Research Article 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Association of Maternal Serum PIGF Levels as a Biomarker in Preeclampsia: A Case Control Tertiary Care Hospital Based Study
*Corresponding author:Krishnaveni C, Department of Anatomy, Viswa Bharathi Medical College, India.
Received:March 20, 2023; Published:March 30, 2023
DOI: 10.34297/AJBSR.2023.18.002469
Abstract
Objectives: Preeclampsia (PE) is a multi-systemic disorder with circulatory pressure of more than 140/90 mm Hg and proteinuria with 0.3 g in a 24 h on dipstick emerging after 20 weeks of pregnancy. In PE subjects, there will be incomplete arterial transformation and cytotrophoblastic cell division. It has pro-angiogenic effects on the feto-uterine circulation and maintenance of the trophoblast development. Due to imbalance in Placental Inhibiting Growth Factor (PIGF) levels in PE women, it constricts the blood vessels and leads to endothelial dysfunction. Low circulating PlGF precedes the manifestation of clinical disease in PE pregnancies and intrauterine growth restriction in fetus. The role of PlGF in PE needs to be focussed more. The present study aim is to compare the maternal serum PIGF levels in normotensive pregnant women to PE women in early and late gestational weeks. Material and Methods: In this case-control study, 150 were preeclamptic women as cases, and 150 as normotensive pregnant women as controls, a total of 300 subjects were associated in the present study. A 5ml of maternal venous blood was collected, the serum was separated and stored at -80˚C till the analysis. By using commercially available Enzyme-Linked Immuno Sorbent Assay kits from Chongqing Biospes Co., Ltd., (suppliers: Info bio-Company, New Delhi) was measured with microplate reader at 450nm (Merilyzer Eiaquant Company). Results: Out of 300 pregnant women, 46 pregnant women were early gestational weeks and 254 were late gestational weeks. The statistical analysis was performed by Statistical Packages for Social Sciences Software 22. The area under the receiver operating characteristic curve is 0.93, 92% sensitivity, and 77% specificity. The significance in the maternal serum PIGF levels was calculated by the Mann-Whitney U test. By comparing the cases and controls it was found that maternal serum PIGF was significantly higher in PE women with z value =2.65 and U= 9253 and with p-value 0.005 significance. Conclusion: Maternal serum PIGF can be used as a preeclamptic diagnostic marker in southeastern Kolar population.
Keywords: Pro-angiogenic Pregnancy Marker, Placental Inhibiting Growth Factor, Maternal circulating marker, vascular endothelial growth factor.
Introduction
Pre-eclampsia (PE) is a pregnancy-specific, vascular angiogenesis imbalance associated with endothelial dysfunction. The abnormalities in the development of the placental vasculature occur at early or late gestation but the development of clinical manifestations was evident at the 20th week of PE pregnancy. Placental growth factor (PlGF), a component of the vascular endothelial growth factor (VEGF) family, play a key role in the remodeling process of maternal arteries in normal pregnancy. This growth factor is released into the bloodstream by the migration of trophoblasts. The placental growth factor (PIGF/ PLGF/PGF) belongs to the cysteine-knot superfamily [1]. Placental protein binds to form a heterodimer with VEGFR-1/FLT-1/soluble variant sFLT-1 (soluble fms Feline McDonough Sarcoma -like tyrosine kinase-1). It also binds to neuropilin receptor1 and 2, recognized in the placenta but its role is still to be highlighted in detail [2]. PIGF is secreted in placental tissues, neurons, and pathological conditions like colon and breast carcinomas. It signals through the vascular endothelial growth factor receptor-1 VEGFR1/FLT1(fms-related tyrosine kinase-1) which stimulates endothelial cell proliferation and migration [1]. PlGF concentrations in early pregnancy are lower in women who later develop PE. However, as the pregnancy progresses, there is a reciprocal relationship between sFLT-1 and PlGF. Low PlGF levels are evident for abnormal placentation, small for gestational age babies which further leads to abnormal growth in the second half of pregnancy. So it supports the hypothesis that PlGF is an indicator of abnormal placentation [2].
Circulating PlGF is prominently elevated in pregnancy from the placenta. The function of PlGF in the placenta is development and maturation of the placental vascular system. Placental expression of PlGF dominates from the second trimester when the uteroplacental circulation is advancing, with myometrial spiral arteries remodeling beginning at 16–18 weeks of gestation [2]. Concentrations of PlGF are low in the first trimester of an uncomplicated pregnancy and increases from 11 to 12 weeks onwards to a peak at 30 weeks, after which it decreases. Standard levels of PlGF are dependent on gestational age, normal levels ranging from approximately 141 pg ml− 1 at around 30 weeks gestation to 23 pg ml− 1 at term [3]. The main role of PlGF in tissues is angiogenesis, imbalance in the concentration of PIGF levels leads to pathological ischemia. PlGF expression in trophoblast (transcriptional activity) is suppressed by hypoxia and increased by a normoxic environment and further related to intrauterine events [2]. With a combination of other angiogenic factors such as soluble endoglin and Doppler sonography are more useful for predicting early-onset PE. The angiogenic factors correlate with disease severity, can be detected several weeks before clinical presentation, and have predictive value for the diagnosis of severe- early onset PE [4]. So PIGF is specific to pregnancy and vascular development. 3 There was a lot of discrepancies to deliberate about the PIGF levels in the diagnostic precision between early onset (EO) and late onset (LO) of PE [5]. In this regard, the study aims to compare the maternal serum concentration of PIGF levels in normotensive pregnant women to PE women in early and late gestational weeks.
Materials and Methods
After approval from the central ethics committee, the information was collected from the period of August 2017 to October 2018, subsequently written informed consent from each study subject involved in this investigation were collected. The subjects were from the Department of Obstetrics and Gynecology at R.L.Jalappa Research Institute and Teaching Hospital.In this casecontrol study, sums of 348 pregnant women were included, out of these 48 (16%) dropouts from the investigation. In this study, 150 pregnant women who were determined with PE dependent on the American College of Obstetrics and Gynecologists in the Department of Obstetrics & Gynecology in R. L. Jalappa Research Institute & Teaching Hospital. The remaining 150 were controls considered as normotensive pregnant women under early and late PE.
Diagnostic Criteria of Preeclampsia: According to the American College of Obstetrics and Gynecologist PE was characterized as new beginning of hypertension (Blood Pressure ≥ 140/90 mm Hg) and proteinuria (urine dipstick of ≥ 1+) following 20 weeks of gestation. Without proteinuria, additionally PE was characterized by extreme hypertension (≥160/110 mm Hg) and hemolysis, low platelets count, and increased liver enzymes (HELLP syndrome) or indications (i.e., headache/migraine, visual changes, and right upper quadrant of stomach discomfort). Renal deficiency in creatinine ≥ 1.1 mg/dl [6]. PE further ordered into early and late beginning, which is related with various pathogenesis, clinical outcome, and prognosis [7]. The PE women with the ≥ 20th week of gestation were incorporated as cases. Another 150 normotensive pregnant women with the ≥ 20th week of gestation without any pregnancy complexity till delivery were controls.
Exclusion Criteria:Pregnant women with gestational hypertension, persistent hypertension, gestational diabetes, history of multiple premature births and abortions, past pregnancy with an abnormal fetus and with developmental anomalies, and thrombophilia-like disorders were eliminated from the study.
Inclusion Criteria:Pregnant women who were diagnosed with preeclampsia after the 20th week of gestation, both primigravida and multigravida were incorporated as preeclamptic women and who were healthy normotensive pregnant women after the 20th week of gestation without any complications till delivery were incorporated as controls.
Maternal serum sample collection and analysis: Under complete aseptic precautionary measures, 5ml of maternal venous blood was collected in red-covered vacutainers; the serum was isolated by centrifugation and stored at -80˚C till further investigation. Before 2 days the samples were stored at -20˚C at that point steadily to 4˚C and afterward gradually brought to room temperature. By using commercially available Enzyme-Linked Immuno Sorbent Assay (ELISA) kits from Chongqing Biospes Co., Ltd., (suppliers: Info bio-Company, New Delhi) was measured by following the complete protocol carefully according to guidelines. The maternal serum analysis was completed by estimating with an ELISA microplate reader at 450 nm (Merilyzer Eiaquant Company).
Statistical Analysis:The outcomes were interpreted by verifying normal distribution which was analyzed by the Mann- Whitney U test due to non-distribution invariance. The AUC (Area Under the receiver operating characteristics (ROC) curve) estimation value of PIGF is 0.93. The statistical analysis of mean, median, sensitivity, and specificity with cut-off variables was determined and deciphered as an ROC curve by utilizing Statistical Packages for Social Sciences Software SPSS (version 22.0; SPSS Inc, Chicago, IL, USA).
Results
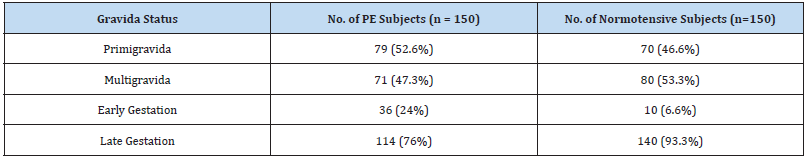
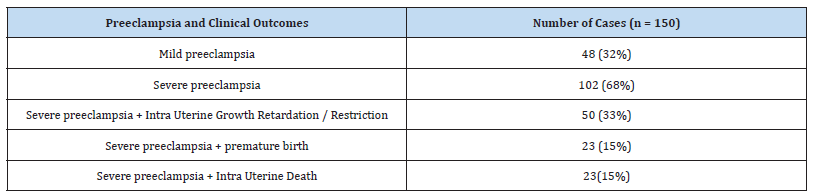
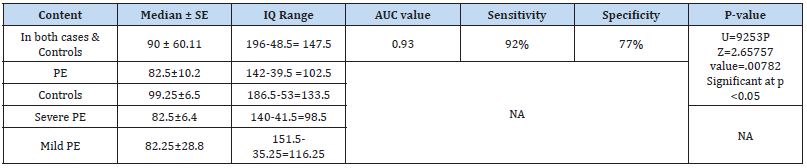
Out of 300 pregnant women based on the gestational weeks, the early gestational weeks were 46 and late gestation was 254. According to their gravida status primigravida were 149 and multigravida were 151 subjects Table 1. The severe PE was 68% and the remaining were mild PE Table 2. The statistical analysis was analyzed by using SPSS software. After analysing the normal distribution curve, the statistical significance of maternal PIGF levels in cases and controls was calculated by the Mann-Whitney U test Table 3. The maternal age, gestational weeks, systolic, and diastolic blood pressures in cases and controls were analysed by the Mann- Whitney U test explained in Table 4. By comparing cases and controls the maternal serum PIGF levels were significantly higher in PE cases (z value =2.65) U= 9253. The p-value is significant with (≤0.05) especially if we compare with early and late gestational weeks, in late gestational weeks the PIGF levels were extremely significant. The AUC curve value is 0.93, 92% sensitivity, and 77% specificity Table 3 and Figure1. In 52.6% primigravida were PE patients. So, this discloses that PE is more susceptible to primigravidas. If we relate the median values of maternal age in cases and controls there was no much change among early and late gestation, but in gestational age, we found a major difference when compared to late and early PE. In late PE the gestational weeks are 37 and whereas in early PE was 30 weeks and in controls, late gestation was extended to 39 weeks. So this designates that due to PE there were more probabilities of early delivery which affects the development of the fetus. If we compare the serum PIGF levels in early and late PE, in early gestational weeks of PE the serum PIGF levels are high in maternal circulation compared to late gestational weeks. In late gestational weeks in controls and PE, the levels were highly significant than early gestational weeks of PE and in controls Table 4. (Figure 1, Table 1-4)
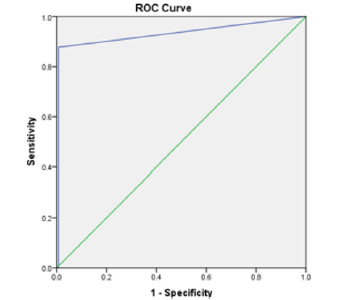
Figure 1:AUC (Area Under the receiver operating characteristics (ROC) curve) estimation value of PIGF is 0.93.
n- Number of subjects, Cases- Preeclamptic pregnant women and Controls – Normotensive pregnant women, PE- Preeclampsia
*Significant with p value ≤ 0.0001, IQ Range- Inter Quartile Range, AUC- Area under the receiver –operating characteristics, SD-Standard Deviation, SE-Standard Error, NA- Not Applicable
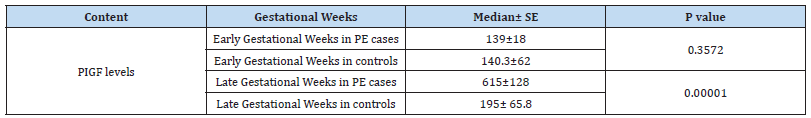
Table 4:Statistical Outcome in Early and Late Preeclamptic Women (Cases) versus Early and Late Normotensive Pregnant Women (Controls).
Significant with p value ≤ 0.0001, n- Number of subjects, SD -Standard Deviation, SE- Standard Error, Numbers of subjects involved in the study were as follows: Early Gestational Weeks in PE cases n=36; Early Gestational Weeks in controls n=10; Late Gestational Weeks in PE cases n=114; Late Gestational Weeks in controls n=140
Discussion
The PIGF can be a diagnostic marker in individuals especially in southern part of India. In PIGF, 4 altered forms were found. PlGF may respond to hypoxia or ischemia, and other comorbidities like obesity, ethnicity [1]. For healthy fetal growth there should be a proper uterine establishment with healthy placental angiogenesis. This plays a vital role in placental perfusion. Imbalance in angiogenesis and vasculogenesis factors affects the remodeling of uterine spiral arteries and angiogenesis which can be one of the reasons for the pathogenesis of PE [8]. PlGF expression is thought to be suppressed by persistent placental hypoxia caused by an underdeveloped uteroplacental circulation. However, studies have been shown that PlGF expression in PE placental tissue is either increased or unchanged. PlGF expression regulation is unknown, but several mechanisms have been investigated. PlGF expression may be influenced by inflammation, as PlGF concentrations are elevated even in sepsis [2]. In a mouse-transgene designed model by Chau et al., study revealed that PIGF promotes angiogenesis for the fetoplacental circulation and supports trophoblast development but the decidual invasion is not influenced. Consequently, abnormal spiral artery remodeling in mice does not lead to placental insufficiency or abnormal blood pressure regulation [2]. In a biomarker study conducted by Giardini et al., suggested that PlGF is a biochemical marker not only for PE but also for placental dysfunction [9]. In a multi-centric study, PIGF levels range from 12 to 3000 pg/mL can be a preferable early clinical marker of adverse suspected PE pregnancy [10]. In another study by Lai et al., Myatt et al and Youssef et al., AUC ranges from 0.61 and 0.734. Poon and Youssef et al in a cohort study evidenced that PIGF is a diagnostic marker in PE. PIGF levels were significantly lower in first trimester serum samples of subjects who later developed PE [11-13]. In a crosssectional study performed by El- Deen et al., with 74 pregnant women, and 50 PE Egyptian women examined for the maternal serum PIGF levels between severe and mild PE. They found there is no statistical significance between severe PE and mild PE groups. When compared with controls to PE women, the PIGF levels were significant [4] which matches with the present study.
The Indian studies on PIGF role or in combinations for prediction of PE availability has been very limited [14]. From India total of 3 studies (original studies) were performed, from north India (all from Delhi) 3 studies and from south India 1 study was done and present study is 2nd in southern part of India. From northern part of India, in Delhi by Agarwal et al., a study was performed on 17 PE cases, the levels of PIGF were compared between early and late PE but not statistically significant between early and late PE but when compared to PE and controls the levels were significant. The cut-off values in early PE were <30 pg/ml with 88.2% sensitivity and 71.4% and in late PE was <32 pg/ml with a sensitivity of 86.7% and specificity of 74.3% [3]. In a cohort study by Ghosh et al., from Delhi, measured maternal serum PlGF levels at 11–14 weeks and 22–24 weeks of gestation. So, it proved that PIGF is an early second trimester marker as maternal serum PIGF levels are <144 pg/ml at 22–24 weeks of gestation were much higher than those of serum PlGF <228 pg/ml at 11–14 weeks of gestation [15]. In a case control study by Varughese et al., from Delhi with 80 study subjects measured the maternal sera PIGF levels. The mean PlGF levels are 236.77 pg/ml in PE whereas in controls the mean PIGF levels are 744.98 pg/ml [16]. Another twin study by Agarwal et al., studied on PIGF and biophysical markers in late first trimester. They deliberate on 291 singleton pregnant women at 11–14 weeks of gestation who are primigravida of age group <40 years. PIGF alone had detection rate of 40% in PE [14]. The reduced levels of PlGF are evident from the first trimester, before 5 weeks the onset of PE. So, this is evident that PIGF levels will be lower from early gestational weeks itself especially in suspected PE. So, the maternal serum PlGF, would identify about 90% and 50% of patients developing early PE and late PE. There was a significant association between PlGF and the severity of PE also [17]. Present study results match with this study results. Chappell et al., in their study observed that PlGF has high sensitivity and predictive value for PE within 14 days in early onset suspected PE. So PIGF alone has a better predictive value [18] Treatment of PE with PlGF is promising but with many uncertainties remain [2].
Acknowledgments
Thanks to the nurses, especially Mrs. Jyothi.K, Mrs. Maya sisters, and other sisters of the OBG department for assisting to complete the project.
Conflict of Interest
None of the authors declare conflict of interest.
Funding Support
There were no funding sources. It is a self-financed project.
References
- Almaani SJ (2019) Placental growth factor in pre-eclampsia: friend or foe? Kidney International 95(4): 730-732.
- Chau K, Hennessy A, Makris A (2017) Placental growth factor and pre-eclampsia. J Hum Hypertens 31(12): 782-786.
- Agarwal R, Chaudhary S, Kar R, Radhakrishnan G, Sharma R, et al. (2017) Serum placental growth factor in late first trimester of pregnancy for prediction of preeclampsia in primigravida - a case control study. Int J Reprod Contracept Obstet Gynecol 6(1): 106-110.
- EL-Deen IM, Seleem AK, Aziz AAA, Sayd EH EL, Dahtory F, et al. (2019) Study of the Vascular Endothelial Growth Factor Gene Expression and Serum Placental Growth Factor Levels in Egyptian Women with Preeclampsia. IOSR Journal of Pharmacy 9(6): 28-37.
- Agrawal S, Shinar S, Cerdeira AS, Redman C, Vatish M (2019) Predictive Performance of PlGF (Placental Growth Factor) for Screening Preeclampsia in Asymptomatic Women: A Systematic Review and Meta-Analysis. Hypertension 74(5): 1124-1135.
- March MI, Geahchan C, Wenger J, Raghuraman N, Berg A, et al. (2015) Circulating Angiogenic Factors and the Risk of Adverse Outcomes among Haitian Women with Preeclampsia. PLoS ONE 10(5): e0126815.
- Kahnamouei aghdam F, Pourfarzi F, Ehsani S (2018) Evaluation of serum placental growth factor in predicting pregnancy outcomes in women with suspected pre-eclampsia. Int J Adv Med 5(1): 11-15.
- He A, Zhou Y, Wei Y (2020) Potential Protein Biomarkers for Preeclampsia. Cureus 12(6): e8925.
- Giardini V, Rovelli R, Algeri P, Giunti L, Lazzarin S, et al. (2020) Placental growth factor as a predictive marker of preeclampsia – PREBIO study – Preeclampsia Biochemical study. The Journal of Maternal-Fetal & Neonatal Medicine 35(16):3029-3035.
- Barton JR, Woelkers DA, Newman RB, Combs CA, How HY, et al. (2020) Placental growth factor predicts time to delivery in women with signs or symptoms of early preterm preeclampsia: a prospective multicenter study. Am J Obstet Gynecol. 222(3): 259.e1-259.e11.
- Youssef A, Righetti F, Morano D, Rizzo N, Farina A, et al. (2011) Uterine artery Doppler and biochemical markers (PAPP-A, PIGF, sFlt-1, P-selectin, NGAL) at 11 + 0 to 13 + 6 weeks in the prediction of late (> 34 weeks) preeclampsia. Prenat Diagn 31(12): 1141-1146.
- Myers JE, Kenny LC, McCowan LM, Chan EH, Dekker GA, et al. (2013) Angiogenic factors combined with clinical risk factors to predict preterm preeclampsia in nulliparous women: a predictive test accuracy study. Br J Obstet Gynaecol 120(10): 1215-1223.
- Lai J, Pinas A, Poon LC, Agathokleous M, Nicolaides KH, et al. (2013) Maternal serum placental growth factor, pregnancy-associated plasma protein-a and free β-human chorionic gonadotrophin at 30-33 weeks in the prediction of preeclampsia. Fetal Diagn Ther 33(3): 164-172.
- Agarwal R, Chaudhary S, Kar R, Radhakrishnan G, Tandon A, et al. (2017) Prediction of preeclampsia in primigravida in late first trimester using serum placental growth factor alone and by combination model, Journal of Obstetrics and Gynaecology 37(7): 877-882.
- Ghosh SK, Raheja S, Tuli A, Raghunandan C, Agarwal S, et al. (2013) Is serum placental growth factor more effective as a biomarker in predicting early onset preeclampsia in early second trimester than in first trimester of pregnancy? Archives of Gynecology and Obstetrics 287(5): 865-873.
- Varughese B, Bhatla N, Kumar R, Dwivedi SN, Dhingra R, et al. (2010) Circulating angiogenic factors in pregnancies complicated by pre-eclampsia. Natl Med J India 23(2): 77-81.
- Akolekar R, Zaragoza E, Poon LC, Pepes S, Nicolaides KH, et al. (2008) Maternal serum placental growth factor at 11 + 0 to 13 + 6 weeks of gestation in the prediction of pre- eclampsia. Ultrasound Obstet Gynecol 32(6): 732-739.
- Chappell LC, Duckworth S, Seed PT, Griffin M, Myers J, et al. (2013) Diagnostic accuracy of placental growth factor in women with suspected preeclampsia: a prospective multicenter study. Circulation 128(19): 2121-2131.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.