Research Article 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Risk Factors of Symptomatic Pelvic Organ Prolapse in Japanese Women
*Corresponding author: Chang-Nian Wei, Department of Public Health, Faculty of Life Sciences, Kumamoto University, Honjo 1-1- 1, Chou-ku, Kumamoto, 860-8556, Japan.
Received: July 06, 2021; Published: July 22, 2021
DOI: 10.34297/AJBSR.2021.13.001898
Abstract
Aim: To estimate the prevalence, risk factors, and subjective health status of symptomatic pelvic organ prolapse in Japanese women
Methods: This was a population-based cross-sectional study of 8,407 randomly selected women, aged 20–90 years. Symptomatic prolapse was self-reported by feeling of a bulge or something falling out from the vagina. Impact of the risk factors on symptomatic pelvic organ prolapse was estimated using logistic regression analysis. Multiple linear regression analysis was used to examine the association between pelvic organ prolapse symptoms and the subjective health status score.
Results: Symptomatic pelvic organ prolapse was reported in 474 participants (5.6%), and the proportion of symptomatic pelvic organ prolapse cases were higher among older women and highest in those aged 70–79 years. Multivariable logistic analysis revealed a significantly higher risk of pelvic organ prolapse in women with ≥1 vaginal deliveries than in nulliparous women. Additionally, self-reported chronic medical conditions, constipation, and heavy lifting at work were strongly associated with symptomatic pelvic organ prolapse. The subjective health status was significantly impaired by pelvic organ prolapse symptoms.
Conclusion: In Japanese women, the number of vaginal deliveries is a risk factor of symptomatic pelvic organ prolapse. Additionally, the symptomatic pelvic organ prolapse negatively affected the self-reported health status similar to other chronic medical conditions.
Keywords: Cross-Sectional Study; Pelvic Organ Prolapse (POP); Risk Factors; Parity; Subjective Health Status
Introduction
Although pelvic organ prolapse (POP) does not directly cause severe morbidity or mortality, the pelvic floor disorders (POP, urinary and fecal incontinence, voiding, and sexual dysfunction) can adversely affect daily life, [1] health status, [2] and quality of life. [3] POP has numerous risk factors, including parity, [4] old age, [5] obesity, [6] hysterectomy, [7] chronic medical conditions, [3] heavy lifting, [8] constipation, [2] smoking, [9] employment/ occupation, [3] and race/ethnicity. [10]
There is limited epidemiological data regarding symptomatic POP in Japan, despite being recognized as a disease that can be treated in outpatient or inpatient settings. This study aimed to identify the prevalence, risk factors, and subjective health status of symptomatic POP in Japanese women.
Methods
This cross-sectional study evaluated 20,900 women who were Japanese Agriculture Group (JA) employees between August 2014 and July 2015. The JA is a cooperative agricultural association that employs individuals to perform agricultural, industrial, managerial, medical, and educational functions. This study sample included JA office staff, service women, teachers, nurses, parttime workers, saleswomen, part-time agricultural workers, and full-time agricultural workers. The JA employees were served by the following six JA medical centers: Kumamoto and Oita centers (Kyushu, south Japan), Yamaguchi and Aichi centers (middle Japan), and Chiba and Iwate centers (north Japan). These centers were chosen through the Kumamoto University community health network. Women were considered eligible to participate in this study if they were aged >20 years and not currently pregnant. To ensure anonymity, the participants were not required to provide their name on the questionnaire and were asked to return the completed questionnaire to the researchers within 2 weeks.
The participants recorded their age (years), height (cm), and weight (kg) in the questionnaire. They then answered the following questions regarding the symptoms of POP (Table 1): Do you experience bulging near your vagina or something protruding? Has your vagina bulged? Can you see or feel your uterus? Can you see something that has dropped in your pelvis? Women who responded “yes” to any of these questions were defined as having symptomatic POP.
Urinary incontinence was defined as the self-reported presence of any of the following symptoms: urine leaking during sneezing or coughing, urine leaking during heavy lifting, urine leaking during the urge to toilet, difficulty in emptying the bladder, or frequent urination (>8 times during the day or >3 times at night). Positive and negative responses were assigned scores of “1” and “0,” respectively.
To define the risk factors of symptomatic POP, the responses to the following questions were recorded: Were they ever examined in a hospital for their symptoms? How many times have they delivered a child? Do they have any chronic medical conditions currently being treated? What is their subjective health status? (Table 1). The selfreported chronic medical conditions were categorized based on the total number of positive responses: 0, 1, 2, and ≥3 conditions. The self-reported health status was categorized as “excellent,” “good,” “fail,” and “poor.” Ages were categorized into groups with 10-year increments (20–29 years to ≥80 years).
Body mass index (BMI) was calculated as weight divided by height squared (kg/m2), and the participants were categorized as being normal weight (<25.0 kg/m2), overweight (25.0–30.0 kg/ m2), or obese (>30.0 kg/m2). Participants were asked to report their parity as the total number of vaginal and cesarean deliveries.
Validity of Methods
When this study was initiated, there was no validated tool for assessing symptomatic POP and the related symptoms in Japanese women. The questionnaire was constructed based on few experts’ group works or modeled on several previously validated questionnaires [11-19] or the tools used to evaluate outpatients during gynecological visits in Japan. Based on these findings, we developed a nine-question self-reported questionnaire to identify participants with symptomatic POP (Table 1). The internal consistency of the questionnaire was good (Cronbach’s α = 0.87). The test-retest reliability was tested, and 50 women were randomly selected from the expectation assessment group, who filled the questionnaire twice at an interval of 2 weeks (r = 0.84).
Statistical Analysis
Standard methods were used for computing the mean, standard deviation (SD), and confidence interval (CI). The χ2 test was used for the analysis of nominal and ordinal data. To determine the contribution of the various proposed causes for POP, multivariate logistic regression analysis was used to control for potential confounding variables and to determine the independent associations between POP and the potential risk factors identified a priori. Urinary incontinence was excluded a priori, because it was considered a part of the outcome and associated with pelvic floor dysfunction, rather than a risk factor of POP. [2] Multiple linear regression analysis was used to measure the independent association between the self-reported health status and activities affected by the risk of POP.
Weighted prevalence estimates and 95% CI were calculated using Statistical Package for the Social Sciences (version 25.0; SPSS Inc., Chicago, IL). Pearson’s χ2 test was used to assess the association between the pelvic floor disorders and the participants’ demographic characteristics. Differences were considered statistically significant for p-values <0.05.
This study used cross-sectional design, which was approved by the Ethics Committee of the Kumamoto University Faculty of Life Sciences (approval number: 839). All participants provided their informed consent, and the study was performed in accordance with the tenets of the Declaration of Helsinki.
Results
The questionnaire was completed by 8,407 women, with a mean age of 61.1 ± 11.5 years (range, 20–90 years) and median parity of 2 (range, 0–8). In total, 474 (5.64%) women were diagnosed with symptomatic POP. The prevalence of symptomatic POP increased with age and peaked in the 70–79 years age group (prevalence rates: 20–29 years, 0.86%; 30–39 years, 3.45%; 40–49 years, 3.59%; 50–59 years, 5.79%; 60–69 years, 6.12%; 70–79 years, 6.27%; and ≥80 years, 5.37%; p < 0.001) (Table 2).
Among these 474 women, 46% (mean age ± SD: 56.2 ± 11.5) reported that they had never been examined, 21% (58.8 ± 11.7) had been examined and treated, 11% (56.0 ± 13.9) had been examined but did not need treatment, 2% (61.6 ± 14.6) needed treatment but did not get treated, and 20% (58.8 ± 15.5) were missing data. Thus, approximately 34% of women with POP sought medical attention and health care, 46% did not, and the data for 20% remained unknown. No significant differences were observed in the ages of women who did and did not seek medical attention. Almost 94% of the participants reported childbirth experience, 7% had undergone the caesarean section, 34.8% reported constipation, and 9% reported dystocia experiences.
In the multivariate logistic regression analysis, factors independently and significantly associated with symptomatic POP were self-reported chronic conditions (2.25-fold increase in odds of those with ≥3 conditions), constipation (1.63-fold increase in odds), and heavy lifting at work (2.03-fold increase in odds of those lifting heavy every day). Furthermore, women with one or more parity showed 2.5- to 4-fold higher odds of having symptomatic POP as compared to nulliparous women. Age, BMI, and occupation did not show an independent association with reported symptomatic POP (Table 3).
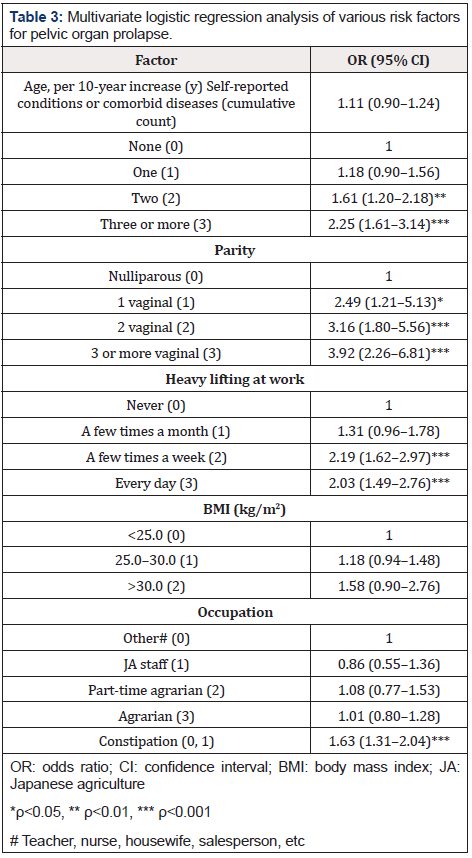
Table 3: Multivariate logistic regression analysis of various risk factors for pelvic organ prolapse.
The multiple linear regression analysis examined the factors independently and significantly associated with self-reported health status (excellent, good, fail, or poor). Self-reported chronic medical conditions, constipation, POP, BMI, heavy lifting at work, age, parity, urinary incontinence, and occupation were used as independent factors. As shown in Table 4, multiple regression results indicated that the self-reported health status had a significant association with self-reported chronic medical conditions, constipation, POP, BMI, heaving lifting at work, and age, but not with parity, UI, and occupation. Furthermore, increased number of chronic medical conditions, constipation, POP symptoms, high BMI, heavy lifting at work, and old age decreased the health status in women.
Discussion
The reported prevalence of symptomatic POP in Japanese women in this population-based cross-sectional study was 5.6%, [20] which was similar to that reported in population-based studies in US (5.7%), [2] UK (6.5%), [21] France (6.4%), [3] Italy (5.5%), [22] Sweden (8.3%), [14] and India (7%). [23] Population-based surveys have found that 6–8% of women report symptoms of POP [2,14]. POP estimates using only questionnaires underreport its true prevalence than that based on physical exanimation. A high prevalence of symptomatic POP is reported in women who undergo surgical prolapse repair. Data suggest a 11–19% lifetime risk in women who undergo surgery for prolapse [1,24,25]. POP has a prevalence of 3–6% when defined by symptoms and up to 50% when based on vaginal examination [26].
In our study, self-reported chronic conditions, parity, and heavy lifting at work were associated with symptomatic POP. The results are consistent with those of other population-based studies [2,9,14,27,28]. Additionally, constipation was a strong and independent risk factor of symptomatic POP, [2,29] though this association was not observed in any other study [9,30]. In our study, the prevalence of constipation showed a significant difference between the asymptomatic and symptomatic POP patients (25.2% vs. 34.8%; p < 0.001). The prevalence of constipation varies depending on the definition [31] of constipation, the surveying method, [32] and the target group [33] of the survey. Even though the National Life Basic Survey results showed that 4.6% of women reported having constipation in Japan, [34] it is not an accurate statistic because it represents the number of people who were aware of their subjective symptoms and not those with objective indicators that can be detected easily by doctors. Therefore, the causal relationship between constipation and POP requires further longitudinal follow-up studies.
Age was not found to be a risk factor of symptomatic POP. Prior studies have reported inconsistent findings regarding the association between age and POP [2,9,14,27,35]. It is possible that older women have lesser awareness of prolapse symptoms or are engaged in fewer activities that could provoke POP symptoms as compared to younger women [2]. Our results showed that about 34% women with symptomatic POP sought medical attention and health care, 46% did not, and 21% had missing data. However, no significant difference in age was noted between those who did and did not seek medical attention. This could be because the medical care and treatment for POP in the outpatient and inpatient is covered by the National Health Insurance in Japan. It is widely accepted that approximately 50% of women develop POP, but only 10–20% of them seek medical care for their condition [36].
Moreover, BMI was not detected as a risk factor of symptomatic POP in this study. Previous studies reported an association between high BMI (>25 kg/m2) and POP, and overweight was considered a risk factor of symptomatic POP [9,11]. Increased BMI was associated with a high prevalence of each pelvic floor disorder. It is important to evaluate the impact of overweight and obesity on pelvic floor disorders, as this is a modifiable risk factor in a population in which the prevalence of obesity was more than 35% in 2010 [37] and 40% in 2018 [38]. In contrast with the US, 21.1% of participants had BMI >25 kg/m2 in our study (Table 2). BMI does not affect the prevalence of POP probably due to the lower obesity rate among adult Japanese women; BMI >25 kg/m2 was observed in less than 19.2% women in 2015. There were no significant changes found in the prevalence of obesity in the last 10 years [34].
Previous research has shown that occupation type and heavy lifting at work are some of the risk factors of POP. [11,25] In order to clarify this, we selected full-time agricultural workers (agricultural work >150 days/year) who frequently engaged in gravity labor, part-time agricultural workers (agricultural work <150 days/ year), [39] JA office staff, and others (teachers, businesswomen, nurses, housewives, etc.) as participants. Our results indicated that the occupation type was not an independent factor affecting symptomatic POP. However, frequent lifting of heavy objects was an independent factor that significantly affected symptomatic POP. This result suggests that no matter what the occupation type, in order to prevent POP, women should avoid lifting heavy objects. Hence, mechanized or semi-mechanized equipment have been actively introduced in workplaces to reduce the physical load and improve women’s health.
Prior studies using multivariable logistic regression found that self-reported health status of fair or poor was strongly associated with symptomatic POP. In our study, the multiple regression model was used to determine the effect of POP on women’s self-reported health status. Self-reported health status was a dependent variable and POP symptoms and chronic conditions were independent variables. POP was significantly associated with self-reported health status and had a similar effect on the women’s health status as other chronic conditions did. Finally, we should pay attention to how POP affects the declining birth rate and aging society in Japan.
This study had several limitations. Because it was a crosssectional study, it could not determine the causal associations. Symptomatic POP was defined by self-reporting without confirmation by medical examination. Furthermore, the respondents were middle-aged and older women; therefore, the data may not be representative of younger women. Our study had a high non-response rate, which may have resulted in overestimation or underestimation of the actual prevalence of symptomatic POP.
Conclusions
Our results indicate that the epidemiological characteristics of POP among Japanese women predict that increase in parity, heaving lifting, constipation, and chronic conditions are its risk factors, but age, BMI, and occupation are not. Our results corroborated with those of several previous studies. Furthermore, like other chronic conditions, POP negatively affects women’s health status.
Acknowledgment
The authors thank the Japanese Agriculture Group for supporting this study, as well as Ms. Yoko Matsuyama and Tajiri Sakamoto for data collection and clerical support. This study was supported by JIKEI hospital (http://jikei-hp.or.jp/).
Disclosure
The authors have no conflicts of interest to disclose.
References
- Wu JM, Matthews CA, Conover MM, Virginia Pate, Michele Jonsson Funk (2014) Lifetime risk of stress urinary incontinence or pelvic organ prolapse surgery. Obstet Gynecol 123(6): 1201-1206.
- Rortveit G, Brown JS, Thom DH, Stephen K Van Den Eeden, Jennifer M Creasman, et al. (2007) Symptomatic pelvic organ prolapses: Prevalence and risk factors in a population-based, racially diverse cohort. Obstet Gynecol 109(6): 1396-1403.
- Fritel X, Varnoux N, Zins M, Gérard Breart, Virginie Ringa, et al. (2009) Symptomatic pelvic organ prolapse at midlife, quality of life, and risk factors. Obstet Gynecol 113(3): 609-616.
- Lukacz ES, Lawrence JM, Contreras R, Nager Charles W, Luber Karl M (2006) Parity, mode of delivery, and pelvic floor disorders. Obstet Gynecol 107(6): 1253-1260.
- Tinelli A, Malvasi A, Rahimi S, Roberto Negro, Daniele Vergara, et al. (2010) Age-related floor modification and prolapse risk factors in postmenopausal women. Menopause 17(1): 204-212.
- Greer WJ, Richter HE, Bartolucci AA, et al. (2008) Obesity and pelvic floor disorders. Obstet Gynecol 112: 341-349.
- Jelovsek JE, Maher C, Barber MD (2007) Pelvic organ prolapse. Lancet 369: 1027-1038.
- Jørgensen S, Hein HO, Gyntelberg F (1994) Heavy lifting at work and risk of genital prolapse and herniated lumbar disc in assistant nurses. Occup Med 44(1): 47-49.
- Kudish BI, Iglesia CB, Solok RJ, Barbara Cochrane, Holly E Richter, et al. (2009) Effect of weight change on natural history of pelvic organ prolapse. Obstet Gynecol 113(1): 81-88.
- Whitcomb EL, Rortveit G, Brown JS, Creasman Jennifer M, Thom David H et al. (2009) Racial differences in pelvic organ prolapse. Obstet Gynecol 114(6): 1271-1277.
- Swift S, Woodamn P, O’Boyle A, Margie Kahn, Michael Valley, et al. (2005) Pelvic organ support study (POSST): The distribution, clinical definition, and epidemiologic condition of pelvic organ support defects. Am J Obstet Gynecol 192(3): 795-806.
- Barber MD, Neubauer NL, Klein-Olarte V (2006) Can we screen for pelvic organ prolapse without a physical examination in epidemiologic studies? Am J Obstet Gynecol 195(4): 942-948.
- Cam C, Sakalli M, Ay P, Turhan Aran, Meltem Cam, et al. (2007) Validation of the prolapse quality of life questionnaire (P-QOL) in a Turkish population. Eur J Obstet Gynecol Reprod Biol 135(1): 132-135.
- Tegerstedt G, Maehle-Schmidt M, Nyrén O, Margareta Hammarström (2005) Prevalence of symptomatic pelvic organ prolapse in a Swedish population. Int Urogynecol J 16(6): 497-503.
- Fukumoto Y, Uesaka Y, Yamamoto K, Shinichiro Ito, Masaki Yamanaka, et al. (2008) Assessment of quality of life in women with pelvic organ prolapse: Conditional translation and trial of P-QOL for use in Japan. Nihon Hinyokika Gakkai Zasshi 99(3): 531-542.
- Baessler K, O’Neill SM, Maher CF, Diana Battistutta (2010) A validated self-administered female pelvic floor questionnaire. Int Urogynecol J 21: 163-172.
- Barber MD, Walters MD, Bump RC (2005) Short forms of two condition-specific quality-of-life questionnaires for women with pelvic floor disorders (PFDI-20 and PFTQ-7). Am J Obstet Gynecol 193(1): 103-113.
- Tegerstedt G, Miedel A, Maehle-Schmidt M, Olof Nyren, Margareta Hammarström (2005) A short-form questionnaire identified genital organ prolapse. J Clin Epidemiol 58(1): 41-46.
- Mouritsen L, Larsen JP (2003) Symptoms, bother and POPQ in women referred with pelvic organ prolapse. Int Urogynecol J 14: 122-127.
- Hasuda T, Ueda A, Wei CN (2017) Prevalence of symptomatic pelvic floor disorders among Japanese women. J Womens Health Care 6: 1-6.
- Mant J, Painter R, Vessey M (1997) Epidemiology of genital prolapse: Observations from the Oxford Family Planning Association study. Br J Obstet Gynaecol 104(5): 579-585.
- (2000) Progetto Menopausa Italia Study Group. Risk factors for genital prolapse in non-hysterectomized women around menopause. Results from a large cross-sectional study in menopausal clinics in Italy. Eur J Obstet Gynecol Reprod Biol 93: 135-140.
- Kumari S, Walia I, Singh A (2000) Self-reported uterine prolapse in a resettlement colony of north India. J Midwifery Womens Health 45(4): 343-350.
- Olsen AL, Smith VJ, Bergstrom JO, J C Colling, A L Clark (1997) Epidemiology of surgically managed pelvic organ prolapse and urinary incontinence. Obstet Gynecol 89(4): 501-506.
- Smith FJ, Holman CD, Moorin RE, Nicolas Tsokos (2010) Lifetime risk of undergoing surgery for pelvic organ prolapse. Obstet Gynecol 116(5): 1096-1100.
- Berber MD, Maher C (2013) Epidemiology and outcome assessment of pelvic organ prolapse. Int Urogynecol J 24(11): 1783-1790.
- Nygaard I, Bradley C, Brandt D, Women's Health Initiative (2004) Pelvic organ prolapse in old women: Prevalence and risk factors. Obstet Gynecol 104(3): 489-497.
- Tegerstedt G, Miedel A, Maehle-Schmidt M, Olof Nyrén, Margareta Hammarström (2006) Obstetric risk factors for symptomatic prolapse: A population-based approach. Am J Obstet Gynecol 194(1): 75-81.
- Weber AM, Walters MD, Ballard LA, Delbert L.Booher, Marion R Piedmonte (1998) Posterior vaginal prolapse and bowel function. Obstet Gynecol 179(6): 1446-1450.
- Samuelsson EC, Victor FT, Tibblin G, K F Svärdsudd (1999) Signs of genital prolapse in a Swedish population of women 20 to 59 years of age and possible related factors. Am J Obstet Gynecol 180(2 Pt 1): 299-305.
- Tokui N, Minari Y (2012) Definition of constipation and constipation constitution. Proceedings of PAMD Institute of Nakamura Gakuen University 5: 49-54.
- Kasugai K, Adachi K, Izawa S (2019) Research for actual situation of constipation in the Japanese. Jpn J Gastroenterol 116: 913-926.
- Fukuda H, Matsushima Y (2005) Studies on constipation and eating behavior of university students. Annual report Faculty of Human and Cultural studies, Tezukayama Gakuin University 7: 91-97.
- (2015) Ministry of Health, Labor, and Welfare [Internet]. Summary of National Health and Nutrition Survey Results.
- Wu JM, Vaughan CP, Goode PS, David T Redden, Kathryn L Burgio et al. (2014) Prevalence and trends of symptomatic pelvic floor disorders in U.S. women. Obstet Gynecol 123(1): 141-148.
- Maher C, Baessler K, Barber M, et al. (2013) Pelvic organ prolapse surgery. In: International Consultation on Incontinence, 5th edn. Paris: Health Publication Ltd.
- Ogden CL, Carroll MD, Kit BK, Flegal KM (2012) Prevalence of obesity in the United States, 2009-2010. NCHS Data Brief p. 82.
- (2020) Prevalence of obesity and severe obesity among adults: United States, 2017-2018. NCHS Data Brief 360: 1-8.
- Ministry of Agriculture, Forestry, and Fisheries, Japan.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.