndrome: a prospective postmortem study. Pediatr Res 55(6): 947-952.
- Alvarez-Lafuente R, Aguilera B, Suarez-Mier MP, et al. (2008) Detection of human herpesvirus-6, Epstein-Barr virus and cytomegalovirus in formalin-fixed tissues from sudden infant death: a study with quantitative real-time PCR. Forensic Sci Int. 178(2-3): 106-111.
- Urquhart GE, Grist NR (1972) Virological studies of sudden, unexplained infant deaths in Glasgow 1967-70. J Clin Pathol 25(5): 443-446.
- Williams AL, Uren EC, Bretherton L (1984) Respiratory viruses and sudden infant death. Br Med J 288 (6429): 1491-1493.
- Fodha I, Vabret A, Ghedira L, Seboui H, Chouchane S, et al. (2007) Respiratory syncytial virus infections in hospitalized infants: association between viral load, virus subgroup, and disease severity. J Med Virol 79(12): 1951-1958.
- Krous HF, Fernandos C, Masoumi H, et al. (2009) Myocardial inflammation, cellular death, and viral detection in sudden infant death caused by SIDS, suffocation, or myocarditis. Pediatr Res 66(1): 17-21.
- Weber MA, Sebire NJ (2010) Molecular diagnostic techniques in the post-mortem investigation of sudden unexpected infant deaths: current and future applications. Open Pathol J 4: 110-119.
- La Grange H, Verster J, Dempers JJ, De Beer C (2014) Review of immunological and virological aspects as contributory factors in Sudden Unexpected Death in Infancy (SUDI). For Sci Int 245: 12-16.
- Rambaud C, Guibert M, Briand E, Grangeot-KL, Coulomb-L’HA, et al. (1999) Microbiology in sudden infant death syndrome (SIDS) and other childhood deaths. FEMS Immunol Med Microbiol 25(1-2): 59-66.
- Weber MA, Sebire NJ (2010) Molecular diagnostic techniques in the post-mortem investigation of sudden unexpected infant deaths: current and future applications. Open Pathol J 4: 110-119.
- Forsyth KD (1999) Immune and inflammatory responses in sudden infant death syndrome. FEMS Immunol Med Microbiol 25(1-2): 79-83.
- Raza MW, Blackwell CC (1999) Sudden infant death syndrome, virus infections and cytokines. FEMS Immunol Med Microbiol 25(1-2): 85-96.
- Highet AR, Berry AM, Goldwater PN (2009) Novel hypothesis for unexplained sudden unexpected death in infancy (SUDI). Arch Dis Child 94(11): 841-843.
- Xiao NG, Xie ZP, Zhou QH, Zhang RF, Zhong LL, et al. (2011) Detection and clinical study on coronavirus HKU1 with acute lower respiratory tract infections of hospitalized children in Changsha. Zhonghua Shi Yan He Lin Chuang Bing Du Xue Za Zhi 25(1): 2-4.
- Goes LG, Durigon EL, Campos AA, Hein N, Passos SD, et al. (2011) Coronavirus HKU1 in children, Brazil, 1995. Emerg Infect Dis 17(6): 1147-1148.
- Debiaggi M, Canducci F, Ceresola ER, Clementi M (2012) The role of infections and coinfections with newly identified and emerging respiratory viruses in children. Virol J 27(9): 247.
- Shapiro-Mendoza CH, Parks SE, Brustrom J, Andrew T, Camperlengo l, et al. (2017) Variations in Cause-of-death determination for sudden unexpected infant deaths. Pediatrics 140(1): e20170087.
- Corey TS, Hanzlick R, Howard J, Nelson C, Krous H (2007) NAME Ad Hoc Committee on Sudden Unexplained Infant Death. A functional approach to sudden unexplained infant deaths. Am J Forensic Med Pathol 28(3): 271-277.
- Randall B, Donelan K, Koponen M, Sens MA, Krous HF (2012) Application of a classification system focusing on potential asphyxia for cases of sudden unexpected infant death. Forensic Sci Med Pathol 8(1): 34-39.
- Moon RY, Byard RW (2014) Need for a working classification system for sudden and unexpected infant deaths. Pediatrics 134(1): e240-241.
- Hunt CE, Darnall RA, McEntire BL, Hyma BA (2015) Assigning cause for sudden unexpected infant death. Forensic Sci Med Pathol 11(2): 283-288.
- National Research Council (US) (2009) Strengthening Forensic Science in the United States a Path Forward. Washington DC: National Academies Press. USA.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.