Research article 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Predictive Value of Monocyte Lymphocyte Ratio in Peritonitis Risk in Peritoneal Dialysis Patients and Its Body Mass Index: A Case-Control
*Corresponding author:Semahat Karahisar Şirali, Ankara Training and Research Hospital, Turkey.
Received: August 09, 2022; Published: August 25, 2022
DOI: 10.34297/AJBSR.2022.16.002298
Abstract
Background: The aim of our study was to determine the predictive value of monocyte to lymphocyte ratio in peritonitis risk in PD patients with and without chronic inflammation and its relationship with body mass index.
Methods: The pre-peritonitis attack monocyte lymphocyte ratio were compared in groups with and without peritonitis and relationship body mass index. SPSS 22.0 program was used and p<.05 was accepted as the significance level.
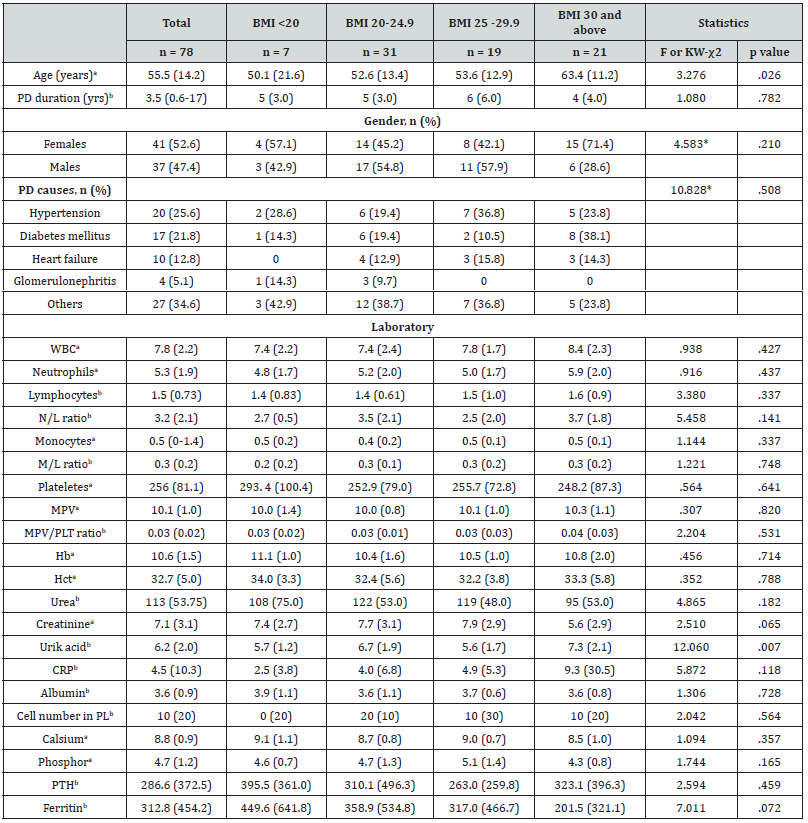
Results: The mean age of 78 patients was 55.5 ± 14.2 years, 51.3% (n = 40) were female. While 43.6% (n = 34) of the cases were diagnosed with peritonitis, 56.4% (n = 44) were diagnosed with not-peritonitis. CRP laboratory upper limit of 5mg/dL (with chronic inflammation) comprised 46.2% (n=36), while those below 5mg/dL and below constituted 53.8% (n=42). The monocyte lymphocyte ratio was found to be significantly higher in the group with peritonitis with chronic inflammation (t=2.51, p=.017), and a positive correlation was found between the mean annual CRP. In the not-peritonitis group with CRP below 5, the neutrophil lymphocyte ratio was found to be significant (z = 2.173, p = .03). When 4 groups were formed according to body mass index, only uric acid level was found to be significantly higher in the group with a BMI of 30 and above (7.3 vs. others; KW-χ2(3) = 12.060, p = .007).
Conclusion: A significantly higher monocyte-lymphocyte ratio before peritonitis attack in PD patients with chronic inflammation would well predict that it may have a predictive value in the diagnosis of peritonitis.
Introduction
Chronic Kidney Disease (CKD) is a common and serious disease associated with premature death and complications of end-stage renal disease. One in 7 to 10 adults worldwide has chronic kidney disease [1]. Peritoneal dialysis (PD) is one of the treatment methods of chronic kidney disease. Peritonitis is still common in PD patients despite technological advances [2]. Peritonitis, the most important complication of patients undergoing PD, is a serious cause of morbidity and mortality. Early diagnosis and prevention of this complication of peritoneal patients with routine follow-up is extremely important.
Although the mechanisms of inflammation and oxidative stress in CKD have not yet been definitively proven, uremic toxins, renin angiotensin aldosterone system (RAAS), comorbid conditions, volume load, hypertension, iron use, and antioxidant deficiency are the factors to blame [3,4]. CRP is a well-known marker of inflammation [5]. Neutrophil lymphocyte ratio (N/L), monocyte lymphocyte ratio (M/L) and platelet lymphocyte ratio (PLT/L) have also been defined as systemic inflammation markers and are associated with poor prognosis in many diseases [6].
It has been reported that high BMI values pose a significant risk for peritonitis and increase the risk of peritonitis with leakage, is associated with microorganism-specific peritonitis, and poses a risk for all-cause mortality [7-10]. Excess adipose tissue has recently been noted as a potential cause of chronic inflammation in CKD patients [11].
There is a chronic inflammation condition in patients with CKD and there is no method to reveal this directly. The aim of the study was to determine the value of monocyte to lymphocyte ratio in determining the risk of peritonitis in PD patients with chronic inflammation and its relationship with BMI.
Material and Method
Of 78 patients who underwent peritoneal dialysis followed in our clinic were evaluated retrospectively. In the last 2 years, 34 patients who had a peritonitis attack and 44 patients who did not have a peritonitis attack were included in the study. According to the International Society for Peritoneal Dialysis (ISPD), a diagnosis of peritonitis was made with abdominal pain, turbid dialysate, presence of more than 100 leukocytes in the peritoneal fluid and 50% of it fragmented. Subgroups were formed because CRP was above 5mg/dL (upper limit of normal) in some of the patients with and without peritonitis. According to the peritonitis agents, the patients were divided into three groups as gram-negative microorganism growth group, gram-positive microorganism growth group, and the group in which no microorganism could be grown in culture. Cases who did not experience any infection event in the last three months were included in the study, and the number of daily changes was four for the whole group and there was no change in number.
All hematological parameters of the participants such as complete blood count, including hemoglobin, hematocrit, white blood cell, neutrophil, lymphocyte, monocytes, neutrophil lymphocyte ratio, monocyte lymphocyte ratio, platelet, mean platelet volume (Sysmex XN 3000), as well as biochemical ones like urea, creatinine, uric acid, sensitive CRP such as albumin, calcium, phosphorus, PTH, ferritin, peritoneal cell count were tested during their routine controls (Cobas 8000 modular analyzer). Age, gender and comorbid diseases were recorded as basic demographic data. BMI was recorded as <20, between 20 and 24.9, 25 and 29.9, and 30-35. BMI was calculated as kilograms divided by height squared.
Study Groups
Laboratory data were compared between the group with peritonitis (P+) and the group without peritonitis (P-). Laboratory data were compared between P+ (Group 1) and P- group (Group 2) with CRP above 5mg/dL (chronic inflammation). Laboratory data were also compared between P+ (Group 3) and P- group (Group 4) with CRP 5mg/dL and below. Data were also compared between Groups 1 and 3 and between Groups 2 and 4. The relationship between BMI, which was divided into four groups, and the total group and subgroups was recorded.
The study complied with the ethical principles of the Declaration of Helsinki and was approved by the ethics committee of our hospital (E-19/44). The aim of the study was to predict the risk of peritonitis attack with the M/L ratio, which is one of the hematological parameters performed in routine controls and its relationship with BMI.
Statistical Analysis
SPSS 22.0 (NY, IMB Inc 2010) program was used in the analysis. Categorical variables were expressed as frequency (n) and percentage (%). Conformity of continuous variables to normal distribution was tested with Shapiro-Wilk and expressed in terms of arithmetic mean, standard deviation, median, minimum and maximum values. The student-t test was used for the comparison of those with normal distribution among continuous variables, while those without normal distribution were analyzed with the Mann- Whitney U test and Kruscall-Wallis Pearson-χ2 and Fisher’s exact tests were used for categorical variables. P<.05 was accepted as the significance level. ROC (Receiver operating characteristic) analysis was used to determine the diagnostic value of M/L and N/L ratios.
Results
The mean age of 78 patients was 55.5±14.2 years, with a minimum age of 25 and a maximum of 87 years. 51.3% (n = 40) of the cases were female and 48.7% (n = 38) were male. While 43.6% (n = 34) of the cases were diagnosed with peritonitis, 56.4% (n = 44) were diagnosed with not-peritonitis. The two groups were found to be similar in terms of age and gender proportion (p>.05, Table 1).
The duration of peritoneal dialysis of the cases was a minimum of 6 months and a maximum of 17 years, and the median level was 3.5 years. PD durations of peritonitis and not-peritonitis groups were found to be similar (Table 1).
Reasons for patients receiving peritoneal dialysis treatment were 25.6% (n = 20) hypertension, 21.8% (n = 17) diabetes, 12.8% (n = 10) heart failure, 5.1% (n = 4) glomerulonephritis and 34.6% (n = 27) were other reasons. In the comparison between the two groups, the hypertension rate in the not-peritonitis group was significantly higher than in the peritonitis group (38.6% vs. 8.8%, χ2 (4) = 15,658, p = .002, Fisher’s exact test) (Table 1).
Conclusion
Comparison of Laboratory Parameters
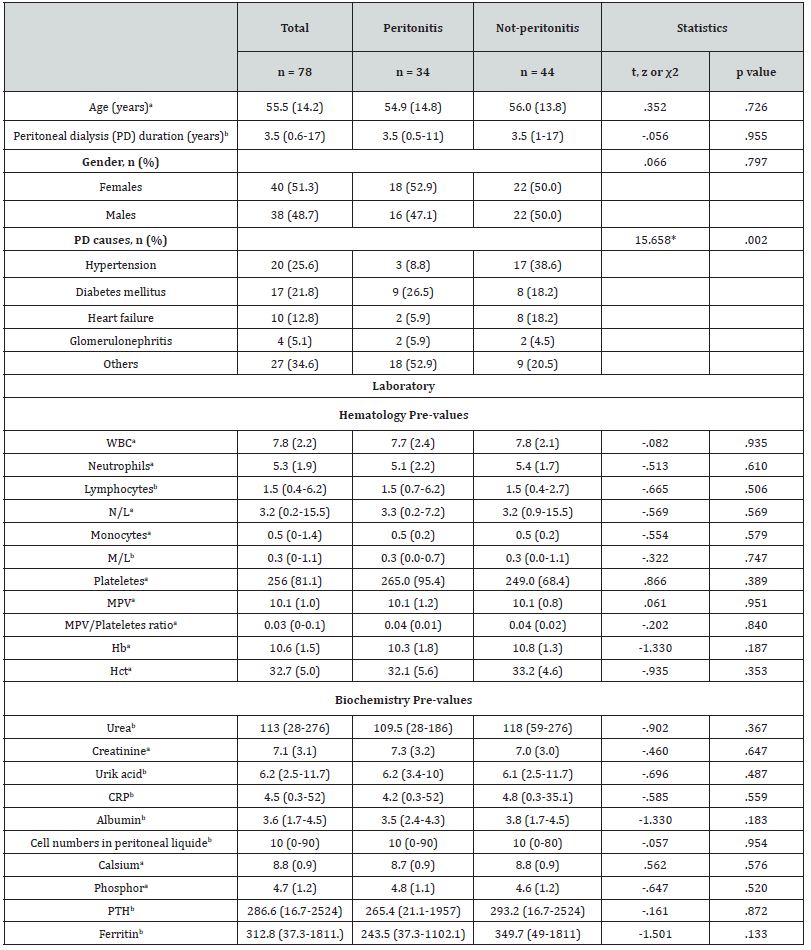
Table 1 shows the comparison of laboratory data of cases with peritonitis (n=34) and not-peritonitis (n=44). Accordingly, all hematological and biochemical parameters were found to be similar in the two groups (p>.05 for all). In the comparison of groups 1 and 2, the M/L ratio was found to be significantly higher in group 1 (t=2.51, p=.017) and the number of monocytes was high (t=2.20, p=.035). There was no significant difference between other parameters (Table 2).
a: Arithmetic mean (standard deviation), b: Median (Interquartile range).
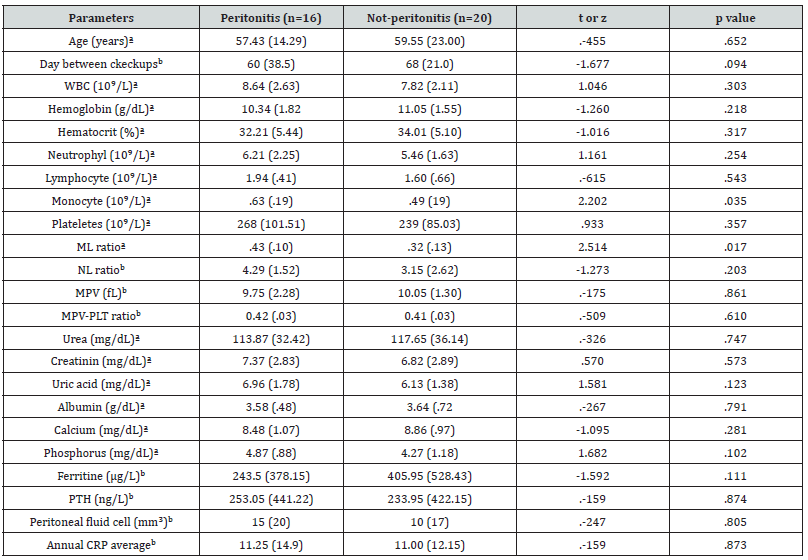
N/L ratio between groups 3 and 4 was found to be significantly higher in group 4 (z=2.17, p=.03). It was not found significant in ROC analysis. In terms of days between controls, the number of days in group 3 was found to be significantly shorter (z=3.00, p=.003) (Table 3).
a: Arithmetic mean (standard deviation), b: Median (Interquartile range).
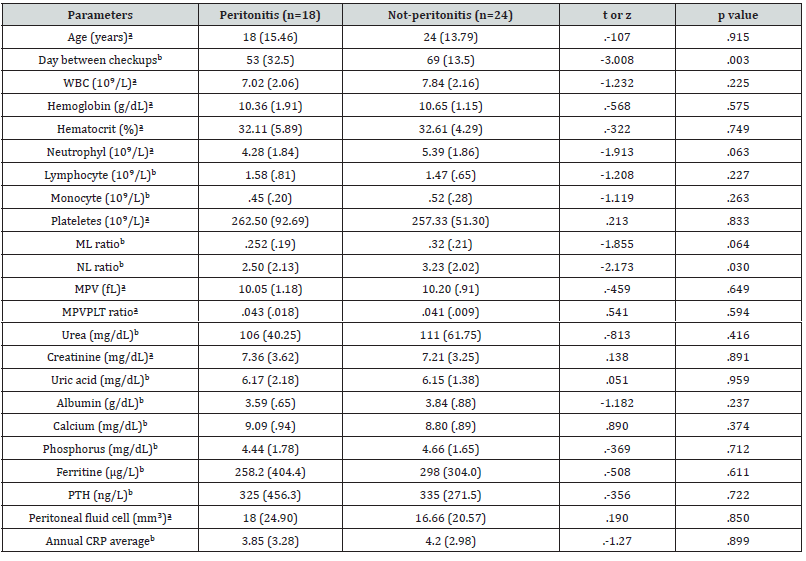
While there was no significant difference between M/L, N/L ratios in comparison of groups 2 and 4, number of monocytes (t= -2.27, p=.03), M/L ratio (t= -3.29, p=.002) in comparison of groups 1 and 3, N/L ratio (t= -3.54, p=.001) was found to be significantly higher in group 1 (Table 4).
a: Arithmetic mean (standard deviation), b: Median (Interquartile range).
With the ROC analysis performed in patients with a cut-off value of 0.36 with an M/L ratio above CRP 5, 75% sensitivity and 70% specificity (NPV77.7, PPV 66) were found to be significant in predicting the risk of peritonitis (Figure 1).
In the group with peritonitis, 13% Gram+, 9% Gram-, 12% growth could not be detected in the distribution of microorganisms. No relationship was found between the microorganism group and the M/L ratio. When 4 groups were formed according to BMI, only uric acid level was found to be significantly higher in the group with a BMI of 30 and above (7.3 vs. others; KW-χ2(3) = 12.060, p = .007). All other parameters were similar between the 4 groups (p>.05 for all) (Table 5).
a: Arithmetic mean (standard deviation), b: Median (Interquartile range), PD: Peritoneal dialysis, PL: peritoneal liquide, PLT: plateles, KW-χ2: Kruskal Wallis test, *: Fisher’s exact test.
BMI groups and microbiological variables (gram+ or gramstaining, and no growth in medium) distributions were found to be similar (Fisher’s exact test=6.659, p=.346). No significant correlation was found between BMI and peritonitis in the total group (t=1,370, p=.175). No correlation was found between BMI and M/L in the CRP≤5 group (r=.052, p=.745). No correlation was found between BMI and M/L in the CRP>5 group (r=.153, p=.372)
Discussion
The prevalence and incidence of chronic kidney disease (CKD) is an increasingly important public health problem worldwide [11]. Peritoneal dialysis is one of the treatments for chronic kidney disease, and peritonitis is the most important cause of morbidity and mortality. Normally, mortality and morbidity are high in chronic kidney disease due to increased inflammation [12,13]. Therefore, early diagnosis and treatment of peritonitis attack is extremely important. Peritonitis developed in 43.6% of the cases in the study. Of those who had peritonitis, 52.9% were female and 47.1% were male, and there was no difference in terms of gender.
In chronic kidney disease, the immune system is suppressed. There is a serious inflammation caused by reasons such as uremic toxins, RAAS, comorbid conditions, volume load, hypertension, iron use, and antioxidant deficiency, and this situation is exacerbated by peritonitis, increasing mortality and morbidity [3,4]. As far as we know, there is no data on the M/L ratio predicting the risk of peritonitis in the literature.
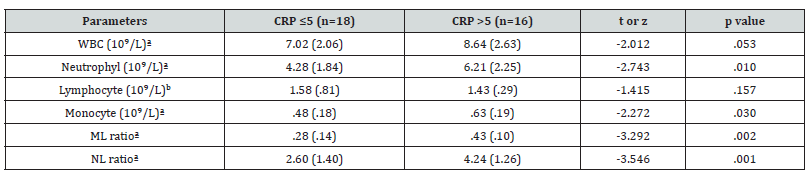
High N/L ratio among hematological parameters has been shown as a predictive and prognostic marker in cardiovascular diseases and various cancers [14-17]. It was expressed as a predictor of worsening renal functions in diabetic patients [18]. M/L, N/L, PLT/L, MPV/PLT (mean platelet volume/platelet) ratios have been shown as biomarkers in critically ill patients [6]. In the study, there was no difference in the N/L ratio between the groups with and without peritonitis, and there was no difference in the ratios of PLT/L, MPV/PLT, while a significant difference was found in the N/L ratio between the groups with and without peritonitis without chronic inflammation.
It has been reported that the M/L ratio is the best predictor of cardiovascular and all-cause mortality in hemodialysis patients, it has an additional value in deaths due to heart failure, and it is a predictor in preoperative evaluations in malignancies and in new-onset CKD [19-22]. No difference in N/L ratio was found. It was found that the M/L ratio could be used as a predictive value in predicting the risk of peritonitis in the subgroup with a CRP level above 5 and chronic inflammation.
The precursors of the immune system, with their soluble factors, defend the host against various factors in a complex way. Monocyte innate is the precursor cell of the immune system and transforms into antigen-presenting dendritic cells [23,24]. It is known that with the suppression of the immune system in chronic kidney disease, the number of lymphocytes decreases, and the number of monocytes decreases. Studies have shown that although the number has decreased, the functions continue to be normal [25,26]. It has been shown that antigen-presenting cells in the peritoneal cell population act quickly against stimuli from other parts of the body, and that peritoneal macrophages stimulate T cells two to three times more strongly than other macrophages in patients undergoing peritoneal dialysis [27,28]. Dendritic cells that develop from monocytes are cells capable of professional and pure T cell activation [29]. Considering these reasons, monocytelymphocyte ratio was thought to be a valuable follow-up method before peritonitis, and CRP level was found to be significantly higher in cases with chronic inflammation above 5.
Studies on inflammatory reactions due to infection in humans are largely based on findings in the blood compartment [29]. In peritoneal dialysis, when leukocytes were compared in infected and uninfected patients, it was observed that proinflammatory and anti-inflammatory mediators were rearranged [30,31]. In the peritoneal cavity, in non-infected individuals, 85% of leukocytes are composed of mononuclear phagocytes, 6% are composed of neutrophils and eosinophils, and flow cytometry has shown that 40% lymphocytes and 2-6% dendritic cells are found. Neutrophils are more sophisticated cells than conventional thinking. receptors were found to function differently according to time and region [32]. While there is an increase in peritoneal macrophages and neutrophils 1-2 days before the infection occurs, neutrophils decrease in 2-3 days during the transition to the healing phase, and macrophages and lymphocytes increase relatively. Resolution reverses the macrophage lymphocyte ratio. Neutrophils remain in the circulation for 4-18 hours, while monocytes remain in the circulation for 1-3 days. This may explain the increase in the N/L ratio before the period when CRP started to rise [33].
CRP, which is an indicator of systemic inflammation in PD patients, was found to be variable between 12% and 65%. There is a high rate of basal inflammation in patients with CKD [34] It is known that systemic and local inflammation affects PD patients. Inflammatory markers are important as predictors of clinical outcomes. For this purpose, the number of studies on the value of hematological rates is increasing day by day.
The presence of three different monocytes in the circulation, their responses to each other and to the uremic environment affect hematological rates. While the monocyte count is normal, there is a tendency to lymphopenia in the uremic environment. This may cause a significant difference in the M/L ratio of CRP above 5 in the case of chronic inflammation.
Bernhard Schmekal [7] reported that the risk of leakage and peritonitis is increased in patients with high BMI compared to those with normal BMI. It was stated by Stefan P McDonald et al. [8] that high BMI occurs in a shorter period of time for the first peritonitis attack and is a risk factor independent of other risk factors. In our study, no significant relationship was found between BMI and increased risk of peritonitis. Dev Jegatheesan et al. [9] pointed out that fungal peritonitis between BMI 30-34.9, grampositive peritonitis in those with BMI ≥35, and an increased risk of gram-negative peritonitis in both groups. Again, in our study, no significant relationship was found between microorganism groups and BMI groups.
Sean Duek Hwang et al. [10] reported that BMI>25.71 is an important risk factor for all-cause mortality. Andresa Marques de Mattos et al. [11] stated that increased adipose tissue is a cause of chronic inflammation in those with CKD. He reported that adipocytes show pro-inflammatory and metabolic activity in peripheral tissues and secrete cytokines such as interleukin 6 (IL- 6). It has been reported that such adipokines secretions are different in subcutaneous and visceral tissue, and IL-6 secretion is two to three times higher in visceral tissue. In line with this information, the expected relationship between BMI and peritonitis risk was not found in our study. The limitations of the study are its retrospective nature, the inability to measure expensive parameters such as traditional TNF α and IL-6, and its regional nature.
Conclusion
Monocyte-lymphocyte ratio was thought to be a valuable follow-up method before peritonitis due to the high risk of infection in patients undergoing peritoneal dialysis, and the CRP level was found to be significantly higher in cases with chronic inflammation over 5mg/dL. We think that this method can be used as a cheap, easy and routinely applicable diagnostic method.
Acknowledgement
I would like to thank my friend, associate professor Zeynep Göker for her support.
Authorship
The subject selection was done by Prof Dr. Murat Duranay, data collection and writing were done by Dr. Semahat Karahisar Şirali, editing and statistics was done by Dr. Refika Büberci. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Informed Consent to Participate
Written informed consent was obtained from all subjects before the study.
References
- Katherine RT, Radica ZA, Kenrik DO, Cami R Jones, Kenn B Daratha, et al. (2019) Clinical Characteristics of and Risk Factors for Chronic Kidney Disease Among Adults and Children: An Analysis of the CURE-CKD Registry. JAMA Netw Open 2(12): e1918169.
- Scott B, Rajnish M (2019) Peritoneal Dialysis Access Associated Infections. Adv Chronic Kidney Dis 26(1): 23-29.
- Yoon JW, Pahl MV, Vaziri ND (2007) Spontaneous leukocyte activation and oxygen-free radical generation in end-stage renal disease. Kidney Int 71(2): 167-172.
- Juan J Carrera, Peter Stenvinkel (2010) Inflammation in end-stage renal disease-What Have We Learned in 10 years? Semin Dial 23(5): 498-509.
- Christiaan LM, Peter S, Friedo WD, Juan JC (2011) Monitoring of inflammation in patients on dialysis: forewarned is forearmed. Nat Rev Nephrol 7(3): 166-176.
- Dragan D, Goran R, Maja S, Ivan Stanojevic, Ivo Udovicic, et all. (2018) Neutrophil-to-lymphocyte ratio,monocyte-to-lymphocyte ratio, platelet-to-lymphocyte ratio and mean platelet volume-to-platelet count ratio as biomarkers in critically III and injured patients: Which ratio to choose to predict outcome and nature of bacteremia? Mediators Inflamm 2018: 3758068.
- Schmekal B (2005) Peritoneal dialysis in patients with high body-mass index. Wien Klin Wochenschr 117(6): 40-45.
- McDonald SP, Collins JF, Rumpsfeld M, Johnson DW (2004) Obesity is a risk factor for peritonitis in the Australian and New Zealand peritoneal dialysis patient populations. Perit Dial Int 24(4): 340-346.
- Jegatheesan D, Johnson DW, Cho Y, Elaine M Pascoe, Darsy Darssan, et al. (2018) The relationship between body mass index and organism-spesific peritonitis. Perit Dial Int 38(3): 206-214.
- Hwang SD, Lee JH, Jhee JH, Song JH, Kim JK, et al. (2019) Impact of body mass index on survival in patients undergoing peritoneal dialysis: Analysis of data from the ınsan memorial end-stage renal disease registry of Korea (1985-2014). Kidney Res Clin Pract 38(2): 239-249.
- Mattos AM, Ovidio PP, Jordao AA, Costa JAC, Chiarello PG (2013) Association of body within peritoneal dialysis. Inflammation 36(3): 689-695.
- Rajiv Saran, Bruce Robinson, Kevin C Abbott, Jennifer Bragg-Gresham, Xiaoying Chen, et al. (2020) US Data System 2019 Annual Report: Epidemiology of Kidney Disease in United States. Am J Kidney Dis 75(1 Suppl 1): A6-A7.
- Kunihiro M, Marije V, Brad C Astor (2010) Chronic Kidney Disease Prognosis Consortium. Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: a collaborative metaanalysis. Lancet 375: 2073-2081.
- Yalcın S, Mahmut IY, Alper S, Mutlu Saglam, Erdinc Cakir, et al. (2013) Neutrophil to lymphocyte ratio independently predicts cardiovascular events in patients with chronic kidney disease. Clin Exp Nephrol 17(4): 532-540.
- Tomoko A, Sawako K, Yoshinari T, Sachiyo Sugiura, Takayuki Katsuno, et al. (2015) Neutrophil/lymphocyte ratio as a predictor of cardiovascular events in incident dialysis patients: a Japanese prospective cohort study. Clin Exp Nephrol 19(4): 718-724.
- Ismail K, Eray E, Aydın Ü, Murat HS, Bülent T, et al. (2013) Role of neutrophil/lymphocyte ratio in prediction of disease progression in patients with stage-4 chronic kidney disease. J Nephrol 26(2): 358-365.
- Yusuf b, Erdem C, Nurhan B, Mustafa K, Fuat E, et al. (2014) Peritonitis incidence was correlated with duration of peritoneal dialysis rather than leptin or neutrophil to lymphocyte(N/L) ratio in peritoneal dialysis patients. Eurasian J Med 46(3):145-150.
- Basem A, Jacques D, Fahad BN, Rabih Nasr, Jennifer Ross, et al. (2012) Neutrophil-to-lymphocyte ratio as a predictor of worsening renal function in diabetic patients (3-year follow-up Study). Ren Fail 34(5): 571-576.
- Fangfang X, Rongyi C, Xuesen C, Bo S, Zhonghua L, et al. (2017) Monocyte/lymphocyte ratioas a beter predictor of cardiovascular and all-cause mortality in hemodialysis patients: A prospective cohort syudy. Hemodial Int 22(1): 82-92.
- Silva N, Bettencourt P, Guimaraes JT (2015) The lymphocyte-to-monocyte ratio: An added value for death prediction in heart failure. Nutrition, Metabolism & Cardiovascular Diseases 25(11): 1033-1040.
- Zhang M, Wang K, Zheng H, Zhao X, Xie S, et al. (2019) Monocyte lymphocyte ratio predicts the new onset of chronic kidney disease: A cohort study. Clin Chim Acta 503: 181-189.
- Ahmed E, Jongchan K, Won SJ, Jee SP, Ji EH, et al. (2018) Predictive value of preoperative monocyte-lymphocyte ratio among patients with localized clear renal cell carsinoma of ≤7 cm on preoperative imaging. Medicine (Baltimore) 97(48): e13433.
- Goldszmid RS, Caspar P, Rivollier A, Sandy White, Amiran Dzutsev, et al. (2012) NK cell-derived interferon-ɣ orchestrates cellular dynamics and the differentiation of monocyte into dendiritic cells at the site of infection. Immunity 36(6): 1047-1059.
- Klinger J, Enriguez J, Arturo JA, Delgado M, Avila G, et al. (2002) Comparison of immunophenotypes in the blood of patients on contnuous ambulatory peritoneal dialysis. Adv Perit Dial 18: 165-169.
- Rajiv Agarwal, Robert P Light (2011) Patterns and prognostic value of total and differential leukocyte count in chronic kidney disease. Clin J Am Soc Nephrol 6(6): 1393-1399.
- Mehdi S, Kousar S, Mohammad M, Mousa M, Roghayeh A (2019) Immune Response as a Mechanism of the Initiation and Progression of Chronic Kidney Disease. Iran J Kidney Dis 13(5): 283-299.
- Kubica U, Olszewski WL, Tarnowski W, Bielecki K, Zıolkowska A, et al. (1996) Normal human ımmune peritoneal cells: Subpopulations and functional characteristics. Scand J Immunol 44(2): 157-163.
- Betjes MGH, Tuk CW, Struıjk G, Kredıet RT, Arısz L, et al. (1993) Antigen-presenting capacity of macrophages and dendiritic cells in the peritoneal cavity of patients treated with peritoneal dialysis. Clin Exp Immunol; 94(2): 377-384.
- Faivre V, Lukaszewicz AC, Alves A, Charron D, Payen D, et al. (2007) Accelerated in vitro differentiation of blood monocytes into dendiritic cells in human sepsis. Clin Exp Immunol 147(3): 426-439.
- Liakopoulos V, Roumeliotis S, Gorny X, Theodoros Eleftheriadis, Peter R Mertens, et al. (2017) Oxidative stress in patients undergoing peritoneal dialysis: A current review of the literatü Oxid Med Cell Longev 2017: 3494867.
- Willem M, Fieren JA (2012) The local inflammatory responses to infection of the peritoneal cavity in humans: their regulation by cytokines, macrophages, and other leukocytes. Mediators Inflamm 2012: 976241.
- Li PK, Ng JK, Mcintyre CW (2017) Inflammation and peritoneal dialysis. Semin Nephrol. 37(1): 54-65.
- Cho Y, Hawley CM, Johnson DW (2014) Clinical causes of inflammaton in peritoneal dialysis patients. Int J Nephrol 2014: 909373.
- Girndt M, Trajanowicz B, Ulrich C (2020) Monocytes in uremia. Toxins (Basel) 12(5): 340.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.