Case Report 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Massive Cerebral Air Emboli After VA-ECMO Cannulization: A Case Report
*Corresponding author: Dylan Ryan, Department of Neurology Duke University School of Medicine, USA
Received: June 20, 2022; Published: June 29, 2022
DOI: 10.34297/AJBSR.2022.16.002262
Abstract
Extracorporeal membrane oxygenation (ECMO) can be a lifesaving and disability preventing intervention in patients who have suffered cardiopulmonary compromise. Though this important piece of medical engineering has provided lifesaving benefits to patients, complications of the therapy can lead to fatal consequences. One of these important consequences for providers to be aware of is massive air embolus, which can lead to cerebral ischemia and death. Here we present a 54-year-old male who presented with transient left sided weakness in the setting of suspected endocarditis who then suffered cardiac arrest, and then massive cerebral air emboli in the setting of VA-ECMO deployment.
Keywords: Massive Air Emboli, Air Emboli, ECMO, Stroke
Introduction
Extracorporeal membrane oxygenation (ECMO) has been more commonly used as a treatment option for patients who have suffered cardiopulmonary arrest, patients awaiting cardiac and/ or pulmonary transplant, and in management of patients with cardiogenic shock. There are two distinct circuit configurations: veno-venous extracorporeal membrane oxygenation (VVECMO), in which blood is drawn into the bypass circuit via a vein, oxygenated, and returned to the venous system [1], and venoarterial extracorporeal membrane oxygenation (VA-ECMO), in which blood is drawn into the bypass circuit via a vein, oxygenated, and returned to the arterial system [2]. In this manner, VA-ECMO can provide both cardiac and respiratory support to patients with severe cardiac dysfunction, such as those post cardiac arrest.
Though ECMO has become an essential lifesaving approach, there are a number of potential complications that come with deployment on patients, such as pump failure, acute limb ischemia [3], left ventricular overload [4], severe hemorrhage, acute thromboembolic disease [5], and massive air embolism. Air within the ECMO circuit can be secondary to catheter leak and/or failure, cannula disconnection, migrating cannula, and unsealed intravascular access lines. Due to this complication, during ECMO use there must be close safety monitoring to ensure patient safety. These sources can lead to micro embolic disease or massive gas emboli that can have devastating consequences for patients. Here, we present a case of massive cerebral air embolism in the setting of cardiac arrest after deployment of VA-ECMO to bring further awareness to this complication of this life saving therapy.
Case Presentation
A 54-year-old male with a past medical history of atrial fibrillation status post ablation on therapeutic anticoagulation with rivaroxaban, recent mitral and tricuspid valve replacements, and heart failure with reduced ejection fraction presented to the emergency department from home with lightheadedness and left sided weakness that had resolved after four minutes. A stroke code was called secondary to report of left sided weakness. His initial neurologic examination was without evidence of focal abnormality with a National Institutes of Health Stroke Scale (NIHSS) of 0. Initial non contrast CT of the brain was without abnormality and CT angiogram of the head and neck was without significant stenotic disease or large vessel occlusion. Brain MRI was obtained and showed acute and subacute, multifocal ischemic infarcts concerning for a central embolic source (Figure 1 A-D). The patient was noted to be febrile to 101.7 F on initial vital signs, fostering concern for endocarditis considering recent valve replacements.
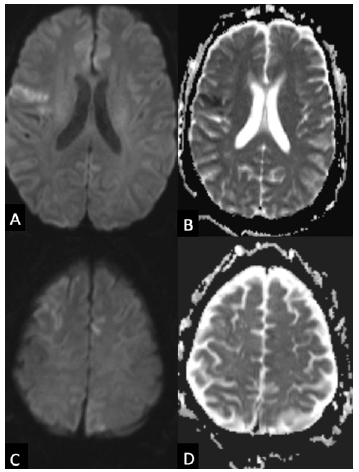
Figure 1:Initial MRI showing multifocal infarcts Axial diffusion weighted images (A, C) show cortical hyperintensities in multiple vascular territories with the apparent diffusion coefficient (B, D) showing decreased values and pseudo normalization suggesting acute and subacute multifocal infarcts.
While in the emergency department, the patient was noted to have an episode of left leg twitching with retained awareness that lasted approximately five minutes. Shortly thereafter, the patient become unresponsive with generalized convulsive activity concerning for generalized tonic clonic seizure. The patient received 2mg of lorazepam and was loaded with 4.5g of intravenous levetiracetam. He was started on meningitis coverage with vancomycin, cefepime, ampicillin and acyclovir. The patient was intubated for airway protection given low Glasgow Coma Scale (GCS).
After intubation the patient was noted to have cardiac arrest in the setting of ventricular fibrillation. During prolonged code, VA-ECMO was deployed, the patient was cannulated and transferred to the intensive care unit. He was started on therapeutic anticoagulation with heparin in the setting of VA-ECMO. Emergent transesophageal echocardiogram (TEE) showed an ejection fraction of approximately 20% with severe left ventricular dysfunction and an echogenic, mobile mass on the mitral valve ring concerning for vegetation versus acute thrombus.
Once therapeutic on heparin, a repeat CT brain was obtained and was notable for multifocal cerebral air emboli with pneumocephalus and surrounding cytotoxic edema (Figure 2 A-B). No clear source for gaseous emboli was detected after evaluation of the ECMO circuit. Repeat CT brain seven hours later revealed markedly worse, diffuse cerebral air emboli with bilateral uncal herniation with diffuse cerebral edema and small right occipital hemorrhage (Figure 2 C-F). The patient was given a bolus of hypertonic saline secondary to diffuse cerebral edema. Previously obtained blood cultures started to grow viridians group streptococcus.
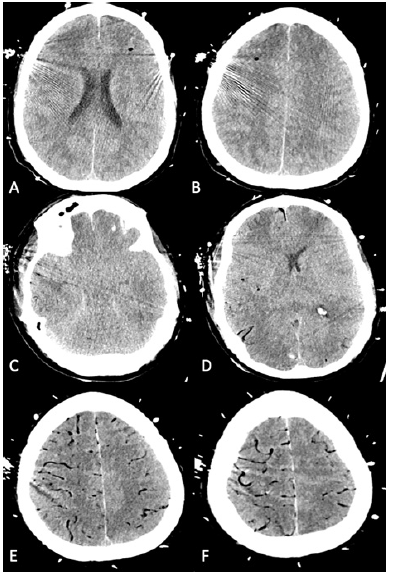
Figure 2:Head CT after ECMO cannulization shows cerebral air emboli Initial axial CT imaging (A, B) shows multiple foci of air emboli with surrounding cytotoxic edema. Repeat axial CT imaging seven hours later shows bilateral uncal herniation (C), right occipital lobe hemorrhage (D) and diffuse vascular air emboli (C, D, E, F)
The patient’s follow up examination was notable for nonreactive pupils, absent corneal reflexes, absent cough, absent gag reflex, and no movement to noxious stimuli. After goals of care discussions with family, formal brain death examination was pursued, and the patient was declared deceased.
Discussion
We have described a case of a 54-year-old male who presented secondary to transient left sided weakness who subsequently suffered cardiac arrest in the setting of ventricular fibrillation placed on VA-ECMO complicated by massive air emboli. In this case there was detection of cerebral air emboli, which led to diffuse cerebral edema and the unfortunate progression to brain death in this patient. This is an unfortunate, devastating complication of ECMO utilization.
It has been shown that ECMO can improve survival without severe disability in patients with potentially reversible respiratory failure [6], and thus can have substantial impact on critically ill patients with cardiopulmonary failure. When ECMO is utilized though, there are important safety considerations to follow due to the possible complications of the therapeutic intervention itself. The intervention itself requires an inflow venous cannula connected to a pump which brings venous blood to a bubble oxygenator [1,2,7]. Associated with the oxygenator is a flow meter which controls oxygen gas flow, a blender that provides a partial pressure of attached oxygen to the oxygenator, and a heat exchanger to regulate the temperature of the blood returning to the patient. The blood then must return via an outflow cannula to the patient’s venous or arterial system [1,2,8]. Currently required in this process is therapeutic anticoagulation, typically with unfractionated heparin, as blood passes through multiple coated cannulas and membranes to reduce risk of thromboembolic disease [2,8]. It is also important to measure the pre-oxygenator and postoxygenator pressures routinely and consistently, as increases in pressure changes suggest increased oxygenator resistance which is often secondary to thrombotic disease [7]. Considering this circuitry, patients are at risk of both extravasation in the setting of therapeutic anticoagulation as well as thromboembolic disease.
Within this framework itself though is an increased risk of massive air emboli. Air embolism within the circuit has been reported as high as 4.9% [9] and can lead to fatal complications for patients. There are multiple possible sources of air emboli in the ECMO circuitry via broken cannulas, migrating cannulas, unsealed vascular access lines, oxygenator tear, and cavitation [10]. Any time gas is detected in the ECMO circuitry, the position of the cannula must be checked, as well as ensuring intravascular lines are properly sealed, as well as the general integrity of the circuitry itself [11]. In the case of cavitation, gas is pulled out of solution in the setting of negative pressure within the venous system, which can lead to gaseous emboli within the ECMO circuitry. Kinking and obstruction via clamping of the venous cannula can lead to this complication, which must be a consideration when air is detected within the circuitry.
Gas within the venous system in VV-ECMO can lead to airlock which will lead to lack of adequate blood flow, which forces the circuit to be disconnected, which can create catastrophic consequences for patients requiring cardiopulmonary bypass [10]. Gas in the arterial system in VA-ECMO can lead to systemic air embolization that can lead to severe hypotension and cerebral ischemia [10]. In our case, there was diffuse cerebral involvement that led to diffuse cytotoxic cerebral edema that led to bilateral uncal herniation and unfortunate demise.
Therapeutic options in the setting of cerebral air emboli involve both supportive interventions as well as selective definitive therapies, though these definitive therapies are challenged in these critically ill patients. When air is detected in the VA-ECMO circuitry, the arterial inflow cannula should be clamped to prevent air entry into the patient’s systemic circulation [10] and flow should be stopped through the circuitry. The patient should be placed in reverse Trendelenberg [10] or left lateral decubitus with head down position [12] if air is thought to be in the venous system as a means to reduce risk of cerebral embolization but should remain supine if in the arterial systemic circulation as head down positions can worsen cerebral edema in the setting of cerebral embolization [12]. Systemic hypothermia has been attempted as well as the volume and pressure of a gas decreases with decreases in temperature [13], though there is little evidence for this measure in the acute setting of gaseous emboli.
Definitive therapies for systemic air embolism include hyperbaric oxygen therapy and hyperoxia [11] and manual air removal via central venous catheterization to the right heart [14]. Hyperbaric oxygen therapy provides partial pressures of oxygen higher than that of atmospheric pressure, which leads to denitrogenation of air bubbles and thus reduces bubble size, which in turn reduces the degree of mechanical obstruction of the bubbles themselves. Hyperbaric oxygen therapy is not widely available in hospitals though, and the challenges of providing these therapies to patients on ECMO are often too challenging at this current time. There have been reports of direct removal of air from the right heart via central venous catheters, but the volume is often quite low [14]. Due to these factors, unless there is already a central venous line, this intervention is not pursued.
In conclusion, the lifesaving and disability preventing benefits of ECMO therapy provide an important tool for providers in caring for patients with severe cardiopulmonary compromise, but complications such as massive cerebral air emboli that carry fatal consequences and limited interventions point to important monitoring considerations for these patients. The risk of these complications must be in the mind of providers caring for these patients, and thus consistent and routine safety assessments, patient simulations, and continued education regarding these complications must be performed to limit these complications in the future.
Acknowledgment
None
Conflict of Interest
No conflict of interest.
References
- Carlo Banfi, Matteo Pozzi, Nils Siegenthaler, Marie Eve Brunner, Didier Tassaux, et al. (2016) Veno-venous extracorporeal membrane oxygenation: cannulation techniques. J Thorac Dis 8(12): 3762-3773.
- Chung M, Shiloh AL, Carlese A (2014) Monitoring of the adult patient on venoarterial extracorporeal membrane oxygenation. ScientificWorldJournal 2014: 393258.
- Theodosios Bisdas, Gernot Beutel, Gregor Warnecke, Marius M Hoeper, Christian Kuehn, et al. (2011) Vascular complications in patients undergoing femoral cannulation for extracorporeal membrane oxygenation support. Ann Thorac Surg 92(2): 626-631.
- Pengyu Ma, Zaiwang Zhang, Tieying Song, Yunliang Yang, Ge Meng, et al. (2014) Combining ECMO with IABP for the treatment of critically Ill adult heart failure patients. Heart Lung Circ 23(4): 363-368.
- Christian Dornia, Alois Philipp, Stefan Bauer, Matthias Lubnow, Thomas Müller, et al. (2014) Analysis of thrombotic deposits in extracorporeal membrane oxygenators by multidetector computed tomography. ASAIO J 60(6): 652-656.
- Giles J Peek, Miranda Mugford, Ravindranath Tiruvoipati, Andrew Wilson, Elizabeth Allen, et al. (2009) Efficacy and economic assessment of conventional ventilatory support versus extracorporeal membrane oxygenation for severe adult respiratory failure (CESAR): a multicentre randomised controlled trial. Lancet 374(9698): 1351-1363.
- Srivats Sarathy, Joseph W Turek, Jian Chu, Aditya Badheka, Marco A Nino, et al. (2021) Flow Monitoring of ECMO Circuit for Detecting Oxygenator Obstructions. Ann Biomed Eng 49(12): 3636-3646.
- Lequier L, Horton SB, McMullan DM, Bartlett RH (2013) Extracorporeal membrane oxygenation circuitry. Pediatr Crit Care Med 14(5 Suppl 1): S7-12.
- ELSO (2017) ECLS Registry Report 1-34.
- Allen S, Holena D, McCunn M, Kohl B, Sarani B (2011) A review of the fundamental principles and evidence base in the use of extracorporeal membrane oxygenation (ECMO) in critically ill adult patients. J Intensive Care Med 26(1): 13-26.
- Kumar A, Keshavamurthy S, Abraham JG, Toyoda Y (2019) Massive Air Embolism Caused by a Central Venous Catheter During Extracorporeal Membrane Oxygenation. J Extra Corpor Technol 51(1): 9-11.
- Jorens PG, Van Marck E, Snoeckx A, Parizel PM (2009) Nonthrombotic pulmonary embolism. Eur Respir J 34(2): 452-74.
- Tovar EA, Del Campo C, Borsari A, Webb RP, Dell JR, et al. (1995) Postoperative management of cerebral air embolism: gas physiology for surgeons. Ann Thorac Surg 60(4): 1138-1142.
- Muth CM, Shank ES (2000) Gas embolism. N Engl J Med 342(7): 476-482.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.