Case Report 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Diabetic Foot Infection Caused by Pasteurella Multicide: A Case Report and Review of The Literature
*Corresponding author: Ming Hui Li, Department of Infection, The first Affiliated Hospital of Shaoxing University, China.
Received: January 18, 2022; Published: January 28, 2022
DOI: 10.34297/AJBSR.2022.15.002111
Abstract
Pasteurella multicide is the most common cause of human soft tissue infection, usually associated with domestic animals contact history. Reports of diabetic foot infections caused by Pasteurella multicide without contact history are rare. Here we report the case of a 51-year-old man with diabetic foot infection who was cured with penicillin. A variety of antibiotics were replaced after admission, his body temperature and right lower extremity swelling had not improved, and symptoms such as heart failure, anemia and renal insufficiency occurred. Finally, Pasteurella multicide was detected by next-generation sequencing and he got better after the use of penicillin. In Non-contact cases, diabetic foot infection caused by Pasteurella multicide may be related to impaired immunity.
Keywords: Pasteurella multicide; Diabetic foot infection; Impaired immunity; Next generation sequencing; Case report
Abbreviations: PM: Pasteurella Multicide; DFI: Diabetic Foot Infection; NGS: Next-Generation Sequencing; WBC: White Blood Cell Count; NE: Neutrophil Percentage; hsCRP: High-Sensitivity C-Reactive Protein; PCT: Procalcitonin; GHbA1c: Glycosylated Hemoglobin A1c; ESR: Erythrocyte Sedimentation Rate; NT-proBNP: N-Terminal Fragment of Brain Natriuretic Peptide Precursor; DM: Diabetes Mellitus; HF: Heart Failure; MODF: Multiple Organ Dysfunction; CSF: Cerebrospinal Fluid; BALF: Bronchoalveolar Lavage Fluid; RA: Rheumatoid Arthritis; CAP: Community Acquired Pneumonia; COPD: Chronic Obstructive Pulmonary Disease
Introduction
Pasteurella multicide (PM), which is mainly colonized in the oral cavity, nasopharynx, and upper respiratory tract of domestic and wild animals, was first isolated by Pasture in 1881 for the use of the development of a vaccine [1]. PM is a Gram-negative coccus that grows well on 5% sheep blood agar at 37℃ [2], and can be divided into five groups (A, B, D, E and F) according to the serotype classification of capsular polysaccharides [3]. Generally, human infections are caused by A and D, the most common causes of soft tissue infections after being scratched or bitten by domestic animals like dogs and cats [4]. In addition, pulmonary and abdominal infections, and other rare invasive manifestations (such as bacteremia, meningitis, endocarditis, and ocular infections) can also occur in neonates, the elderly, and immunocompromised individuals [5]. PM is sensitive to penicillin, which is often used as a first-line treatment, second-and third generation cephalosporins, and fluoroquinolones or tetracyclines as alternatives [6].
Diabetic foot infection (DFI) is one of the severe complications of diabetes precipitated by neuropathic or neuroischemic feet, which increases the risk of lower-extremity amputation and mortality [7]. There are few reports of PM regarding immunocompromised hosts. In this paper, a case of diabetic foot infected with PM without a contact history was reported and the related literatures were reviewed.
Case Presentation
A 51-year-old male patient presented to our emergency department complaining of recurrent fever and swelling and pain in his right lower extremity. He also felt chills, fatigue, and muscle soreness. The symptoms began to recur a week ago and gradually aggravated. The swelling range of the right lower limb spread from the ankle to the beneath of knee joint. He is a self-employed, with a history of type 2 diabetes for 17 years and was hospitalized for DFI in our hospital one year ago. After cefotaxime and levofloxacin intravenous anti-infection and insulin with acarbose for lowering blood glucose, he was improved and discharged. He used insulin and acarbose to control blood glucose at home, but the blood glucose level was rarely monitored. There is no special family history or genetic disease history, no history of surgery and trauma, no history of infectious diseases, no animal contact history.
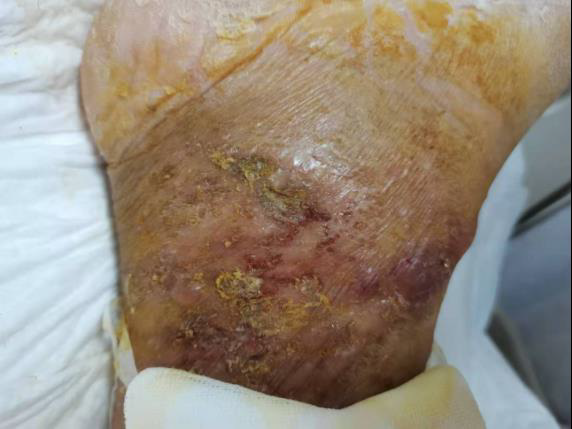
The relevant examinations were quickly performed in the emergency department: WBC 14.24 *109/L, NE 89.5%, hsCRP 310.9mg/L, PCT 90.08ng/ml, GHbA1C 9.20%. Glucose and protein were detected in urine, consistent with the diagnosis of diabetes. D-dimers and arterial blood gas were normal. After intravenous administration of 1g q8h of Imipenem-cilastatin, he still had low fever and distending pain in his right lower extremity. On admission, His temperature was 38.8°C, heart rate was 100 bpm, respiratory rate was 19 breaths per minute, blood pressure was 164/112 mmHg. There was a small ulcer measuring approximately 2*1cm in his right lower extremity with high skin temperature and pus exudation could be seen. The dorsalis pedis artery pulsation existed. Re-examination in the ward: CRP 152.41mg/L, PCT 10.03ng/ml, ESR 108mm/h. Right ankle CT showed soft tissue swelling around. Deep vein ultrasound revealed muscular calf vein thrombosis of right, and the swelling just decreased slightly after anticoagulation with low molecular weight heparin. Echocardiography indicated mild mitral and tricuspid regurgitation and reduced left ventricular diastolic function. No abnormal lesions were found in abdominal, and chest enhanced CT scans. Empirical anti-infection therapy including intravenous linezolid 0.6g q12h combined with piperacillin-tazobactam 4.5g q12h was administered, along with symptomatic treatments such as diuresis and active control of blood glucose. His inflammatory indices gradually decreased, but symptoms remained unremarkable. The patient then asked to be transferred to the superior hospital for further treatment.
Moxifloxacin 0.4gqd and cefazolin sodium 1g q8 were given intravenously as initial treatments in the new hospital and blood glucose was controlled at 5.5-7.2 mmol/L. Blood cultures and pus cultures were further performed, which turned out negative. An MR plain scan of the ankle joint showed obvious swelling of soft tissue around the right ankle joint and effusion around the tendon of the right peroneal longus, peroneal shortus, and hallux long flexor. In addition, bone marrow edema was seen in the right calcaneus, talus, and distal tibia. In combination with the high inflammation indicators and magnetic resonance report, the diagnosis was considered as DFI.
Thus, under ultrasound guidance, 1.5ml of reddish liquid was sucked from the swelling site, and 53 Pasteurella multicide sequences were checked by NGS (Figure 1). Subsequently, the antibiotics were promptly changed to 6.4 million U penicillin in combination with levofloxacin 0.5g qd according to the NGS and drug susceptibility results. At this point, it could be observed that his right lower extremity swelling gradually improved, and inflammation indicators dropped rapidly. NGS re-examination only detected PM 2 sequences, suggesting that the treatments were effective.
However, in the course of disease, other thorny problems arose. There was a continuous increase in NT-proBNP, whereas a progressive decline in the hemoglobin (Figure2A, 2B). Transthoracic cardiac ultrasound indicated multiple valve regurgitation and a moderate decrease in left ventricular diastolic function. Bone marrow biopsy showed more than ten medullary cavities, with hematopoietic cells accounting for about 30%, and visible megakaryocytes, as well as a hemopoietic system without obvious abnormalities. Through consultation with the Cardiology and Hematology departments, the cardiac damage may have resulted from long-term ischemia and hypoxia as the patient was in a state of severe anemia. After intensive nutrition, iron supplementation, and intermittent infusion of red blood cell suspension transfusions, the NT-proBNP was maintained at a slightly elevated level.
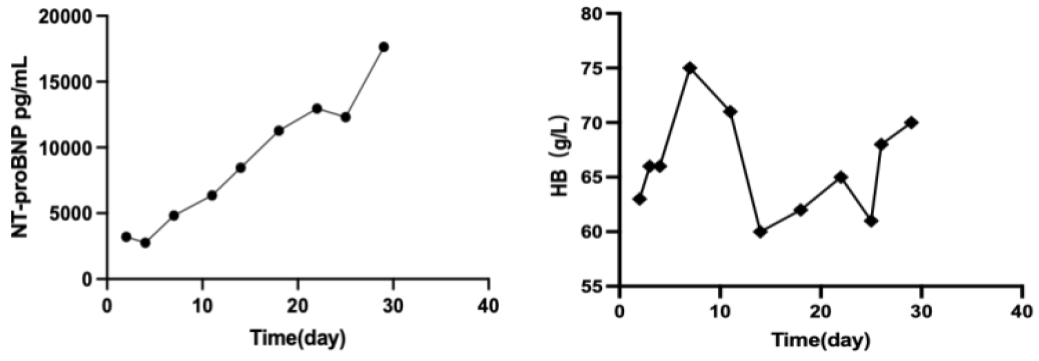
Figure 2: (A) NT-proBNP gradually increased as long-term anemia led to myocardial ischemia and hypoxia injury. (B) Hemoglobin fluctuated between 60-75g/L, in a state of moderate anemia.
After his condition stabilized, the patient was transferred back to local hospital to continue the consolidate treatment, and ultimately discharged free of symptoms, on injection Benzylpenicillin 1.2 million units per month for 2 months.
Discussion
We undertook a focused review of literatures previously reported on PM infection in diabetic patients. A total of 31 articles were retrieved, from which 22 case reports were picked out, including 16 cases with animal contact history: 12 cases with scratched and bitten history and 4 cases with licking history. 5 cases had no contact history, 1 case was unknown. The clinical manifestations were mainly soft tissue infections to varying degrees, and invasive manifestations such as respiratory tract infection, meningitis, intra-abdominal infection, and bacteremia could also be seen. Related cases are shown in [Table1].
PM often resides in the nasopharynx and respiratory tract of animals [25]. Human soft tissue infections can be caused after being scratched or bitten by animals, and even pyogenic arthritis or osteomyelitis can occur in severe cases. Licks of cats and dogs can also cause soft tissue infection, but often occur in artificial joints, which possibly may be contacted with secretions during feeding [18]. Non-contact infection associated with polysaccharide capsules is usually prevalent in the immunocompromised, such as diabetes, cirrhosis, and dialysis [26]. In addition, it is reported that PM is colonized in the respiratory tract of COPD patients, making them more susceptible to infection [27]. In this case, the patient was neither bitten by a cat or dog, nor exposed to secretions, nor changed artificial joints, but the NGS result did show PM infection. Given our patient’s long history of diabetes, we consider that the infection was caused by diabetes-induced immunodeficiency. Therefore, it is extremely important to adequately control the blood glucose of patients with diabetes and enhance their immunity.
DFI is a common complication in diabetic patients, manifested as a bone or soft tissue infection below the ankle [28]. According to the updated IWGDF [29] (The International Working Group on the Diabetic Foot) guideline, treatment should be based on clinical characteristics, severity of infection, possible pathogens or cultures, recent antibiotic history, and local antibiotic resistance. This patient was diagnosed as severe infection because he had systemic inflammatory response syndrome and bone marrow edema in his right ankle. With regard to pathogens, given the predominant distribution of Gram-negative bacteria (such as Escherichia coli) in China and the poor initial treatment, the antibiotic was adjusted to broad-spectrum antibacterial piperacillin-tazobactam combined with linezolid to cover methicillin-resistant staphylococcus aureus (MRSA) for empiric therapy. Nevertheless, he did not appear to be improving and there was a progressive decline in the blood routine test, possibly as a side effect of linezolid. Thanks to the NGS report [30], antibiotics were ultimately changed to penicillin and levofloxacin. Consequently, the patient’s conditions were markedly improved, thereby underscoring the importance of identification of the exact pathogens during empirical treatment.
Conclusion
Soft tissue infections induced by PM usually have domestic animal contact history, while non-contact ones are often in immunodeficient patients. Thus, it is very important to identify the pathogens promptly and timely by using the existing means and technologies in clinical practice, such as NGS. At the same time, clinicians are supposed to pay more attention to the rational use of antibiotics to reduce antibiotic resistances.
Author Contributions
Di Yang reviewed the literature and contributed to manuscript drafting; Ming-Hui Li was responsible for the revision of the manuscript for important intellectual content; all authors issued final approval for the version to be submitted.
Statement Confirming Patient Consent
Patient provided and signed the informed consent form.
Competing Interest’s Statement
The authors have no competing interests.
References
- Hurtado R, Maturrano L, Azevedo V, Aburjaile F (2020) Pathogenomics insights for understanding Pasteurella multocida adaptation. Int J Med Microbiol 310(4):151417.
- Mogilner L, Katz C (2019) Pasteurella multocida. Pediatr Rev 40(2): 90-92.
- Peng Z, Wang X, Zhou R, Chen H, Wilson BA, et al. (2019) Pasteurella multocida: Genotypes and Genomics. Microbiol Mol Biol Rev 83(4): e00014-19.
- Guan L, Xue Y, Ding W, Zhao Z (2019) Biosynthesis and regulation mechanisms of the Pasteurella multocida capsule. Res Vet Sci 127: 82-90.
- Hasan J, Hug M (2021) Pasteurella Multocida. Treasure Island (FL): StatPearls Publishing, Island.
- Bourély C, Cazeau G, Jouy E, Haenni M, Madec JY, et al. (2019) Antimicrobial resistance of Pasteurella multocida isolated from diseased food-producing animals and pets. Vet Microbiol 235: 280-284.
- Pitocco D, Spanu T, Di Leo M, Vitiello R, Rizzi A (2019) Diabetic foot infections: a comprehensive overview. Eur Rev Med Pharmacol Sci 23(2 Suppl): 26-37.
- Grehn M, Müller F, Hany A, Meier P (1984) Pasteurella multocida septicemia not associated with primary liver disease. Eur J Clin Microbiol 3(3): 258-260.
- Tsunematsu Y (1980) [Toxoplasmosis]. Kansenshogaku Zasshi 54(1): 1-2.
- Tattevin P, Souala F, Gautier AL, Rauch M, Bouet J, et al. (2005) Diabetes in patients with pasteurellosis. Scand J Infect Dis 37(10): 731-733.
- Yada T, Arashima Y, Kawano K, Fukui T (1991) A case of Pasteurella multocida subsp. multocida complicated with diabetes mellitus. Kansenshogaku Zasshi 65(10): 1365-1368.
- Fajfar Whetstone CJ, Coleman L, Biggs DR, Fox BC (1995) Pasteurella multocida septicemia and subsequent Pasteurella dagmatis septicemia in a diabetic patient. J Clin Microbiol 33(1): 202-204.
- Jones AG, Lockton JA (1987) Fatal Pasteurella multocida septicaemia following a cat bite in a man without liver disease. J Infect 15(3): 229-235.
- Lee WS, Chen FL, Wang CH, Ou TY, Lin YH, et al. (2019) Community-acquired bacteremic pneumonia due to Pasteurella multocida subspecies multocida in a patient with poor-control diabetes mellitus. J Microbiol Immunol Infect 52(1): 163-164.
- Satomura A, Yanai M, Fujita T, Arashima Y, Kumasaka K, et al. (2010) Peritonitis associated with Pasteurella multocida: molecular evidence of zoonotic etiology. Ther Apher Dial 14(3): 373-376.
- Kukrety S, Parekh J, Townley T (2016) Pasteurella multocida Bacteremia in an Immunocompromised Patient. Case Rep Med 7392847.
- Schlichthaar H, Rohrer T, Schuster G, Lehnert H (1995) Interstitielle Pneumonie und Sepsis durch Pasteurella-multocida-Infektion. Dtsch Med Wochenschr 120(46): 1582-1586.
- Braithwaite BD, Giddins G (1992) Pasteurella multocida infection of a total hip arthroplasty. A case reports. J Arthroplasty 7(3): 309-310.
- Gonda H, Noda Y, Ohishi T, Tanigawa Y, Sato S, et al. (2001) A clinical study on patients detected Pasteurella multocida from sputum. Kansenshogaku Zasshi 75(9): 780-784.
- Yu GV, Boike AM, Hladik JR (1995) An unusual case of diabetic cellulitis due to Pasturella multocida. J Foot Ankle Surg. 34(1): 91-95.
- Milani-Nejad N, Tyler K, Grieco CA, Kaffenberger BH (2017) Pasteurella multocida ecthyma complicated by necrotizing fasciitis. Dermatol Online J 15: 23(4).
- Marcantonio YC, Kulkarni PA, Sachs S, Ting K, Lee J, et al. (2017) Disseminated Pasteurella multocida infection: Cellulitis, osteomyelitis, and myositis. IDCases 10: 68-70.
- Malek AE, Fares JE, Raad II, Ericsson C (2019) Pasteurella multocida vertebral osteomyelitis, myositis and epidural abscess in a diabetic cirrhotic patient. IDCases 18: e00630.
- Katechakis N, Maraki S, Dramitinou I, Marolachaki E, Koutla C, Ioannidou E, et al. (2019) An unusual case of Pasteurella multocida bacteremic meningitis. J Infect Public Health 12(1): 95-96.
- Pardal Peláez B, Sarmiento García A (2021) [Microbiology of infections caused by dog and cat bites: A review]. Rev Chilena Infectol 38(3): 393-400.
- Martin TCS, Abdelmalek J, Yee B, Lavergne S, Ritter M (2018) Pasteurella multocida line infection: a case report and review of literature. BMC Infect Dis 18(1): 420.
- Bhat S, Acharya PR, Biranthabail D, Rangnekar A, Shiragavi S (2015) A Case of Lower Respiratory Tract Infection with Canine-associated Pasteurella canis in a Patient with Chronic Obstructive Pulmonary Disease. J Clin Diagn Res 9(8): DD03-DD04.
- Saeed K, Esposito S, Akram A, Ascione T, Bal AM, et al. (2020) International Society of Antimicrobial Chemotherapy. Hot topics in diabetic foot infection. Int J Antimicrob Agents 55(6): 105942.
- Lipsky BA, Senneville É, Abbas ZG, Aragón Sánchez J, Diggle M, et (2020) International Working Group on the Diabetic Foot (IWGDF). Guidelines on the diagnosis and treatment of foot infection in persons with diabetes (IWGDF 2019 update). Diabetes Metab Res Rev 36 Suppl 1: e3280.
- Zhao N, Cao J, Xu J, Liu B, Chen D, et al. (2021) Targeting RNA with Next- and Third-Generation Sequencing Improves Pathogen Identification in Clinical Samples. Adv Sci (Weinh) 8(23): e2102593.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.