Research Article 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Plasma Polyunsaturated Fatty Acid Levels in Patients with Chronic Kidney Disease on Dialysis
*Corresponding author:Pham Van Tran, Department of Biochemistry, 103 Military Medical Hospital, Ha Noi, Viet Nam.
Received:November 30, 2022; Published:December 19, 2022
DOI: 10.34297/AJBSR.2022.17.002388
Abstract
Aim: Determination of plasma concentrations of some polyunsaturated fatty acids and plasma omega 3/omega 6 ratios in patients with chronic kidney disease (CKD) on dialysis. Methods: The study was conducted on 188 people, from 18 to 70 years old, including 64 CKD patients on dialysis (study group), 60 CKD patients with conservative treatment (disease control group) and 64 normal healthy people (healthy control group). The concentration of unsaturated fatty acids was measured by gas chromatography-mass spectrometry. Total cholesterol, triglycerides, HDL-C, LDL-C, Total protein, albumin, CRP, urea, glucose, creatinine in plasma were measured by photometric methods. Results: The plasma saturated fatty acids of the three groups were roughly equivalent. The omega 3 Arachidonic acid (AA), Eicosapentaenoic acid (EPA), Docosahexaenoic acid (DHA) decreased in the study group compared with the other groups (p<0.05), EPA levels and DHA of the disease control group was lower than that of the healthy control group (p<0.05), there was no difference in ALA concentration between these two groups (p>0.05). Omega 6 such as Linoleic acid (LA), Arachidonic acid (AA) decreased, the lowest in the group 1, the highest in the group 3 (p<0.05). The ratio of omega 3/omega 6 (ω3/ω6) and total polyunsaturated fatty acids/ω6 decreased in groups 1 and 2 compared with group 3 (p<0.05). The group of patients with dialysis time less than 60 months had higher plasma omega concentration than the group with dialysis time over 60 months (p<0.05). Conclusion: The concentration of ω3 and ω6 and the ratio of ω3/ω6 in CKD patients on dialysis decreased compared with the group of CKD patients on conservative treatment and the healthy control group. The longer the dialysis time, the lower the concentration of unsaturated fatty acids. This confirms that the supplementation of unsaturated fatty acids for patients with chronic kidney disease on dialysis is necessary.
Introduction
Chronic kidney disease (CKD) has become a public health problem in many countries. Worldwide, more than 500 million people are suffering from CKD, of which the majority (80%) come from developing countries [1]. In Vietnam, it is estimated that about 10,000 patients receive CKD. Patients with CKD on long-term dialysis often experience many complications related to anemia, bone dystrophy, cardiovascular disease, and malnutrition.
The 20-carbon unsaturated fatty acids are essential fatty acids that the body cannot synthesize on its own, a deficiency will lead to imbalances, autoimmune disorders, inflammatory conditions. These polyunsaturated fatty acids are important precursors for the synthesis of prostaglandins, which include: 8, 11, 14-eicosatrienoic (dihomo-ϒ-linolenic-DGLA) or 20:3n-6, 5, 8, 11, 14-eicosatetraenoic (arachidonic acid-AA) or 20:4n-6 and 5, 8, 11, 14, 17-eicosapentaenoic (EPA) or 20:5n-3.
Eicosapentaenoic acid (EPA; also, Eicosapentaenoic acid) is an omega-3 fatty acid. In physiological literature, it is given the name 20:5(n-3). It also has the trivial name timnodonic acid. In chemical structure, EPA is a carboxylic acid with a 20-carbon chain and five cis double bonds; the first double bond is located at the third carbon from the omega end. EPA acts as a precursor for prostaglandin- 3 (which inhibits platelet aggregation), thromboxane-3, and leukotriene-5 eicosanoids. EPA is both a precursor and the hydrolytic breakdown product of eicosapentaenoic ethanol amide. A prescription drug containing only EPA was shown to reduce heart attack, stroke, and cardiovascular death by 25% relative to a placebo in those with statin-resistant hypertriglyceridemia [2].
Arachidonic acid is a polyunsaturated fatty acid present in the phospholipids (especially phosphatidylethanolamine, phosphatidylcholine, and phosphatidylinositides) of membranes of the body’s cells, and is abundant in the brain, muscles, and liver. Skeletal muscle is an especially active site of arachidonic acid retention, accounting for roughly 10-20% of the phospholipid fatty acid content typically [3].
In addition to being involved in cellular signaling as a lipid second messenger involved in the regulation of signaling enzymes, such as PLC-ϒ, PLC-δ, and PKC-α, -β, and -ϒ isoforms, arachidonic acid is a key inflammatory intermediate and can also act as a vasodilator [4].
While most studies on PUFA and CKD have focused on n-3 fatty acids [5], numerous large human studies have shown that dietary or tissue levels of n-6 PUFA are inversely associated with inflammation [6] and with clinical events such as blood pressure, diabetes, and cardiovascular diseases (CVD). Low plasma PUFA levels are associated with decreased renal function in the elderly [39]. In addition, people with lower plasma PUFA levels have a higher risk of kidney failure [64].
Plasma concentrations of saturated fatty acids in CKD patients decreased compared with healthy subjects. [2]. Observational and interventional studies have shown an association of omega 3/omega 6 concentration and ratio with dialysis time [4,7]. Diets rich in omega 3 reduce blood LDL-C levels, but can also reduce HDL-C, significantly improve cardiovascular risk factors, prolong survival for CKD patients [8,9]. We studied this topic with the goal of determining the concentration of some polyunsaturated fatty acids, the ratio of plasma omega 3/omega 6 in patients with chronic kidney disease on dialysis. From there, it can be recommended to add these substances to the diet for patients with CKD on dialysis.
Methods
Selection of Research Subjects
The study was conducted on 188 people, from 18 to 70 years old, including 64 CKD patients on dialysis ≥ 3 months (study group, group 1), 60 CKD patients with conservative treatment (diseased control group, group 2) and 64 normal healthy people (healthy control group, group 3). The study period was from November 2019 to July 2020 at Military Hospital 103.
Blood samples were collected in the morning after a 12-h fast. Fatty acids were measured using a fasting plasma sample. We collected data according to a research medical record including medical inquiries, clinical examination, and laboratory tests. The plasma biochemical tests and fatty acids were performed at the Department of Biochemistry of Military Hospital 103 and Department of Biochemistry, Institute of 69.
Measurement of Plasma Unsaturated Fatty Acids
Plasma fatty acid composition was analysed by gas–liquid chromatography Thermos Scientific Trace ITQ 900™ GC/MS (Thermofisher, Hanoi, Vietnam) at the 69 Institute, Hanoi, Vietnam. After separation of lipids by thin layer chromatography and trans-methylation, all fatty acids ranging from myristic acid (14:0) to DHA (22:6) were recorded. This study analyses the relative proportions of essential PUFA [LA (n-6), ALA (n-3) and LC n-3 (representing the sum of EPA, DPA and DHA)] in the plasma of dialysis patients.
Measurement of Plasma Biochemical Parameters
Total cholesterol, triglycerides, HDL-C, LDL-C, total protein, albumin, urea, glucose, creatinine in plasma were measured by enzymatic photometric method on the AU5800 using the kit of the same manufacturer (Beckman Coulter, Ha Noi, Vietnam).
Statistical Analyses
The values were expressed as mean±SD or median (10th and 90th percentiles) or percentage of total. Differences of PUFA studied between difference groups were assessed after correction for age and sex using a mixed model procedure. All statistical analyses were performed using statistical software SPSS version 20. Statistical significance was set at P<0.05. Because P-values were not adjusted for multiple testing, they must be considered descriptive.
Results
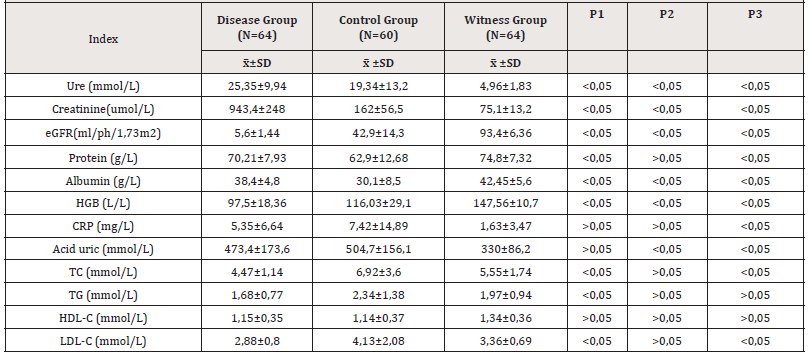
The study was carried out on 188 people, in which the CKD patient on dialysis group had 36 men (56.3%) and 28 women (43.7%); the diseased control group had 42 men (70%) and 18 women (30%), and the healthy control group had 52 men (81.3%) and 12 women (18.7%). The mean age of these three groups is 37.22±9.6, 43.83±18.7, 36.28±18.3 years, respectively (Table 1&2).
Note*: GFR: (Glomerular filtration rate); HGB: Hemoglobine; CRP: C reactive protein; GOT: Glutamate Oxaloacetate Transferase; GPT: Glutamate Pyruvate Transferase; TC: Total Cholesterol; TG: Triglyceride; HDL: Hight Dennity Lipoproteine; LDL: Low Density Lipoproteine; Results are presented as mean and SD; p1: (comparison between study group and diseased control group), p2: (comparison between study group and healthy control group), p3: (comparison between diseased control group and healthy control group).
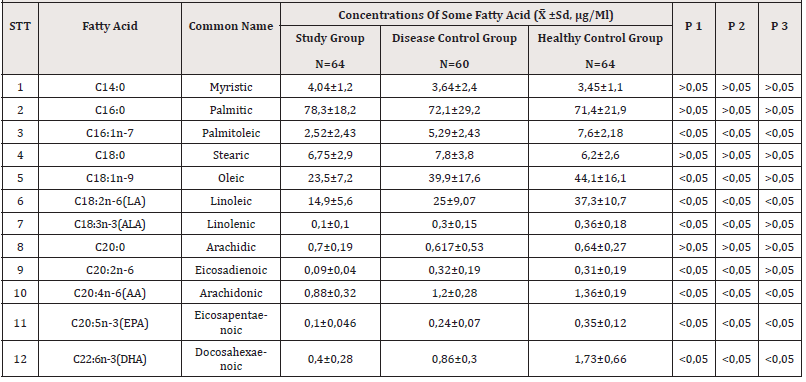
The plasma saturated fatty acids of the three groups were roughly equivalent. C16:1n-7 and C18:1n-9 decreased in the study group compared with the disease control group and the healthy control group (p<0.05). Omega 3 such as ALA, EPA, DHA decreased in the study group compared with the other groups (p<0.05), the concentration of EPA and DHA in the disease control group was lower than in the healthy control group (p<0 ,05), there was no difference in ALA concentration between these two groups (p>0.05). The omega 6 (LA and AA) decreased, the lowest in the study group, the highest in the healthy control group (p<0.05) (Table 3).
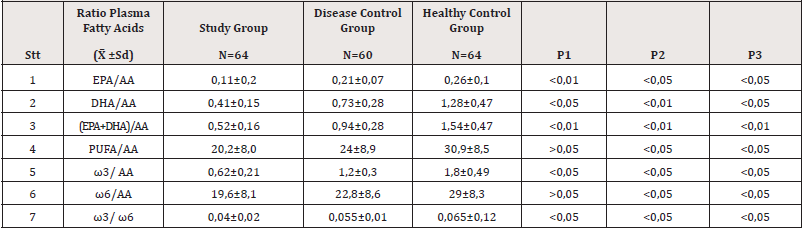
Compared with the healthy control group, the ratio of omega 3/ omega 6 and total polyunsaturated fatty acids/omega 6 decreased in the other groups (Table 4).
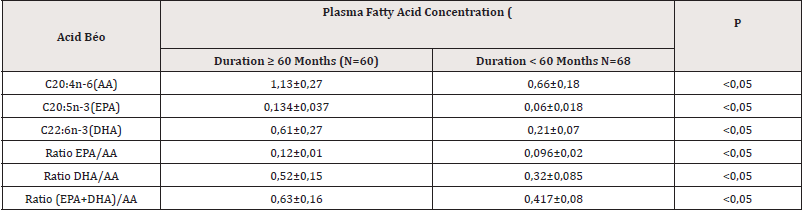
Table 4:The relationship between the concentration of some polyunsaturated fatty acids and the time of dialysis.
The group of CKD patients with dialysis time less than 60 months had higher plasma omega concentration than the group of patients with dialysis time over 60 months (p<0.05).
Discussion
We investigated plasma unsaturated fatty acid concentrations in patients with chronic kidney disease on dialysis. In this study, CKD patients on dialysis had low albumin, total protein and HGB levels reflecting poor nutritional status. Increased plasma uric acid reflects renal dysfunction in uric acid excretion as renal function declines. Elevated plasma CRP levels have been observed in approximately 20% to 65% of patients with end-stage CKD indicating inflammation in this subject.
The rate of dyslipidemia of patients with chronic kidney disease on dialysis in our study was high, the increase in TG accounted for the highest rate, then the increase in LDL-C and increase in TC, HDL-C accounted for a lower percentage. Controlling dyslipidemia to reduce the risk of cardiovascular disease is an important goal for this population [2,5].
Our study showed that the differences in the concentrations of saturated and unsaturated fatty acids between the study groups followed two trends Table 3. The concentration of saturated fatty acids in the disease group was higher than in the other two groups, but this difference was not statistically significant (p>0.05), which is also consistent with some studies around the world [10]. Our results are like those of other authors confirming that the changes of saturated fatty acid levels in the plasma of patients with CKD are not affected by dialysis [11,12].
In contrast, the concentration of unsaturated fatty acids in CKD patients with or without dialysis decreased compared with healthy people Table 2, especially omega 3 and omega 6. This is consistent with published worldwide reports [2,11]. The general decrease in unsaturated fatty acids compared with the control group in parallel with the decrease in triglycerides may be due to malnutrition, inflammation, and atherosclerosis. [6]. Low plasma EPA and DHA may be due to poor intake of foods containing EPA and DHA (fish and meat) or may be due to reduced absorption of fatty acids such as omega 3 associated with dietary potassium restriction. On the other hand, the traditional Vietnamese diet contains very little omega 3 and omega 6 [11].
Thus, the low plasma levels of polyunsaturated fatty acids in CKD patients on dialysis may be a factor in the association with inflammation and cardiovascular complications that often occur in these patients [11,7]. Table 3 results show a decrease in the ratio of omega 3/omega 6 of the study group compared with the other groups (p<0.05). The difference in omega 3/omega 6 ratio between the disease group and the control group is consistent with the study of other authors. These studies indicate that the optimal ratio may vary with the disease under consideration. This is consistent with the fact that chronic diseases are multigenic and multifactorial [2,13].
ALA is a precursor of the family of omega 3 fatty acids, ALA is converted to EPA and DHA through long-lasting and desaturated reactions. Linoleic acid (LA) is a precursor to the family of omega 6 fatty acids. In the body, LA is converted to arachidonic acid (AA). AA and EPA will compete for the same metabolic enzymes. Under the action of cyclo-oxygenase and lipo-oxygenase, EPA will produce prostaglandins and thromboxane group 3 (PGE3, PGI3, TXA3) and group 5 leukotrienes (CTB5, LTC5 - CTE5) which have anti-inflammatory effects and inhibit the aggregation platelet collection. Progressive, persistent inflammation in CKD patients with CRP was consistently higher than in controls Table 1. Thereby reducing polyunsaturated fatty acids in plasma, especially omega 3, leading to a decrease in the ratio of omega 3/omega 6. The ratio of EPA/AA, DHA/AA showed a close association. more closely associated with cardiovascular complications than EPA, DHA, or AA concentrations in several studies [14,15].
The right omega-3/omega-6 ratio will bring many health benefits and prevent disease, some clinical studies have shown that reducing the omega-6/omega-3 ratio helps reduce diseases. chronic. One study found that replacing corn oil with olive and canola oils to achieve an omega-3/omega-6 ratio of 1/4 resulted in a 70% reduction in total mortality [14]. Thus, the reduction in the ratio of omega 3 to omega 6 in the study group compared to the control group is consistent with published studies [14,15]. There was a statistically significant difference in unsaturated fatty acid levels between the dialysis group above and below 60 months Table 4. The group of patients with prolonged dialysis time had lower concentrations of omegas and the ratio of omega 3 to omega 6 compared to the group with less time on dialysis. This result is consistent with many studies around the world [3,4,7].
Szczuko, et al., (2020) showed decreased PUFA levels in patients with chronic kidney disease. There is also an inverse correlation between K+ secretion and EPA and DHA concentrations [7]. As chronic kidney disease progresses, the elimination of K+ increases, the activity of 6-desaturase decreases, the synthesis of EPA and DHA decreases, the ratio C18:3n-6/C22:4n-6 decreases sharply after 5 years. This index can be used to diagnose disease progression [7].
In addition, several factors may contribute to this altered unsaturated fatty acid reduction: loss of appetite and/or restriction of dietary intake, loss of omega 3 on dialysis [13]. The decrease in omega 3 and omega 6 in the study group was inversely correlated with the duration of dialysis. This is the basis for adding these substances to the diet of CKD patients on dialysis [16-20].
Conclusion
The concentration of omega 3 and omega 6 and the ratio of omega 3/omega 6 in the group of CKD patients on dialysis decreased significantly compared with the group of chronic kidney disease patients on conservative treatment and the healthy control group. The longer the dialysis time, the lower the concentration of unsaturated fatty acids. This shows the need for unsaturated fatty acid supplementation in patients with CKD on dialysis.
Author Contribution
Hoang Thi Minh, Nguyen Van Ky processed the research data, performed the analysis. Dam Thi Phuong Lan, Vu Quang Hop, Pham Thai Binh drafted the manuscript and designed the calculations, Tong Quoc Dong, Nguyen Thi Mai Ly, Pham Van Tran research design, aided in interpreting the results and worked on the manuscript. All authors discussed the results and commented on the manuscript.
Ethical Approval
The study was approved by the Ethical Committee of the Military Hospital 103, Vietnam.
Statement of Human Rights
The study was performed following the ethical standards of the responsible committee on human experimentation (institutional and national).
Statement of Informed Consent
Internet informed consent was obtained from the participants at the end of the e-questionnaire for their anonymized information to be published in this article..
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research and/or authorship of this article: This work was supported by Military Hospital 103, Vietnam.
ORCID ID: Pham Van Tran https://orcid.org/0000-0002-4421- 7813.
References
- Mills KT, Yu Xu, Weidong Zhang, Joshua D Bundy, Chung-Shiuan Chen et al. (2015) A systematic analysis of worldwide population-based data on the global burden of chronic kidney disease in 2010. Kidney Int 88(5): 950-957.
- Nakamura N, Takeshi Fujita, Ryuichiro Kumasaka, Reiichi Murakami, Michiko Shimada, et al. (2008) Serum lipid profile and plasma fatty acid composition in hemodialysis patients-comparison with chronic kidney disease patients. in vivo 22(5): 609-611.
- Sikorska Wiśniewska M, Adriana Mika, Tomasz Śledziński, Sylwia Małgorzewicz, Piotr Stepnowskiet al. (2017) Disorders of serum omega-3 fatty acid composition in dialyzed patients, and their associations with fat mass. Renal failure 39(1): 406-412.
- Z Rasic Milutinovic, G Perunicic, S Pljesa, Z Gluvic, S Sobajic, I Djuric, D Ristic, et al. (2007) Effects of N-3 PUFAs supplementation on insulin resistance and inflammatory biomarkers in hemodialysis patients. Renal failure 29(3): 321-329.
- Shen H, Yan Xu, Jingfen Lu, Chunfang Ma, Yadong Zhou, et al. (2016) Small dense low-density lipoprotein cholesterol was associated with future cardiovascular events in chronic kidney disease patients. BMC nephrology x. 17(1): 143.
- Pecoits Filho R, B Lindholm and P Stenvinkel (2002) The malnutrition, inflammation, and atherosclerosis (MIA) syndrome–the heart of the matter. Nephrology Dialysis Transplantation 17(suppl_11): 28-31.
- Szczuko M, Małgorzata Kaczkan, Sylwia Małgorzewicz, Przemysław Rutkowski, Alicja Dębska Ślizień et al. (2020) The C18: 3n6/C22: 4n6 ratio is a good lipid marker of chronic kidney disease (CKD) progression. Lipids in Health and Disease 19: 1-14.
- Baum SJ, Penny M Kris Etherton, Walter C Willett, Alice H Lichtenstein, Lawrence L Rudel, et al. (2012) Fatty acids in cardiovascular health and disease: a comprehensive update. Journal of clinical lipidology 6(3): 216-234.
- Czernichow S, D Thomas, and E Bruckert (2010) n-6 Fatty acids and cardiovascular health: a review of the evidence for dietary intake recommendations. British journal of nutrition 104(6): 788-796.
- Shearer GC, Juan J Carrero, Olof Heimbürger, Peter Barany, Peter Stenvinkel (2012) Plasma fatty acids in chronic kidney disease: nervonic acid predicts mortality. Journal of Renal Nutrition 22(2): 277-283.
- Benjamin Gollasch, Inci Dogan, Michael Rothe , Maik Gollasch, Friedrich C Luft (2020) Effects of hemodialysis on blood fatty acids. Physiological Reports 8(2): 14332.
- Ross IA, Thomas Boyle, Widmark D Johnson, Robert L Sprando, Michael W O'Donnell, et al. (2010) Free fatty acids profile of the fetal brain and the plasma, liver, brain and kidneys of pregnant rats treated with sodium arsenite at mid-organogenesis. Toxicology and industrial health 26(10): 657-666.
- Friedman AN, Sharon M Moe, Susan M Perkins, Yong Li, Bruce A Watkins (2006) Fish consumption and omega-3 fatty acid status and determinants in long-term hemodialysis. American journal of kidney diseases 47(6): 1064-1071.
- Domei T, Hiroyoshi Yokoi, Shoichi Kuramitsu, Yoshimitsu Soga, Takeshi Arita, et al. (2012) Ratio of serum n-3 to n-6 polyunsaturated fatty acids and the incidence of major adverse cardiac events in patients undergoing percutaneous coronary intervention. Circulation Journal 76(2): 423-429.
- Shoji T, Ryusuke Kakiya, Tomoshige Hayashi, Yoshihiro Tsujimoto, Mika Sonoda, et al. (2013) Serum n-3 and n-6 polyunsaturated fatty acid profile as an independent predictor of cardiovascular events in hemodialysis patients. American Journal of Kidney Diseases 62(3): 568-576.
- Kapoor R and YS Huang (2006) Gamma linolenic acid: an anti-inflammatory omega-6 fatty acid. Curr Pharm Biotechnol, 7(6): 531-534.
- Shearer GC, Juan J Carrero, Olof Heimbürger, Peter Barany, Peter Stenvinkel (2012) Plasma fatty acids in chronic kidney disease: nervonic acid predicts mortality. J Ren Nutr 22(2): 277-283.
- Peck LW, ER Monsen and S Ahmad (1996) Effect of three sources of long-chain fatty acids on the plasma fatty acid profile, plasma prostaglandin E2 concentrations, and pruritus symptoms in hemodialysis patients. Am J Clin Nutr 64(2): 210-214.
- Varga Z, I Kárpáti, G Paragh, L Buris, G Kakuk (1997) Relative abundance of some free fatty acids in plasma of uremic patients: relationship between fatty acids, lipid parameters, and diseases. Nephron 77(4): p417-421.
- Organization WH International Association for the Study of Obesity (IASO), International Obesity Task Force (IOTF). The Asia-Pacific perspective: redefining obesity and its treatment. Melbourne: Health Communications Australia 2000: 20.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.