Review Article 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Reduction of Sarcopenia Using HIFEM and RF in Two Age Stratified Cohorts: A Prospective Clinical Trial
*Corresponding author:Diane Duncan, M.D., FACS, Plastic Surgical Associates, Fort Collins CO, 1701 East Prospect Road, USA.
Received:February 23, 2021; Published:March 30, 2023
DOI: 10.34297/AJBSR.2023.18.002464
Abstract
The definition of sarcopenia is the progressive reduction of muscle mass with declining physical ability due to loss of strength and mobility. Usually associated with aging and a sedentary lifestyle, this condition can also result from an injury or extended illness. If allowed to progress, a poor quality of life with limitation of longevity can follow. Of course, the top recommendation for correction of muscle mass loss and functional performance improvement is to advise the patient to exercise more. In some cases, this solution is not tenable, as the person may be too weak to regenerate muscle mass efficiently. Another obstacle is habit; the older population traditionally does not maintain a strong exercise regime or may not have the opportunity to do so safely. The EGWSOP (European Working Group on Sarcopenia in Older People) [1] notes that age related sarcopenia is commonly associated with other comorbid conditions such as obesity, diabetes, osteoporosis, rheumatoid arthritis, anorexia, HIV, and other chronic debilitating conditions.
The aim of our prospective study was to evaluate the effects of consecutive application of HIFEM plus radiofrequency (RF) procedures transabdominally plus seated application of HIFEM in two age stratified cohorts. Fourteen patients were under the age of 60, and 16 patients were aged 60 and over. Three patients were male and 27 were female. Four weekly treatments with transcutaneous abdominal HIFEM plus radiofrequency and six twice weekly treatments of seated HIFEM alone were performed over a period of four weeks. Measurements taken included comfortable gait speed, maximum gait speed, balance, number of steps completed in two minutes, and timed up and go test (TUG). DEXA scans and bioimpedance analysis were performed.
A Likert 5-point patient satisfaction survey and therapy comfort survey was completed for each patient. Treatment results were evaluated at 1 month and 3 months following completion of treatment. Mean improvement in comfortable gait speed was 32.9% and maximum gait speed increase averaged 25.9%. Timed up and go test times improved by 21.1%. The number of steps completed in a measured 2-minute time period improved by 18.4%. Balance improvement, a significant predictor of longevity, increased by a measured 11.6% in the entire group. The study noted improvement in all test values over the broad group of patients, while patients > 60 years improved more profoundly. The conclusion was that combined HIFEM plus RF treatments effectively improved sarcopenia, especially in the older age group.
Keywords: Sarcopenia, Muscle wasting, Frailty, Balance, Core Strength, Longevity
Introduction
Sarcopenia is the involuntary loss of muscle mass and function [2] manifesting after the third decade of one´s life. 1-2% of muscle mass is lost yearly, with up to 3% of yearly muscle mass loss occurring in geriatric age [3]. By age 80, muscle mass reduction overall is estimated to be 50%. Multiple factors play a role in sarcopenia development, including hormonal changes, a decrease in nutrition and activity, and an increase in inflammation [4]. However, the most physically deleterious cause of sarcopenia is the decrease in the number of neuromuscular junctions and type-II muscle fiber [5], which affects the functionality of muscles due to decreased time in force development upon activation [6]. Osteosarcopenia, defined as sarcopenia presenting with osteoporosis, is a common occurrence in geriatric patients [7].
This overall decline in the musculoskeletal system contributes to adverse outcomes, such as increased incidence of falls, a decline in functionality, an increase in frailty, and mortality, with a correlation to decreased quality of life and cardiometabolic risk factors such as diabetes or hypertension [8-10]. Sarcopenia can also be used as a measurement of fragility, with the found correlation between the low core muscle area and an increased postoperative complication rate [11]. There are various ways to evaluate sarcopenia based on muscle quantity and quality [12]. The assessment of muscle quality is determined by the physical performance tests, which are considered the most valid as it shows the changes in muscle functionality [16]. Among the physical performance tests, the gait speed, timed-up-and-go test (TUG), the Berg Balance test, and the stair step test are the most common [13-15]. To assess the changes in muscle mass quantity, the bioelectrical impedance analysis (BIA) can be used [16]. The combination of qualitative and quantitative tests to assess sarcopenia is recommended [17]. For detecting the extent of osteoporosis and measuring the bone mineral density (BMD) by dual-energy x-ray absorptiometry (DXA) can be used [18].
One of the ways to slow down the symptoms of sarcopenia and osteoporosis is through resistance exercise (RE) [19]. However, the drop-out rate from the training programs in older adults can be up to 33% [20], as higher age and poorer physical performance are associated with a higher drop-out rate [21]. Therefore, noninvasive HIFEM technology may serve as an alternative to exercise. The technology is based on the alternating magnetic field that depolarizes the motor neurons in skeletal muscle tissue, causing contractions of supramaximal nature, which cannot be achieved voluntarily [22], with the muscle tissue adapting to this workload by inducing hypertrophy [23]. The standalone HIFEM procedure was shown to strengthen the rectus abdominis and reduce abdominal separation [24], enhance the gluteal muscles [25], and tone the upper arm and calf muscles [26].
The HIFEM technology is also used to strengthen the pelvic floor muscles (PFM) for treating urinary incontinence in both men and women, thus improving their quality of life [27,28]. To enhance the effectiveness of HIFEM technology, it has been combined with synchronized radiofrequency (RF). The heating of the muscle tissue enhances the regenerative processes through activating satellite cells (SC), muscle-delivered stem cells, which induce differentiation of the muscle fibers and increase blood flow to boost metabolic exchange [29]. The superior effects of HIFEM+RF have been shown on the abdomen [30], gluteal muscles [31], inner [32] and outer thighs [33], and upper arms [34]. Using a standalone HIFEM procedure over the pelvis and simultaneous application of HIFEM+RF over the abdomen aims to strengthen the abdominal and pelvic floor muscles, aka the core muscles.
In many cultures, progressive weakness and frailty with advancing age is an expectation. Our study was performed in order to validate a solution which might be helpful globally for our aging population. The technologies mentioned above have established roles in aesthetic medicine, especially muscle toning, which infers muscle strengthening. This study investigates the effects and safety of consecutive application of synchronized application of HIFEM+RF with a standalone HIFEM procedure for possible treatment of sarcopenia.
Methods
Eligible patients (≥21 years, BMI <35 kg/m2) had their medical records screened for possible exclusion criteria defined as medical conditions contraindicating the application of electromagnetic field and radiofrequency, such as implants (metal and electronic), cardiovascular disease, malignant tumor, pregnancy, and postpartum. Thirty patients (27 women, 3 men, 26-75 years old, BMI 19.7-33.5 kg/m2) who met inclusion criteria were enrolled in this prospective, open-label, single-center, one-arm study. Out of the enrolled patients, sixteen (n=16) patients were above 60 years old (60-75 years old, BMI 20.5-30.4 kg/m2). All study subjects were instructed about the treatment procedure and provided written informed consent. The study protocol followed the Declaration of Helsinki. Patients were instructed to maintain their lifestyle regarding their diet and physical activity regimen.
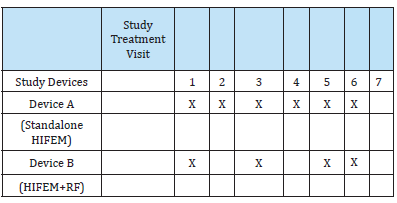
In this study, two devices were used in combination. Device A utilizes standalone HIFEM energy (EMSELLA, BTL Industries Inc., Boston, MA) to treat pelvic floor muscles. This device includes a chair applicator with a 2.5T generator. Patients sit upright on the applicator while fully clothed for the 28-minute treatment duration. The intensity of the HIFEM field is set according to the patient´s maximal tolerated level (0-100%). Device B utilizes the simultaneous application of HIFEM with synchronized radiofrequency (Emsculpt Neo, BTL Industries, Boston, MA) and is equipped with a large applicator. Patients lay on the medical table in a supine position. The large applicator is fixed with a secure belt over the abdomen for a 30-minute treatment. The HIFEM and RF intensities are set to a patient´s maximal tolerated level (0-100%). The treatment protocol included seven (n=7) treatment sessions. Four HIFEM+RF abdominal procedures spaced 5-10 days, with six standalone HIFEM procedures on the pelvic floor spaced 2-4 days, were applied. Both procedures were used consecutively at the first, third, and fifth treatment visits, starting with HIFEM+RF (see Table 1 for clarity). Two follow-up visits were scheduled 1 month and 3 months after the treatments (Table 1).
The Primary Outcomes
The primary evaluation method included five standard tests for assessing sarcopenia. First, the maximum and comfortable gait speed was evaluated by timing a 7.5-meter walk (approx. 25 feet) at a comfortable and maximal speed. The measurement unit is meters per second (m/s). The timed up-and-go test (TUG) tested the time (in seconds, s) a patient needs to get up from a chair, walk 10 feet, turn, walk back to the chair, and sit down. The balance test consisted of a total of 3 attempts. First, patients stood only on the left leg while trying to maintain balance, then the right leg, and for the third attempt, patients could choose either leg. The time (in seconds, s) was measured with a 45-second cap. The attempts were then averaged to obtain one number. The last test included measuring the number of steps (N) and simulating the stair-step test. The patients were asked to step onto a 6-inch-high platform and step off, then the number of steps was counted for two minutes. The data were collected at baseline, 1-month, and 3-month followup visits.
The Secondary Outcomes
The secondary outcome was based on InBody evaluation and dual-energy X-ray absorptiometry (DXA) measurements. The n Body 770 (Cerritos, CA, USA) was used to measure the changes in skeletal muscle mass (SMM, lb) and visceral adipose tissue (VAT, cm2). DXA (Hologic Horizon W, Hologic Inc., Danbury, CT, USA) scans were used to evaluate the changes in bone mineral density (BMD, T-score). The T-score is calculated as (the patient´s BMDyoung BMD mean)/SD of young [29]. There are three diagnostic categories: normal BMD (T-score≥-1.0), osteopenia (T-score<1.0 and >-2.5), and osteoporosis (T-score ≤ -2.5) [29]. Additionally, digital photographs and waist circumference were taken. Data for secondary outcomes were collected at baseline, at 1-month, and 3-month follow-up visits. The statistical significance was tested by One Factor ANOVA Repeated Measures with Tukey HSD post-hoc test. The significance level α was set at 5%.
Results
All patients finished the treatment sessions and both follow-up visits. No adverse events or side effects were reported or observed. The patients did not report any discomfort with the consecutive application of both devices and resumed their daily activities right after the treatments without any issues. The BMI of the subjects did not significantly change throughout the study (p-value > 0.05).
Primary Outcomes
At baseline, the average comfortable gait speed and maximal gait speed were 1.4 m/s and 2.1 m/s. At 1-month post-treatment, the average comfortable gait speed increased to 1.6 m/s (+10.9%), and the maximal gait speed increased to 2.3 m/s (+5.8%). The results peaked at a 3-month follow-up visit with an average increase in comfortable and maximal gait speed by 32.9% (p-value <0.001) and 25.9% (p-value <0.001), respectively. In addition, patients above 60 years old showed an increase of 45.6% (p-value < 0.001) and 37.5% (p-value = 0.01) in comfortable and maximal gait speed at 3-month follow-up (for more detailed information, see Table 1). On average, patients needed 7.1s to complete the TUG test at the baseline visit. However, the time to complete the TUG test gradually reduced over 1-month and 3-month follow-up visits after the treatments, demonstrating an improvement by -16.9% (p-value < 0.001) and -21.1% (p-value < 0.001), respectively. Patients over 60 showed similar improvement to the whole group (-18.9% at 1-month follow-up, -20.6% at 3-month follow-up).
For the balance test, patients (n=13) that reached the 45-second cap at baseline and both follow-up visits were excluded from the evaluation. At baseline, the patients (n=17) could hold their balance up to 22.2s on average. All evaluated patients demonstrated an improvement in balance at 1-month and 3-month follow-up. The patients could hold their balance for 23.5s at the 1-month followup and 28.8s (+29.7%, p-value = 0.04) at the 3-month follow-up. Patients over 60 improved their balance by +30.3% at a 3-month follow-up (p-value = 0.04). On average, patients could complete 70 steps in two minutes at baseline visits. At the 1-month follow-up, the number of steps increased by 16.4% (p-value <0.001), with an 18.4% increase at the 3-month follow-up. In addition, patients over 60 could do 20.9% more steps at a 3-month follow-up (p-value < 0.001).
Secondary Outcomes
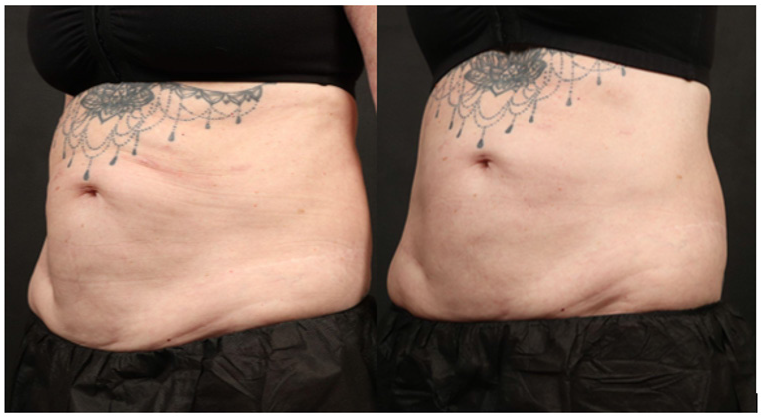
On average, study subjects had a circumference reduction of -3.6 cm at 1-month follow-up and -3.5 cm at 3-month followup (p-value < 0.001). The changes are visible in Figure 1. Due to scheduling conflicts, two patients did not undergo the In Body or DXA measurements; therefore, only 28 patients were included in evaluating In Body parameters and DXA scans. The baseline bioimpedance analysis (In Body, Cerritos, CA) measurements showed an average skeletal muscle mass of 58.7±16.8 lbs. At 1-month follow-up, patients (n=28) gained +0.5 lb of SMM, with +0.9 lb at 3-month follow-up (p-value < 0.05). Patients over 60 (n=15) showed an average SMM of 54.5±15.2 lb at baseline and gained +0.93 and +1.4 lb of skeletal muscle mass increase at 1-month and 3-month follow-up (p-value = 0.009), respectively..
The evaluation of VAT showed an exciting finding. At the end of the study, 18.2% of patients with high VAT (above 100 cm2) showed a reduction in VAT and were in the normal VAT values (p-value = 0.007) at the 3-month follow-up visit. On the other hand, 25% of patients with extremely high VAT (above 160 cm2) showed a reduction in VAT and came down the range of high VAT values (p-value < 0.001) at the 3-month follow-up visit.
At baseline, the DEXA measurements showed 23 patients in the normal levels of BMD, 4 in the osteopenia levels, and 1 in the osteoporosis levels. During the study, no changes were observed in the BMD (p-value > 0.05) (Table 2 & Figure 1).
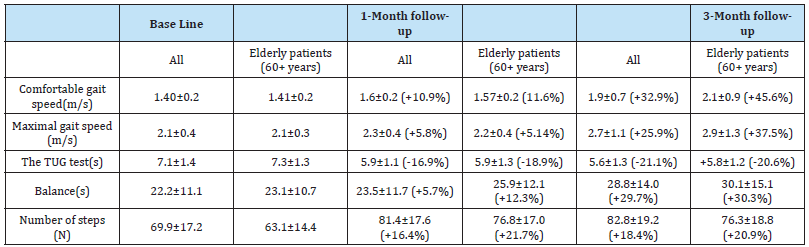
Table 2:An overview table of primary outcomes, value ± standard deviation, and percentage of change (%). For the comfortable gait speed, maximal gait speed, TUG test, and the number of steps, 30 subjects were included in the All group, with 16 patients in the elderly patient group. Seventeen (n=17) patients were evaluated for balance improvement in the All group, with 13 patients evaluated in the elderly group.
Discussion
This prospective study aimed to investigate the effect of consecutive application of standalone HIFEM over the pelvis and simultaneous HIFEM and radiofrequency over the abdomen to improve the symptoms of sarcopenia. The treatments were well tolerated, and patients could continue their daily activities uninterrupted. The muscle quality assessment through the evaluation of 5 functional tests and bioelectrical impedance analysis demonstrates the viability of the HIFEM modality for the treatment of sarcopenia. No adverse events or side effects occurred during this study. Sarcopenia is a syndrome of decline in muscle mass and function, which interferes with the daily activities of patients [30]. The strength of the core muscles is associated with an increase in comfortable and maximum gait speeds [31] and balance, including the step test as a measurement of dynamic balance and the TUG test [32,33]. This presented study showed a significant improvement in all primary outcomes, with more profound results in the group over 60 years old.
The result of all study participants peaked at 3-month followup, especially comfortable and maximum gait speeds (+32.9% and +25.9%, respectively), the TUG test (-21.1%), balance (+29.7%), and the number of steps (+18.4%). In the group with patients over 60 years old, the comfortable and maximum gait speeds (+45.6% and +37.5%, respectively), the TUG test (-20.6%), balance (+30.3%) peaked at 3-month follow-up, with the number of steps test (+21.7%) peaking at 1-month follow-up. However, the favorable results were maintained with a +20.9% increase in the number of steps at 3-month follow-up. Resistance Exercise (RE) strengthens the muscle by stimulating muscle protein synthesis and hypertrophy [34,35]. The HIFEM+RF technology has been shown to induce hypetrophy 17, with results comparable to 12- 16 weeks of intense exercise [36]. Measuring the quantitative increase in muscles by bioelectrical impedance analysis detected a +0.9 lb increase of SMM, with patients over 60 showing a+1.4lb increase of SMM, both at 3-month follow-up, corresponding with the improvement in functional performance of patients.
Bone strengthening via resistance training is based on mechanical loading, a process of changing bone formation in response to mechanical stress [37]. However, bone strengthening may be independent of changes in BMD as BMD is just one of many determinants of bone strength [38]. This study’s strengths are the combination of five functional tests to assess muscle function and the bioelectrical impedance analysis to measure the changes in skeletal muscle mass directly. The zero drop-out rate also ensured a comprehensive statistical analysis of the changes occurring in all patients after the treatments. Although the unequal representation of male (n=3) and female (n=27) subjects was a drawback, it may be beneficial to extrapolate this research to more males. DXA for assessing bone strength only captures the bone mass and not bone size, shape, or structure, which also contributes to bone strength38. Future research should consider using quantitative computed tomography (pQCT) to measure changes in the bones and establish a control group.
Conclusion
The evaluation of the five functional tests for sarcopenia assessment combined with the bioelectrical impedance analysis showed that the consecutive use of standalone HIFEM over the pelvis and HIFEM with synchronized radiofrequency over the abdomen significantly improve the functional performance of skeletal muscles in all age groups and thus battle the signs of sarcopenia, especially in patients over 60 years of age.
References
- Cruz-Jentoft AJ, Bahat G, Bauer J, Boirie Y, Bruyère O, et al. (2019) Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing 48(1): 16-31.
- Kim JW, Kim R, Choi H, Lee SJ, Bae GU, et al. (2021) Understanding of sarcopenia: from definition to therapeutic strategies. Arch Pharm Res 44(9-10): 876-889.
- Walston JD (2012) Sarcopenia in older adults. Curr Opin Rheumatol 24(6): 623-627.
- Hepple RT (2003) Sarcopenia--A Critical Perspective. Sci Aging Knowledge Environ (46): pe31.
- Inoue T, Maeda K, Nagano A, Akio Shimizu, Junko Ueshima, et al. (2021) Related Factors and Clinical Outcomes of Osteosarcopenia: A Narrative Review. Nutrients 13(2): 291.
- Cruz-Jentoft AJ, Bahat G, Bauer J, Yves Boirie, Olivier Bruyère, et al. (2019) Sarcopenia: revised European consensus on definition and diagnosis. Age and Ageing 48(1): 16-31.
- Kim Y, Park KS, Yoo JI (2021) Associations between the quality of life in sarcopenia measured with the SarQoL® and nutritional status. Health and Quality of Life Outcomes 19(1): 28.
- Hasselager R, Gögenur I (2014) Core muscle size assessed by perioperative abdominal CT scan is related to mortality, postoperative complications, and hospitalization after major abdominal surgery: a systematic review. Langenbecks Arch Surg 399(3): 287-295.
- Mijnarends DM, Meijers JMM, Halfens RJG, Sovianne ter Borg, Yvette C Luiking, et al. (2013) Validity and Reliability of Tools to Measure Muscle Mass, Strength, and Physical Performance in Community-Dwelling Older People: A Systematic Review. Journal of the American Medical Directors Association 14(3): 170-178.
- Orlandini L, Rolland Y, Cesari M (2021) Measurements of Physical Performance. In: Sarcopenia. John Wiley & Sons, Ltd 253-270.
- Ackermans LLGC, Rabou J, Basrai M (2022) Screening, diagnosis and monitoring of sarcopenia: When to use which tool? Clinical Nutrition ESPEN 48: 36-44.
- Blake GM, Fogelman I (2007) The role of DXA bone density scans in the diagnosis and treatment of osteoporosis. Postgrad Med J 83(982): 509-517.
- Hong AR, Kim SW (2018) Effects of Resistance Exercise on Bone Health. Endocrinol Metab (Seoul) 33(4):435-444.
- Zech A, Drey M, Freiberger E (2012) Residual effects of muscle strength and muscle power training and detraining on physical function in community-dwelling prefrail older adults: a randomized controlled trial. BMC Geriatrics 12: 68.
- Geirsdottir OG, Chang M, Briem K, Jonsson PV, Thorsdottir I, et al. (2017) Gender, Success, and Drop-Out during a Resistance Exercise Program in Community Dwelling Old Adults. J Aging Res.
- Duncan D (2020) A Novel Technology Combining RF and Magnetic Fields: Technical Elaboration on Novel RF Electrode Design. AJBSR 11(2): 147.
- Duncan D, Dinev I (2020) Noninvasive Induction of Muscle Fiber Hypertrophy and Hyperplasia: Effects of High-Intensity Focused Electromagnetic Field Evaluated in an In-Vivo Porcine Model: A Pilot Study. Aesthetic Surgery Journal 40(5): 568-574.
- Kinney BM, Kent DE (2020) MRI and CT Assessment of Abdominal Tissue Composition in Patients After High-Intensity Focused Electromagnetic Therapy Treatments: One-Year Follow-Up. Aesthetic Surgery Journal 40(12): NP686-NP693.
- Palm M (2020) Magnetic Resonance Imaging Evaluation of Changes in Gluteal Muscles After Treatments with the High-Intensity Focused Electromagnetic Procedure. Dermatologic Surgery 47(3): 386-391.
- Katz B, Duncan D (2021) Lifting and Toning of Arms and Calves Using High-Intensity Focused Electromagnetic Field (HIFEM) Procedure Documented by Ultrasound Assessment. J Drugs Dermatol 20(7): 755-759.
- Azparren J, Brandeis J (2021) HIFEM PROCEDURE ENHANCES QUALITY OF LIFE OF ELDERLY MEN WITH POST-PROSTATECTOMY INCONTINENCE. :6.
- Samuels JB, Pezzella A, Berenholz J, Alinsod R (2019) Safety and Efficacy of a Non‐Invasive High‐Intensity Focused Electromagnetic Field (HIFEM) Device for Treatment of Urinary Incontinence and Enhancement of Quality of Life. Lasers Surg Med 51(9): 760-766.
- Duncan D, Chilukuri S, Kent D, Hoffmann K, Tingsong L, et al. (2022) Non-invasive Alternatives for Liposuction. In: Enhanced Liposuction - New Perspectives and Techniques.
- Jacob C, Kent D, Ibrahim O (2021) Efficacy and Safety of Simultaneous Application of HIFEM and Synchronized Radiofrequency for Abdominal Fat Reduction and Muscle Toning: A Multicenter Magnetic Resonance Imaging Evaluation Study. Dermatol Surg 47(7): 969-973.
- DiBernardo B, Goldberg D, Katz B, Mc Coy JD, Chilukuri S, et al. (2022) Augmentation of Gluteal Muscles by HIFEM Procedure is Considerably Enhanced by Simultaneous Radiofrequency Heating. Presented at: 41st Annual Conference of the American Society for Laser Medicine and Surgery. San Diego, CA
- Duncan D (2022) Safety and Efficacy of Simultaneous Application of High intensity Focused Electromagnetic Field and Synchronized Radiofrequency for Non-invasive Fat Reduction and Muscle Toning in Inner Thighs: Magnetic Resonance Imaging Evaluation. JCAD 15(8): 28-32.
- Palm M, Halaas Y, Kinney BM, Goldfarb R (2022) Spot Reduction of Localized Fat Deposits on the Lateral Thighs by Simultaneous Emission of Synchronized Radiofrequency and HIFEM energy: Magnetic Resonance Multicentre Study. Dermatol Surg 49(1): 48-53.
- Jacob C, Weiss R (2017) Upper Arm Toning and Strengthening with Simultaneous Application of HIFEM and Synchronized RF.
- Dimai HP (2017) Use of dual-energy X-ray absorptiometry (DXA) for diagnosis and fracture risk assessment; WHO-criteria, T- and Z-score, and reference databases. Bone 104: 39-43.
- Qin H, Jiao W (2022) Correlation of muscle mass and bone mineral density in the NHANES US general population, 2017–2018. Medicine (Baltimore) 101(39): e30735.
- Aguiar LT, Camargo LBA, Estarlino LD, Teixeira-Salmela LF, Faria CDC de M, et al. (2018) Strength of the lower limb and trunk muscles is associated with gait speed in individuals with sub-acute stroke: a cross-sectional study. Braz J Phys Ther. 22(6): 459-466.
- Kahle N, Tevald MA (2014) Core muscle strengthening’s improvement of balance performance in community-dwelling older adults: a pilot study. J Aging Phys Act 22(1): 65-73.
- Kojima K, Kamai D, Ishitani S, Watanabe S (2017) Availability of the Two-step Test to evaluate balance in frail people in a day care service. J Phys Ther Sci 29(6): 1025-1028.
- Atherton PJ, Smith K (2012) Muscle protein synthesis in response to nutrition and exercise. J Physiol 590(5): 1049-1057.
- Bellamy LM, Joanisse S, Grubb A, Cameron J Mitchell, Bryon R Mc Kay, et al. (2014) The Acute Satellite Cell Response and Skeletal Muscle Hypertrophy following Resistance Training. PLOS ONE 9(10): e109739.
- Halaas Y, Duncan D, Bernardy J, Ondrackova P, Dinev I, et al. (2021) Activation of Skeletal Muscle Satellite Cells by a Device Simultaneously Applying High-Intensity Focused Electromagnetic Technology and Novel RF Technology: Fluorescent Microscopy Facilitated Detection of NCAM/CD56. Aesthetic Surgery Journal 41(7): NP939-NP947.
- Frost HM (2003) Bone’s mechanostat: A 2003 update. Anat Rec A Discov Mol Cell Evol Biol 275(2):1081-1101.
- Järvinen TLN, Kannus P, Sievänen H (1999) Have the DXA-Based Exercise Studies Seriously Underestimated the Effects of Mechanical Loading on Bone? J Bone Miner Res 14(9): 1634-1635.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.