Case Report 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Critical Complication Post Percutaneous Coronary Intervention: Acute Stent Thrombosis
*Corresponding author: Ching-Wen Wei, RN, NP, PhD, Assistant Professor, Department of Nursing, Tzu Chi University of Science and Technology, Hualien, ROC, Taiwan.
Received: December 01, 2021; Published: January 19, 2022
DOI: 10.34297/AJBSR.2022.15.002103
Abstract
Acute stent thrombosis (AST) is a rare but lethal complication that occurs after an individual receives coronary arteries stenting. If overlooked, it may lead to cardiogenic shock and ultimately death. The authors present a case of a 65-year-old male with coronary artery disease who experienced acute chest pain one hour after receiving percutaneous coronary intervention (PCI) with stenting. The nurse practitioner and the cardiac team were able to quickly diagnose AST through clinical signs, electrocardiography (ECG) and emergent coronary angiogram (CAG). The case subsequently underwent a second PCI for reperfusion of coronary arteries, combined with dual antiplatelets therapy (DAPT), and was successfully discharged afterwards. In patients who receive PCI, there is a 0.5% to 1 % chance that AST may occur within the first 24 hours. In patients experiencing chest pain or tightness, he or she should be monitored with 12-leads ECG every 15 to 30 minutes. CAG should be performed early to check the stented vessels for occlusions. DAPT may be initiated well before the first PCI in patients who are prone to thrombotic events to early prevent AST.
Keywords: Acute stent thrombosis, coronary heart disease, Percutaneous Coronary intervention with stenting, Chest pain, Dual antiplatelet therapy
Introduction
The academic research consortium criteria (ARC criteria) defines AST as thrombosis detected within the 5mm proximal or distal to the newly placed coronary stents during coronary angiogram [1]. In the situation where a patient begins to have chest symptoms, physicians should be on high alert for acute stent thrombosis [2]. AST has a prevalence of 0.5 to 1 %, with a mortality rate up to 20 to 45 %. Post-stenting thrombotic events can lead to myocardial infarction, deadly cardiac arrhythmia and irreversible heart failure. Even with early intervention, large areas of myocardial infarction and heart failure leads to poor prognosis [3,4].
Risk factors of AST include low left ventricular ejection fraction (LVEF), unstable angina, coagulopathy, diabetes mellitus and End- Stage Renal Disease (ESRD) [5,6]. Clinically, patients may present with conscious disturbance, low blood pressure, shortness of breath, reduced urine output and poor perfusion of peripheral vessels. If left unrecognized, progressive damage to heart function will occur [3]. Therefore, it is of paramount that first line workers are constantly monitoring patient’s general condition, ECG changes, and variation of lab data such as cardiac enzymes and renal function [7]. Differentiating AST from occlusion of other vessels or end coronary vessel thrombosis is also important as their clinical signs may be similar [5]. This is a 65-year-old male patient developed chest symptoms one hour after receiving PCI. Although the nurse practitioner suspected it might be acute coronary syndrome (ACS), his symptoms were not typical and the ECG and the cardiac enzymes were within normal limits, the diagnosis of AST was thus delayed.
Case report
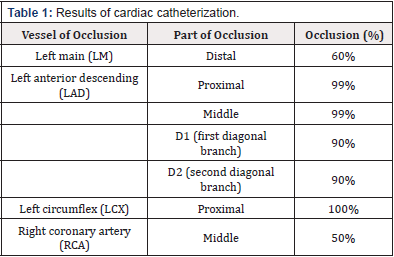
This is a 65-year-old male with a known medical history of hypertension, hyperuricemia and hyperlipidemia. Patient first complained of intermittent chest pain after exertion for one year. His chest pain is limited to the left anterior chest and is associated with dyspnea and cold sweating. Exercising ECG revealed ST segment depression with chest tightness, compatible with coronary artery disease. Coronary angiography showed severe occlusion of left anterior descending (LAD), left circumflex (LCX), and right coronary artery (RCA) (Table 1). Three Drug-eluting stents (DES) were deployed from Left main (LM) to LAD, LAD proximal to middle, LAD middle to distal. Balloon angioplasty was done to LM and LCX.
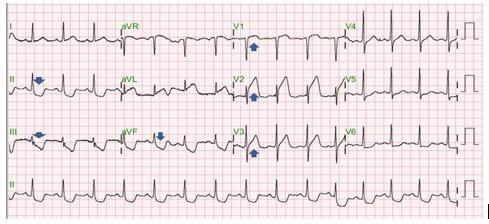
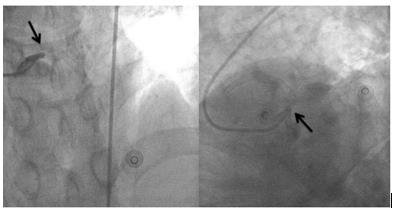
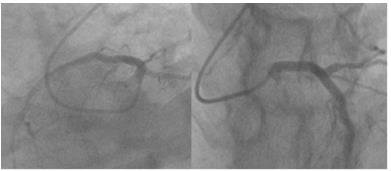
An hour after PCI, the patient begins to complain of chest pain for about 15 minutes. The pain is limited to the left anterior chest wall and described as dull pain with a pain score of 2. No radiation of pain to the chin, shoulder, upper arm or back is noted. Aggravated on exertion but by lying supine the pain may be alleviated. Pointof- care ultrasound of heart ruled out the possibility of cardiac tamponade and pericardial effusion. Chest X-ray revealed no engorgement of the aorta and the mediastinum. No water-bottle sign was seen. Laboratory work up found no elevation of creatine kinase (CK), creatine kinase-MB (CK-MB), or Troponin-I. 12 leads ECG showed no obvious ischemic change of the heart. His chest pain worsened. Follow-up cardiac enzymes were within normal limits, but ECG showed ST segment elevation in precordial leads V1 to V3. Mirroring leads in II, III, aVF showed ST segment depression (Figure 1). Based on these findings, a ST segment elevation myocardial infarction (STEMI) was diagnosed. Since the patient received PCI in the last 24 hours, and the anatomical location of ischemic change according to the ECG corresponds to previously stented locations. Acute stent thrombosis was highly suspected as a complication post coronary arteries intervention. Since the patient continues to be symptomatic and acute stent thrombosis was diagnosed. A second PCI was performed and found a large amount of thrombus accumulating at previously placed stents in LM to LAD and LM to LCX, causing complete blockage of arterial perfusion distal to LM (Figure 2 & 3). Confirming the scenario of acute stent thrombosis, and discharged after second PCI 1 week.
Discussion
Acute stent thrombosis (AST) denotes an ischemic change to the heart due to thrombosis of recently placed coronary artery stents within the first 24 hours of PCI. Based on the time of events can be classified into early (acute or subacute), late, and very late stage [8]. Among these, the acute AST is the rarest but also deadliest with mortality rate up to 45% [2]. When patient complains of chest symptoms, continuous monitoring of ECG is essential. In Taiwan, our cardiac association recommends ECG to be obtained within the first 10 minutes of symptoms. If the first ECG was insignificant or inconclusive, a 12 leads ECG should be obtained for every 15 to 30 minutes [9]. If clinically confirmed or highly suspected, early coronary angiogram should be performed in establishing early diagnosis and treatment [6]. Close monitoring of input and output (I/O), bodyweight, and cardiac sonography for signs of heart failure after receiving PCI are considered as routines. Prescribing statins early with a target LDL less than 70 mg/dl to help reduce cardiac mortalities and ischemic stroke of the brain [10]. Beta-blockers should be given to patients without pulmonary edema, signs of shock, and who are hemodynamically stable to improve left ventricle function and overall survival [11]. Angiotensin- converting enzyme inhibitors (ACEI) or ARB reduces 30-day mortalities in patients with a LVEF <40%, diabetics, and hypertension [10].
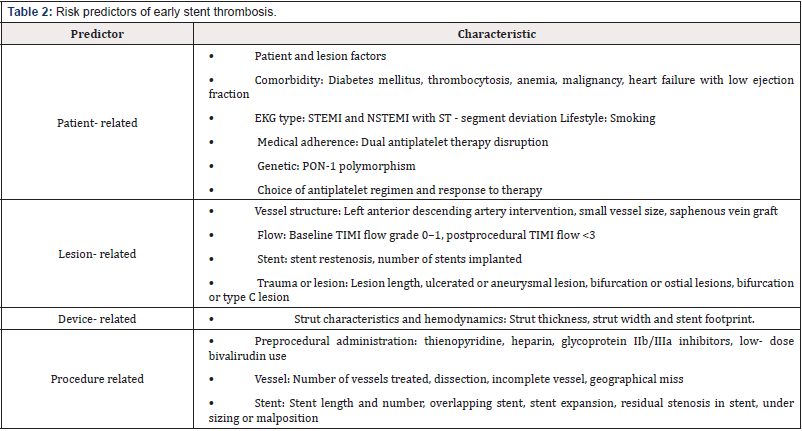
Several causes of AST have been established in the past. Based on a systematic review, predictors of early ST were include patient, lesion, device, and procedure (Table 2) [12]. Dangas et al. [13] based on two large RCTs (HORIZONS-AMI trial and ACUITY trial), developed a Stent Thrombosis Risk Score that predicts the risk of ST in patients post PCI. The score evaluates baseline platelet count, insulin-dependent diabetes mellitus, baseline thrombolysis in myocardial infarction (TIMI)flow grade 0-I, early anticoagulant therapy (pre-PCI), ulcerated or aneurysmal lesion, number of treated vessels, and final TIMI flow grade < III. Kumar et al. [14] examined the validity of the score in 569 patients who had STEMI and underwent PCI and found the risk score was significantly higher in those who developed AST. Baseline platelet count and insulin-dependent diabetes mellitus were statistically significant in predicting AST. Patients with a total score of 5 or higher were at a 5-fold risk than 0 those with a scoreless or equal to 2. Demonstrating a linear relationship between the score and the risk of developing AST, with a specificity of 91% and sensitivity of 30%.
The medical team should focus on modifiable risk factors such as types of stents. New generations of stents provide better functionalities, durability, and less complications. New DES significantly reduce stent restenosis and inflammation of vessels; biodegradable polymer-coated and polymer-free devices reduce polymer induced reactions; self-expandable stents for bifurcation lesions. When main vessels are found to have occlusions, IVUS or Optical coherence tomography (OCT) can be used for optimal stent size choice to reduce the effect of procedure-related factors [15]. Acute stent thrombosis can be linked to P2Y12, a chemoreceptor for adenosine diphosphate (ADP). Antiplatelet or anticoagulation agents should be administered early to prevent AST [16]. Drugs including Aspirin which inhibits TAX2 in prevention and treatment of ACS and ischemic stroke. Clopidogrel and Ticagrelor bind to ADP P2Y12 receptor to prevent platelet activation. Glycoprotein IIb/IIIa inhibitors such as Abciximab, Tirofiban, Eptifibatide for unstable angina and NSTEMI. In 2018, European Heart Association guidelines was reported, in patients without bleeding tendency and previous bleeding histories in prevention of AST before PCI as a dual antiplatelet therapy [17].
Conclusion
Our case experienced acute chest symptoms one hour after PCI. This case was educational to first line workers, which keep monitor ECG post PCI to early detect of AST was important if patient complained chest pain. It could be help minimize the damage done to the myocardium and prompt early intervention and reperfusion and ultimately prevent mortality.
References
- Cutlip DE, Windecker S, Mehran R, Boam A, Cohen DJ, et al. (2007) Clinical end points in coronary stent trials. Circulation 115(17): 2344-2351.
- Reejhsinghani R, Lotfi AS (2015) Prevention of stent thrombosis: challenges and solutions. Vasc Health Risk Manag 11: 93-106.
- Sommer P, Armstrong EJ (2015) Stent thrombosis: current management and outcomes. Curr Treat Options Cardiovasc Med 17(3): 365.
- Tyczyński P, Karcz MA, Kalińczuk L, Fronczak A, Witkowski A, et al. (2014) Early stent thrombosis. Aetiology, treatment, and prognosis. Postepy Kardiol Interwencyjnej 10(4): 221-225.
- Wada H, Mattson PC, Iwata H (2017) Stent or scaffold thrombosis: past, current and future perspectives. EMJ 5: 55-61.
- Zhang F, Tongo ND, Hastings V, Kanzali P, Zhu Z, et al. (2017) ST-segment elevation myocardial infarction with acute stent thrombosis presenting as intractable hiccups: an unusual case. Am J Case Rep 18: 467-471.
- Modi K, Soos MP, Mahajan K (2021) Stent Thrombosis. StatPearls [Internet]. Treasure Island (FL): Stat Pearls Publishing.
- Ullrich H, Münzel T, Gori T (2020) Coronary stent thrombosis- predictors and prevention. Dtsch Arztebl Int 117(18): 320-326.
- Li YH, Wang YC, Wang YC, Liu JC, Lee CH, et al. (2018) 2018 Guidelines of the Taiwan Society of Cardiology, Taiwan Society of Emergency Medicine and Taiwan Society of Cardiovascular Interventions for the management of non-ST-segment elevation acute coronary syndrome. J Formos Med Assoc 117(9): 766-790.
- Ibanez B, James S, Agewall S, Antunes MJ, Bucciarelli-Ducci C, et al. (2018) 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: The Task Force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC). Eur Heart J 39(2): 119-177.
- Bugiardini R, Cenko E, Ricci B, Vasiljevic Z, Dorobantu M, et al. (2016) Comparison of early versus delayed oral β blockers in acute coronary syndromes and effect on outcomes. Am J Cardiol 117(5): 760-767.
- Gori T, Polimeni A, Indolfi C, Räber L, Adriaenssens T, et al. (2019) Predictors of stent thrombosis and their implications for clinical practice. Nat Rev Cardiol 16(4): 243-256.
- Dangas GD, Claessen BE, Mehran R, Xu K, Fahy M, et al. (2012) Development and validation of a stent thrombosis risk score in patients with acute coronary syndromes. JACC Cardiovasc. Interv 5(11): 1097-1105.
- Kumar R, Tariq S, Fatima M, Saghir T, Batra MK, et al. (2020) Validity of the stent thrombosis risk score in predicting early stent thrombosis after primary percutaneous coronary intervention. J Saudi Heart Assoc 32(2): 256-262.
- Maehara A, Matsumura M, Ali Ziad A, Mintz Gary S, Stone Gregg W, et al. (2017) IVUS-Guided versus OCT-Guided coronary stent implantation. J Am Coll Cardiol 10(12): 1487-1503.
- Ge J, Yu H, Li J (2017) Acute coronary stent thrombosis in modern era: etiology, treatment, and prognosis. Cardiology 137(4): 246-255.
- Neumann FJ, Sousa-Uva M, Ahlsson A, Alfonso F, Banning AP, et al. (2019) 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur Hear J 40(2): 87-165.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.