Research Article 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Repeated UVB Radiations Induced Thyroid Alteration and Oxidative Stress in Male Swiss Albino Mice
*Corresponding author: Payal Mahobiya, Endocrinology Lab, Department of Zoology, Dr. Harisingh Gour Vishwavidyalaya Sagar, India
Received: March 15, 2023; Published: April 05, 2023
DOI: 10.34297/AJBSR.2023.18.002471
Abstract
Due to the ozone layer depletion an increased global warming observed, which allows UVB radiation to penetrate the atmosphere and have deadly effects on people. It is thought that UVB short- and long-term radiation are exposed to daily has biological effects on organisms and humans. Concerning effect of radiations, we examine the automatic mechanism by which a specific amount of radiation induces oxidative stress and hyperthyroidism. For these examinations 12 adult male albino mice were divided into two groups: control group, UVB exposed group (2hrs/day). Mice were radiated daily for 15 consecutive days. Specimens of the thyroid gland were processed for hormonal, biochemical, histological and immunofluorescence examinations. A morphometric study and statistical analysis of the findings were performed for different groups. Compared to the control group, the specimens of the UVB exposed mice showed alteration in mean height of the thyroid follicular epithelial cells and caspase-3 expression.
Keywords: Radiation, Thyroid, Hyperthyroidism
Introduction
Radiation from several sources is always present around all living things on Earth. Irradiation of the entire body damages cellular, physiological, and biochemical reactions within living cells. Radiation effects are influenced by various factors, including age, gender, exposure time, and affected tissue. These radiations are emitted and transmitted by various sources and are absorbed by the animal body. UV rays are non-ionizing rays ranging from 200nm to 400nm. UV radiations are found in sunlight and have enough energy to easily penetrate body cells, causing changes in chemical and biological activities. Thyroid hormones regulate metabolic and energy homeostasis, thermogenesis, and the transcription of genes that control cell proliferation and basal metabolic rate to control an organism’s growth throughout [1]. UVB radiation has a wide range of effects on all living things on Earth, including the infliction of cancer [2]. The amount of ozone in the stratosphere determines the amount of UVB radiation that reaches the Earth’s surface. Animals and humans may be harmed by biological reactions to changes in UVB radiation. The United Nations has conducted regular assessments of the effects of UVB radiation on people and the environment [3].
Changes in body weight, thyroid weight, hormonal alterations, biochemical changes and expression of antibodies were studied in this study. Furthermore, radiations were used for the first time to demonstrate the direct free radical scavenging potential of this flavonoid in thyroid tissue.
Methods and Materials
Chemicals
Analytical-grade chemicals and reagents were utilized. In Mumbai, India, Himedia Laboratories Pvt. Ltd. sold naringin (C27H32O14). NBT, NADPH, Methionine, Reduced Glutathione, and the remaining chemicals were acquired from Central Drug House (P) Ltd, New Delhi, India. Thiobarbituric Acid, Hematoxylin, Eosin, and the other chemicals were acquired from Himedia, India, Caspase-3 antibodies were purchased from science emporium.
UVB Irradiations
The UVB light, produced in Germany with a wavelength of 280 nm, was used to expose the Swiss albino mice. For 15 days, the irradiance was set at two hours each day [4].
Study Animals
We bought male Swiss albino mice from Mhow, India’s College of Veterinary Sciences and Animal Husbandry, measuring 22-28 grammes. Animals were acclimated to the housing at a temperature of 20 to 25 °C, relative humidity of 50 to 55 %, and a cycle of 12 hours of light and 12 hours of darkness.
Ethical Statement
Institutional animal ethics committee, Dr Harisingh Gour Vishwavidyalaya, Sagar (M.P.) (A Central University) (Approval No.:379/CPCSEA/IAEC-2021/004), at Department of Pharmaceutical Sciences, provided its ethical endorsement. International standards were followed in the handling and usage of laboratory animals.
Study Test Design
The animals were randomly divided into four groups with six mice in each group.
a) Group 1: The animals received standard food and water ad libitum for 15 days (control group). b) Group 2: The animals was exposed to UVB irradiation at 280nm for two hours every day for 15 days./
Throughout the therapy, body weight and daily food and drink intake were noted. Animals were starved overnight and then killed by cervical decapitation after the study period. After the connective tissues around the thyroid gland were removed, the thyroid gland was removed through a transverse abdominal incision and weighed.
Sample Collection
After being extracted, whole blood samples were put in evacuated tubes containing an EDTA anticoagulant solution. The plasma obtained after centrifuging the second tube at 2200rpm for 15 minutes was stored at -20°C until the levels of the thyroid hormones were determined.
Measurement of Body Weight and Thyroid Weight
Body weight was recorded on the beginning, sixth, eleventh, and fifteenth days of the trial. Thyroid weight was assessed following exsanguinations. An electronic weighing balance (Sartorius BP 210S) was used to measure thyroid weight.
Tissue Homogenization
The thyroid gland was removed for biochemical testing, cleaned with PBS solution, and kept at -20oC until needed. The thyroid gland extract was made with homogenization in 0.02 M Tris-Cl (pH 7.4). After centrifuging homogenate tissue at 12500 rpm for 30 min at 4°C, the supernatant was kept at -20°C for the biochemical tests.
Hormonal Analysis
Using an enzyme-linked immunosorbent test kit given by The Calbiotech Inc. (California, USA), serum samples were examined for T3, T4 and TSH levels [5].
Analysis of Enzymatic and Non-Enzymatic Assays
The remaining part of testis was used for biochemical analysis to measure Lipid peroxidation [6], Catalase [7], Superoxide dismutase [8], GSH [9], Hydrogen peroxide (Sinha, 1972), Glutathione Reductase [10] and Nitric oxide [11] levels in all the above groups.
Histopathological Analysis
Through the heart, 0.02M phosphate-buffered saline (PBS) and 4% PFA were used to perform whole-body perfusion (Stefanini et al., 1967). After being removed, the thyroid gland was placed in the fixative for two days. For haematoxylin and eosin staining, fixed tissues were treated, dried, and cut into 5μm slices from paraffinembedded thyroid. 10X and 40X magnification microscope analyses of images.
Immunofluorescence
Caspase-3 immunoreactivity was detected in the thyroid by immunofluorescence by using method of Niranjan & Srivastava, 2019 with slight modifications. Paraffin sections of thyroid samples were deparaffinized and rehydrated. Antigens were retrieved by incubating the sections in a microwave oven in sodium citrate buffer (10 mM; pH 6.0) for 15 min. Sections were brought to room temperature and rinsed with PBS. Sections were incubated with primary antibodies overnight at 4˚C. Antibodies against caspase-3 (cat. no. BS-0081R) was used. Slides were incubated with the secondary antibody FITC for 60 min at 4˚C. The sections were counterstained using DAPI for 4 minutes at RT, then slides were mounted by glycerine-based media. The immunofluorescence images were taken using EVOS 5000 Invitrogen fluorescent microscope. For semiquantitative analysis of caspase-3, the immunoreactive cells were analysed with Image J software.
Statistical Analysis
All statistical data analyses employed one-way ANOVA (analysis of variance), and the results are presented as mean SE (standard error). The irradiated and control groups were compared using Dunnett’s test. The threshold for significance was established at, correspondingly, *p<0.05, **p<0.01, and ***p<0.001.
Result
Body and Thyroid Weight
The body weight measured over the study period was analysed using the body weight mean. Body weight differences between the UVB-irradiated group and the control group were found to be statistically significant (p<0.01). In comparison to the control group, the thyroid weight in the UVB-irradiated groups reduced considerably (p<0.01), according to the findings (Figure 1).
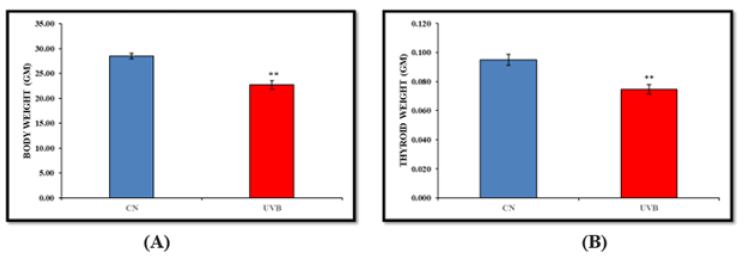
Figure 1:Effect of UVB irradiation on body weight (A) and thyroid weight (B). Values are presented as mean ± SE (n=6). Significant difference from control group (*p < 0.05, **p<0.01).
Effects of UVB Irradiation on Thyroid Hormones
Thyroid hormone levels are noticeably higher in the UVBirradiated group compared to the control group. T3, T4 levels significantly increased in the UVB-irradiated group compared to the control group, while TSH levels significantly decreased (Figure 2).
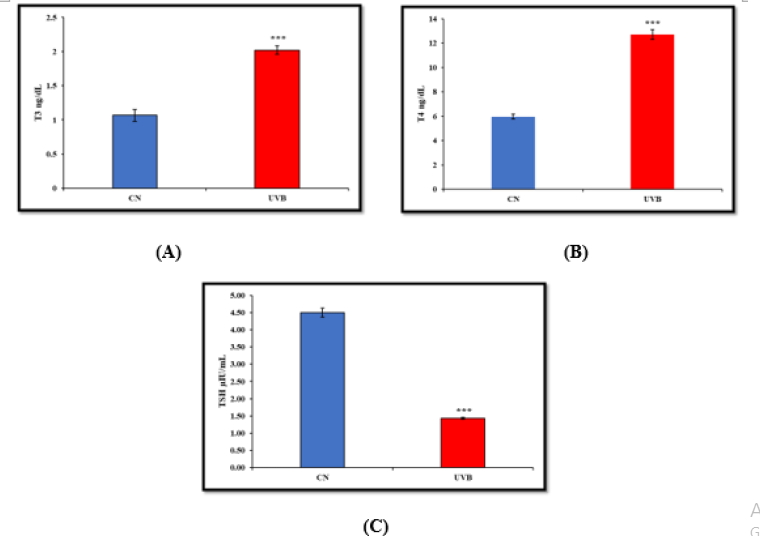
Figure 2:Effect of UVB irradiation on Hormonal Parameters i.e., (A) T3 (B) T4 (C) TSH (D) FT3 (E) FT4. Values are presented as mean ± SE (n=6). Significant difference from control group (*p < 0.05, **p<0.01, ***p<0.001).
Effect of UVB Irradiation on Enzymatic and Non- Enzymatic Assays
a) LPO Estimation-Thyroid MDA levels in the UVBirradiated group were considerably higher than those in the control group (Figure 3A).
b) SOD EstimationIn comparison to the control group, the UVB-irradiated group’s SOD activity was significantly higher (Figure 3B).
c) Catalase ActivityThe UVB-irradiated group had significantly more catalase activity than the control group (Figure 3C).
d) H2O2 ActivityWhen compared to the control group, the H2O2 activity seen in the UVB-irradiated group was significantly higher (Figure 3D).
e) NO Activity-NO activity was substantially higher in the UVB-irradiated group than it was in the control group (Figure 3E).
f) GR ActivityIn comparison to the control group, the GR activity was significantly higher in the UVB-irradiated group (Figure 3F).
g) GSH ActivityWhen compared to the control group, the GSH activity seen in the UVB-irradiated group was significantly lower (Figure 3G).
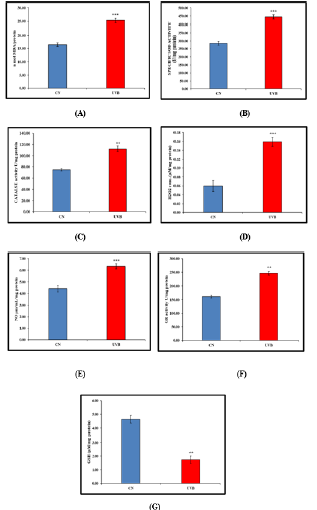
Figure 3:Effect of UVB irradiation on Enzymatic and Non-Enzymatic parameters i.e., (A) LPO (B) SOD (C) CAT (D) H2O2 (E) NO (F) GR (G) GSH. Values are presented as mean ± SE (n=6). Significant difference from control group (*p < 0.05, **p<0.01, ***p<0.001). LPO (Lipid per Oxidation), SOD (Super oxide Dismutase), CAT (Catalase), H2O2 (Hydrogen per Oxide), NO (Nitric Oxide), GR (Glutathione reductase), GSH (reduced glutathione)
Effect Of UVB Irradiation on Histology of Thyroid Gland
The thyroid follicular structure was shown in the control group to have a normal histological appearance, but the UVB-irradiated group showed disruption of the follicular structure (Figure 4).
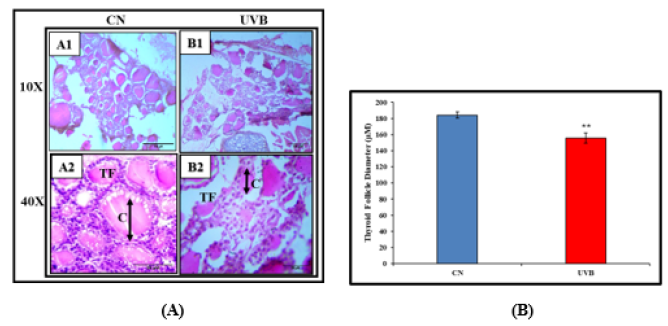
Figure 4:Effect of UVB Radiation on Thyroid histology. TS of the thyroid gland of the control group (A1&A2), UVB exposed group (B1&B2). (A) photograph showing H &E staining (100X and 400X), (B) Graph of the diameter of thyroid follicles, and the values represent Mean±SE (n=6). Colloidal fluid (C) and Thyroid follicular (TF).
Immunofluorescence Expression of Caspase-3 In Thyroid Gland
Caspases are a group of genes that play a role in maintaining homeostasis by regulating cell death and inflammation [12]. The fluorescence expression of Caspase-3 was seen to be increased in case on UVB irradiated group as compared to control (Figure 5).
Discussion
In order to provide hopeful evidence for a link between exposure to EMF and an increased prevalence of health risks, possible impacts of EMF on biological systems were widely examined [13]. Numerous in-depth studies have been conducted over the last few decades to examine the potential biological effects of electromagnetic fields (EMF) on various human systems. The endocrine system, namely the pineal gland, has received the most attention out of all the researched systems [14]. Effects from low radiation doses are more challenging to forecast and analyse. There is no limit to how efficient adaptive mechanisms promote cellular protection. As a result, it is impossible to draw a firm judgement concerning exposure to low radiation levels [15,16]. Due to the sensitivity of the thyroid gland to EMFs, this exposure led to morphological modifications and a decrease in blood T4 and T3 levels. These alterations remained over the whole trial, proving that normal thyroid function takes more time to rebound after exposure to EMFs.
Loss of body weight is typically linked to hyperthyroidism [17]. Our findings were consistent with previous studies in that UVB irradiation resulted in considerable thyroid and body weight loss [18,19]. The thyroid gland is impacted by UV radiation, and long-term radiation exposure predominantly targets many genes, proteins, and lipids and leads to cancer [20]. We discovered that UVB radiation-induced oxidative stress produced free radicals and induced hyperthyroidism. Hormonal parameters such as T3, T4 were all considerably higher in hyperthyroidism, whereas TSH was much lower. By causing damage to crucial components, including body protein and thyroid tissue, UVB irradiation was shown to dramatically diminish the body weight and thyroid weight of the experimental mice. In vivo cellular oxidative stress is thought to be modulated by any fluctuation in circulatory thyroid hormones. Reactive oxygen species are produced due to increased mitochondrial respiration, which is thought to be the primary mechanism of this physiological/pathological alteration [21].
We observed that Swiss albino male mouse thyroid glands exposed to hyperthyroidism experienced a heightened rise in ROS. Other investigations have revealed elevated levels of oxidative stress in several organs, including the testis, skeletal muscle, heart, pancreas, and brain, which is mediated by the hyperthyroid condition [22,23]. Physiological levels of ROS are created during typical cell metabolism. However, excessive ROS production can result in cell apoptosis, necrosis, or autophagy in several pathological circumstances, including inflammatory bowel disease, diabetes, cancer, or obesity (Niture et al., 2014). Organisms have complete, integrated endogenous enzymatic repair mechanisms to deal with ROS damage. Important non-enzymatic antioxidants include glutathione (GSH), vitamin E, vitamin C, h-carotene, and uric acid, which are either ingested with food or produced endogenously. The endogenous enzymatic antioxidants are represented by Cu2+, Zn2+ and MnSODs, catalase, and GPx [24]. The decrease in antioxidant capacity in hyperthyroid patients is most likely due to increased free radical production. Increased enzyme activity in hyperthyroidism patients is likely due to increased ROS production [25]. In the mouse thyroid, exposure to UVB significantly increased the level of lipid peroxidation. Earlier research on lipid peroxidation showed a similar impact (Jagetia et al., 2003,26). Lipid peroxidation is inversely related to oxidative stress, reducing some defensive systems effectiveness.
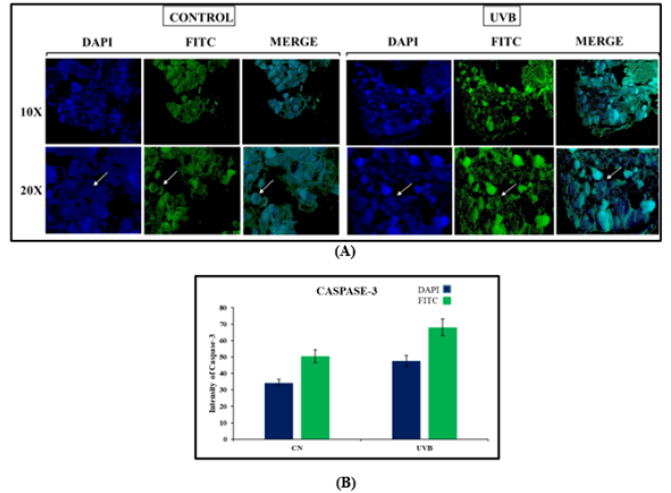
Figure 5:Immunofluorescence expression of Caspase-3. TS of the thyroid gland of the control group, UVB exposed group. (A) Photograph showing DAPI staining and arrow indicate the expression of Caspase-3 antibody in thyroid gland (100X and 200X), (B) Graph of the fluorescence intensity in thyroid follicles, and the values represent Mean±SE (n=6).
Whole-body UVB exposure reduces an organism’s cellular potential for general antioxidant defence and depletes known antioxidants like GSH [24]. Mice exposed to UVB radiations exhibited significantly reduced thyroid GSH activity. It has been observed that GSH depletion enhances lipid peroxidation since it is known to impair glutathione peroxidase activity both in vitro and in vivo. The current study shows a similar relationship between the decrease in GSH and the rise in lipid peroxidation. According to our study, thyroid Glutathione Reductase (GR) activity significantly increased due to UVB exposure. According to some authors, the thyroid hormone impact directly contributes to the enhanced activity of glutathione reductase, and administration of peroxidative products to rats has been shown to dramatically promote the activity of several detoxification enzymes [27,28]. Catalase, hydrogen peroxide, and superoxide dismutase activity were significantly increased throughout our research. According to Messiah et al., hyperthyroid rats hearts and erythrocytes had greater SOD, CAT, and H2O2 activity levels [29]. The expression of antioxidant enzymes may be impacted by hyperthyroidism depending on the cell type, mitochondrial activity, and ROS concentration inside the cell [30].
The alterations in thyroid gland function in the exposed group were further supported by the histological examination of the thyroid follicles in the current investigation, which exhibited glaring light microscopic and ultrastructural abnormalities. In many instances, these follicles were disorganised and lost. While some follicles seemed to have involuted walls, others had fragmented follicular walls. While other cells had vacuolated cytoplasm, the follicular cells lost their epithelial covering in the lumina. Additionally, some follicular cells possessed darkly coloured nuclei, and interfollicular septa had cellular infiltration. The deficiency of TSH’s glandstimulating actions and hypoactivity might be employed to explain the thyroid follicles’ histological alterations. Morphometrical data supported the changes in epithelial height.
Immunofluorescence revealed increased amounts of cells positive for Caspase after UVB exposure. In the present study, the enhanced Caspase-3 expression was detected in thyroid follicular cells in the UVB exposed group. To date, little information exists on the effect of UVB radiation on caspase-3 expression. In the present study, UVB radiation induced significant increase in caspase-3 expression indicating UVB provokes apoptosis in the thyroid follicular cells. Apoptosis is a physiological process of selected cell deletion. As an antagonist of cell proliferation, apoptosis contributes to keeping the cell number in thyroid tissue and helps to remove superfluous and damaged cells, but excessive apoptosis could cause destruction of thyroid tissue [31]. In a similar study in China to explore the effects of expressions of caspase-3 in mice testes at different concentrations and time of lead acetate, it increased the expressions of caspase-3, which induces apoptosis of germ cells [32-40]. The occurrence of thyroid cell apoptosis and the expression of caspase-3 in the adult male mice following UVB radiation administration were investigated. Compared with the control group, the protein levels of caspase-3 were significantly higher in UVB exposed group. Furthermore, UVB exposure led to inflammation in the thyroid follicular cell apoptosis, as demonstrated in the DAPI-FITC assay. These results suggested that thyroid follicular cells are affected by UVB radiation.
Conclusion
Our findings conclusively demonstrate that UVB irradiationinduced hyperthyroidism caused an inclined in thyroid hormone levels. UVB exposure has significant negative impacts on thyroid function and weight. The results also showing the negative impact over the antioxidant mechanisms. Our study’s result is novel to demonstrate how repeated UVB radiations induced hyperthyroidism and caused deformation of thyroid follicles and the alterations in the expression of caspase-3 antibody.
Author’s Contributions
Payal Mahobiya designed the experiment plan. Shashank Shakyawal managed the experimental animals, performed the treatment and completed data analysis and wrote the manuscript. Both the authors read and approved the final manuscript.
Acknowledgment
This research work was financially assisted by UGC NON-NET FELLOWSHIP for carrying out this research work successfully. The authors wish to thank the Department of Zoology, School of Biological Sciences, Dr. Harisingh Gour Vishwavidyalaya, Sagar, M.P., India, for providing infrastructure facilities.
Conflict of Interest
The authors declare that there is no conflict of interest.
References
- Schriks M, Roessig JM, Murk AJ, Furlow JD (2007) Thyroid hormone receptor isoform selectivity of thyroid hormone disrupting compounds quantified with an in vitro reporter gene assay. Environ Toxicol Pharmacol 23(3): 302-307.
- Neugart S, Schreiner M (2018) UVB and UVA as eustressors in horticultural and agricultural crops. Scientia Horticulturae 234: 370-381.
- Andrady A, Aucamp P, Bais A, Ballare C, Bjorn, et al. (2005) Environmental effects of ozone depletion and its interactions with climate change: progress report, 2004. Photochem Photobiol Sci 4(2): 177-184.
- Akindele AJ, Adeneye AA, Salau OS, Sofidiya MO, Benebo AS, et al. (2014) Dose and time-dependent sub-chronic toxicity study of hydroethanolic leaf extract of Flabellaria paniculata Cav.(Malpighiaceae) in rodents. Front Pharmacol 23(5): 78.
- Kumar A, Prakash A, Dogra S (2010) Naringin alleviates cognitive impairment, mitochondrial dysfunction and oxidative stress induced by D-galactose in mice. Food Chem Toxicol 48(2): 626-632.
- Placer ZA, Cushman LL, Johnson BC (1996) Estimation of lipid peroxidation, malindialdehyde in biochemical system. Anal Biochem 16(2): 359-364.
- Bergmeyer HU (1983) Methods of enzymatic analysis, Weinheim, Germany: Verlag Chemie 3(3): 273-286.
- Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44(1): 276-287.
- Sedlak J, Lindsay RH (1968) Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman's reagent. Anal Biochem 25(1): 192-205.
- Carlberg I, Mannervik B (1975) Purification and characterization of the flavoenzyme glutathione reductase from rat liver. J Biol Chem 250(14): 5475-5480.
- Griess P (1879) Bemerkungen zu der Abhandlung der HH. Weselsky und Benedikt „Ueber einige Azoverbindungen” ︁. Berichte der deutschen chemischen Gesellschaft 12(1): 426-428.
- Mcllwain DR, Berger T, Mak TW (2013) Caspase function in cell death and disease. Cold Spring Harb Perspect Biol 5(4): a008656.
- Wolf FI, Torsello A, Tedesco B, Fasanella S, Boninsegna A, et al. (2005) 50-Hz extremely low frequency electromagnetic fields enhance cell proliferation and DNA damage: possible involvement of a redox mechanism. Biochim Biophys Acta 1743(1-2): 120-129.
- Graham C, Cook MR, Sastre A, Gerkovich MM, Kavet R, et al. (2000) Cardiac autonomic control mechanisms in power-frequency magnetic fields: a multistudy analysis. Environ Health Perspect 108(8): 737-742.
- Daino K, Nishimura M, Imaoka T, Takabatake M, Morioka T, et al. (2018) Epigenetic dysregulation of key developmental genes in radiation‐induced rat mammary carcinomas. Int J Cancer 143(2): 343-354.
- Sharma S, Singla N, Chadha VD, Dhawan DK (2019) A concept of radiation hormesis: stimulation of antioxidant machinery in rats by low dose ionizing radiation. Hell J Nucl Med 22(1): 43-48.
- Ganong WF (2005) Review of Medical Physiology. 22 [sup] nd ed. San Francisco: Lange 444.
- Venditti P, Meo SD (2006) Thyroid hormone-induced oxidative stress. Cell Mol Life Sci 63(4): 414-434.
- Petrulea M, Muresan A, Duncea I (2012) Oxidative stress and antioxidant status in hypo-and hyperthyroidism. Antioxidant enzyme 8: 197-236.
- Sinnott B, Ron E, Schneider AB (2010) Exposing the thyroid to radiation: a review of its current extent, risks, and implications. Endocr Rev 31(5): 756-773.
- Guerrero A, Pamplona R, Portero Otín M, Barja G, López Torres M, et al. (1999) Effect of thyroid status on lipid composition and peroxidation in the mouse liver. Free Radic Biol Med 26(1-2): 73-80.
- Elnakish MT, Ahmed AA, Mohler PJ, Janssen PM (2015) Role of oxidative stress in thyroid hormone-induced cardiomyocyte hypertrophy and associated cardiac dysfunction: an undisclosed story. Oxid Med Cell Longev 2015: 854265.
- Rao G, Verma R, Mukherjee A, Haldar C, Agrawal NK (2016) Melatonin alleviates hyperthyroidism induced oxidative stress and neuronal cell death in hippocampus of aged female golden hamster, Mesocricetus auratus. Exp Gerontol 82: 125-130.
- Karbownik M, Reiter RJ (2000) Antioxidative effects of melatonin in protection against cellular damage caused by ionizing radiation (44547). Proc Soc Exp Biol Med 225(1): 9-22.
- Naazeri S, Rostamian M, Hedayati M (2014) Impact of thyroid dysfunction on antioxidant capacity, superoxide dismutase and catalase activity.
- Koc M, Taysi S, Emin Buyukokuroglu M, Bakan N (2003) The effect of melatonin against oxidative damage during total-body irradiation in rats. Radiat Res160(2): 251-255.
- Sandstrom BE, Carlsson J, Marklund SL (1987) Variations among cultured cells in glutathione peroxidase activity in response to selenite supplementation. Biochim Biophys Acta 929(2): 148-153.
- Morini P, Casalino E, Sblano C, Landriscina C (1991) The response of rat liver lipid peroxidation, antioxidant enzyme activities and glutathione concentration to the thyroid hormone. Int J Biochem 23(10): 1025-1030.
- Messarah M, Saoudi M, Boumendjel A, Boulakoud MS, El Feki A, et al. (2011) Oxidative stress induced by thyroid dysfunction in rat erythrocytes and heart. Environ Toxicol Pharmacol 31(1): 33-41.
- Costilla M, Macri Delbono R, Klecha A, Cremaschi GA, Barreiro Arcos ML, et al. (2019) Oxidative stress produced by hyperthyroidism status induces the antioxidant enzyme transcription through the activation of the Nrf-2 factor in lymphoid tissues of Balb/c mice. Oxid Med Cell Longev: 7471890.
- Richburg JH (2000) The relevance of spontaneous-and chemically-induced alterations in testicular germ cell apoptosis to toxicology. Toxicol Lett 112-113: 79-86.
- Yan Zhang, Chunhong Wang, Duanlian Zhang (2006) Effects of lead on expressions of TGFβ1 and Caspase-3 in mice testes. Chinese Journal of Public Health 22(7): 869-870.
- Castilho RF, Kowaltowski AJ, Vercesi AE, (1998) 3, 5, 3′-triiodothyronine induces mitochondrial permeability transition mediated by reactive oxygen species and membrane protein thiol oxidation. Arch Biochem Biophys 354(1): 151-157.
- Fauré M, Lissi EA, Videla LA (1991) Evaluation of the antioxidant properties of thyroid hormones and propylthiouracil in the brain-homogenate autoxidation system and in the free radical-mediated oxidation of erythrocyte membranes. Chem Biol Interact 77(2): 173-185.
- Koiri RK, Trigun SK, Mishra L, Pandey K, Dixit D, et al. (2009). Regression of Dalton’s lymphoma in vivo via decline in lactate dehydrogenase and induction of apoptosis by a ruthenium (II)-complex containing 4-carboxy N-ethylbenzamide as ligand. Invest New Drugs 27(6): 503-516.
- Landriscina C, Gnoni GV, Quagliariello E (1976) Effect of thyroid hormones on microsomal fatty acid chain elongation synthesis in rat liver. Eur J Biochem71(1): 135-143.
- Niranjan MK, Srivastava R (2019) Expression of estrogen receptor alpha in developing brain, ovary and shell gland of Gallus gallus domesticus: Impact of stress and estrogen. Steroids 146: 21-33.
- Niture SK, Khatri R, Jaiswal AK (2014) Regulation of Nrf2—an update. Free Radical Biology and Medicine 66: 36-44.
- Srivastava R, Chaturvedi CM (2012) Age, photoperiod and estrogen dependent variations in the shell gland and the expression of AVT in the ovary of Japanese quail. Steroids 77(6): 578-588.
- United Nations Environment Programme EE, Assessment P (2004) Environmental effects of ozone depletion and its interactions with climate change: Progress Report 2003. Photochemical & photobiological sciences: Official journal of the European Photochemistry Association and the European Society for Photobiology 3(1): 1.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.