Research Article 
 Creative Commons, CC-BY
Creative Commons, CC-BY
The Anti-Aging Effects of Marine Peptides on The Improvement of Facial Skin: A Mechanistic Study and A Pilot-Scale Study
*Corresponding author:Danial Steve, Ph.D., NutraPio Health Industrial Co., Ltd., North Carolina, USA.
Received:March 06, 2023;Published:March 13, 2023
DOI: 10.34297/AJBSR.2023.18.002430
Abstract
In recent years, various marine derived products have been becoming prevalent and popular in the beauty from within market due to solid scientific research that proved their potential to improve the skin conditions and reverse facial aging. In our study, a proprietary and patented combination consisting of 5 marine peptides was investigated regarding its benefits against facial antiaging. The human dermal fibroblast tests indicated mRNA expressions of AQP3, and type III collagen were significantly enhanced by the composition compared to each individual ingredient. The pilot-scale study in which 10 individuals orally supplemented with the composition for 28 days had increased contents of both type I and type III collagens while the ratio of type I to type III collagens decreased to a level comparable to that of young adolescents. This should be the first research demonstrating the efficacies of marine peptides combination on facial skin improvement from the perspective of its ability to reverse the ratio of type I to type III collagen, which definitely provided a valuable target for the futuristic intervention.
Introduction
The fragmentation of the dermal collagen matrix occurs in aged skins due to internal and external factors [1]. The fragmented collagen originally produced and organized in fibroblasts cannot be reconnected because of the lost mechanical tension for fragmented fibroblasts [2]. Without the stretch of the collagen fibers, the fibroblasts cannot only produce sufficient amounts of collagen but also meanwhile generate a lot of collagen-degrading enzymes [3]. Therefore, it is essential to stimulate the damaged fibroblasts and their proliferation as an effective strategy to prevent further skin deterioration. Various clinical trials have shown that orally administered marine peptides can be easily absorbed to stimulate the fibroblasts for synthesizing collagen [4-5]. The structure of prolyl-hydroxyproline functioned as chemotactic stimuli for fibroblasts in vivo and attracted these cells to the damaged sites and tissues for restoration [6]. Moreover, prolyl-hydroxyproline could also enhance proliferation human dermal fibroblasts as well as hyaluronic acid synthesis in a vitro study [7].
On the other hand, it should not be ignored that the transportation of water and glycerol in the basal layer keratinocytes of epidermis in normal skin cannot be finished without a membrane transporter called Aquaporin-3 (AQP3). It was found AQP3-knockout mice have reduced stratum corneum water content [8,9]. Here we also hypothesize the supplementation of marine peptides could activate the expression of AQP3 and thus facilitate the hydration of epidermis. Type III collagen is a rare and precious collagen on the market at present. It exists in the superficial dermis as small fibers. The type III collagen content is at its peak for fetus, so the softness, elasticity and moisture of the baby’s skin are unmatched compared to those of the adults. The baby’s skin can also renew quickly without leaving scars after injury due to high amount of type III collagen, making it known as “baby collagen” [10].
Unfortunately, it dwindles continuously from adults to senile individuals, resulting in the inability of the skin to repair scars, sharply deteriorated elasticity and collapsed skin [11]. In our study, we investigated the effects of the combination of 5 marine peptides on ameliorating and reversing skin aging. The mechanisms of relative gene expressions in human dermal fibroblasts were also investigated. Unprecedentedly, a pilot scale study involving 10 healthy subjects was conducted for the observation of collagen I/III change after its oral administration for 28 days.
Materials and Methods
Testing Product
The composition was a proprietary and patented peptide matrix consisting of collagen peptide, elastin peptide, salmon hydrolyzed peptide, proteoglycan and salmon seed peptide. The patented composition was owned by provided by ActiveGene Institute of Life Sciences Co., Ltd. The testing products consisted of composition and the fillers.
Effect on Human Dermal Fibroblasts on Vitro Test
The solution of the composition was prepared at 10%, 5%, 0.5%, 0.1% solution with sterile PBS. The Human dermal fibroblast cells were inoculated (0.4~1.6 x 105 cells/well) into the 6-well cell culture plate with collagen peptides. The RNA extraction and reverse transcription cDNA were operated by the RNA extraction kit and PrimeScript™ RT Master Mix. The primer verification of RT-PCR and RT-PCR tests were conducted by TB green ®Premix ExTaq™ II (Takara). Primer sequence AQP3-3F and AQP3-3R are GCAGCCTGTCCATCTGTG and ACCCTACTTCCCAAAAGCC respectively. The effects were calculated as an increasing percentage based on blanks.
Effects on The Expression of Type III Collagen on Vitro Test
When the fibroblast planking rate reached 75%~85%, the culture solution in the culture bottle was discarded, 1ml of 0.25% trypsin was added for digestion at 37oC for 3-5 minutes. 5mL of DMEM culture solution containing serum was then added to stop digestion until 80% of the cells were in suspension observed under inverted microscope. Then the cells in the culture bottle were collected and centrifuged for 1 minute at 1000 rpm/min. The cell suspension was inoculated into a 6-well plate with 2mL of liquid per well. Then it was put in the incubator (37oC, 5% CO2, 95% RH) for 24h±2h. After incubation and culture, the cell supernatant was collected, and ELISA kit was used to detect the content of type III collagen in the supernatant. The effects were calculated as an increasing percentage based on blanks.
Effects on the expression of type I and type III collagen before and after the oral administration for 28 days. Normal skin samples were provided by volunteers (n=30; of which male =16 and female =14) who signed the agreement that clearly states the benefits and risks by participating such a study. The product was orally supplemented to those individuals for 28 days. Before and after the trial, the tissue samples were fixed in 4% formaldehyde for 12h, dehydrated, and olefin-embedded prior to sectioning at 3μm. Type I and III collagen were assessed in fixed sections using polyclonal antibodies and the streptavidin–biotin–peroxidase complex (SABC) kit.
Results
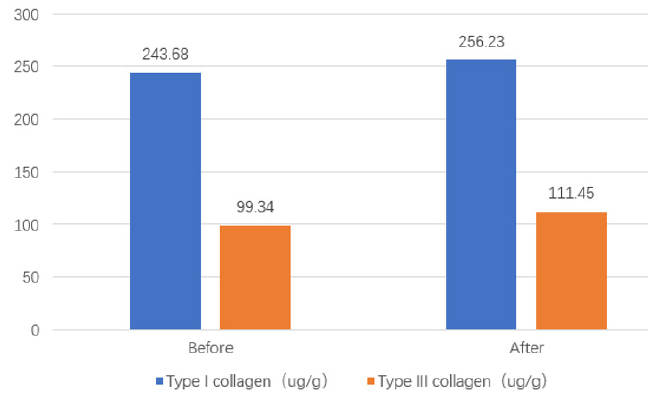
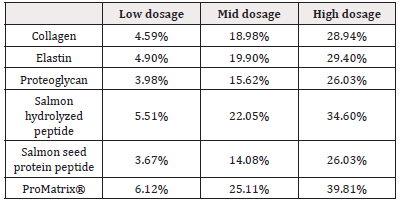
The gene expressions of AQP3 and type III collagen were significantly enhanced when the composition was co-cultured with human dermal fibroblasts (Table 1&2). The huge improvement was not observed when they were co-cultured with each individual ingredient. Before and after the test, both the content of type I and type III collagen were improved from 243.68μg/g and 99.34μg/g to 256.23μg/g and 111.45 μg/g respectively. The ratio of type I to type III collagen decreased from 2.45 to 2.30 (Figure 1).
Discussion
There have been many animal and clinical research advances in the protection and improvement of skin by marine derived peptides regarding skin elasticity, hydration, trans-epidermal water loss, photo aging, atopic skin and so on [12-13]. In our study, we observed the efficacies of a proprietary combination consisting of 5 marine peptide ingredients in facial anti-aging. Our vitro tests of human dermal fibroblast indicated mRNA expressions of AQP3 and type III collagen were significantly stimulated by the composition while individual ingredients did not generate such strong benefits. Marine peptides can repair and increase the activity of fibroblasts and thus increase the production of hyaluronic acid and collagen fibrils [14]. In particular small molecular tripeptides and di-peptides could stimulate the activity of fibroblasts with lost mechanical strength [15-16].
Accumulating evidence indicates that the water-, glycerol- and hydrogen peroxide-transporting channel aquaporin-3 (AQP3) expressed in plasm membranes is crucial in keratinocyte function while its abnormalities caused several human skin diseases [17]. Hydration of the stratum corneum is an important determinant of skin appearance and highly depends on AQP3. Exposure of mice to high humidity or skin occlusion increased stratum corneum hydration in the wild type, but not in AQP3-null mice [18]. It was also found the magnitude of water moving into stratum corneum to off-set evaporative water was much lower than that with AQP3 [19]. In our research, the increased mRNA expression of AQP3 probably indicates the improvement in the stratum corneum hydration and trans-epidermal water loss. Both our vitro study and literature show certain structures in the composition should be similar to epidermal growth factor; however, this remains to be investigated in the futuristic studies [20].
The proportion of type I collagen to type III collagen in adolescent skin is about 2.27 [21]. With the growth of age, this proportion gradually increases, resulting from the fact that type I collagen and type III collagen decrease at the same time while type III collagen decreases faster. The technology of the invention cannot only slow down the decline rate of type I collagen and type III collagen but also reverse the proportion of type I collagen and type III collagen to the level for teenagers (less than 19 years old). Our research should be the first research demonstrating the efficacies of marine peptides combination on facial skin improvement given it reverses the ratio of type I to type III collagen and provides a valuable target for the futuristic intervention.
References
- Haratake A, Watase D, Fujita T, Setoguchi S, Matsunaga K, et al. (2015) Effects of oral administration of collagen peptides on skin collagen content and its underlying mechanism using a newly developed low collagen skin mice model. Journal of Functional Foods 16: 174-182.
- Fisher GJ, Varani J, Voorhees JJ (2008) Looking older: fibroblast collapse and therapeutic implications. Archives of dermatology144(5): 666-672.
- Varani J, Dame MK, Rittie L, Fligiel SE, Kang S, et al. (2006) Decreased collagen production in chronologically aged skin: roles of age-dependent alteration in fibroblast function and defective mechanical stimulation. The American journal of pathology, 168(6): 1861-1868.
- Proksch E, Schunck M, Zague V, Segger D, Degwert J, et al. (2014) Oral intake of specific bioactive collagen peptides reduces skin wrinkles and increases dermal matrix synthesis. Skin pharmacology and physiology 27(3): 113-119.
- Proksch E, Segger D, Degwert J, Schunck M, Zague V, et al. (2014) Oral supplementation of specific collagen peptides has beneficial effects on human skin physiology: a double-blind, placebo-controlled study. Skin pharmacology and physiology 27(1): 47-55.
- Postlethwaite AE, Seyer JM, Kang AH (1978) Chemotactic attraction of human fibroblasts to type I, II, and III collagens and collagen-derived peptides. Proceedings of the National Academy of Sciences of the United States of America 75(2): 871-875.
- Ohara H, Ichikawa S, Matsumoto H, Akiyama M, Fujimoto N, et al. (2010) Collagen-derived dipeptide, proline-hydroxyproline, stimulates cell proliferation and hyaluronic acid synthesis in cultured human dermal fibroblasts. Journal of dermatology 37(4): 330-338.
- Hara-Chikuma M, Verkman AS (2008) Roles of aquaporin-3 in the epidermis. The Journal of investigative dermatology 128(9): 2145-2151.
- Choi SY, Ko EJ, Lee YH, Kim BG, Shin HJ, et al. (2014) Effects of collagen tripeptide supplement on skin properties: a prospective, randomized, controlled study. Journal of cosmetic and laser therapy: official publication of the European Society for Laser Dermatology 16(3): 132-137.
- Jensen BA, Reimann I, Fredensborg N (1986) Collagen type III predominance in newborns with congenital dislocation of the hip. Acta orthopaedica Scandinavica 57(4): 362-365.
- Volk SW, Wang Y, Mauldin EA, Liechty KW, Adams SL, et al. (2011) Diminished type III collagen promotes myofibroblast differentiation and increases scar deposition in cutaneous wound healing. Cells, tissues organs 194(1): 25-37.
- Pyun HB, Kim M, Park J, Sakai Y, Numata N, et al. (2012) Effects of Collagen Tripeptide Supplement on Photoaging and Epidermal Skin Barrier in UVB-exposed Hairless Mice. Prev Nutr Food Sc 17(4): 245-253.
- Berardesca E, Abril E, Serio M, Cameli N (2009) Effects of topical gluco-oligosaccharide and collagen tripeptide F in the treatment of sensitive atopic skin. Int J Cosmet Sci 31(4): 271-277.
- Fisher GJ, Varani J, Voorhees JJ (2008) Looking Older. Archives of Dermatology 144(5).
- Sontakke SB, Jung J, Piao Z, Chung HJ (2016) Orally Available Collagen Tripeptide: Enzymatic Stability, Intestinal Permeability, and Absorption of Gly-Pro-Hyp and Pro-Hyp. J Agric Food Chem 64(38): 7127-7133.
- Cadet J, Douki T, Ravanat JL (2015) Oxidatively generated damage to cellular DNA by UVB and UVA radiation. Photochem Photobiol 91(1): 140-155.
- Bollag WB, Aitkens L, White J, Hyndman KA (2020) Aquaporin-3 in the epidermis: more than skin deep. Am J Physiol Cell Physiol 318(6): C1144-C1153.
- Hara M, Ma T, Verkman AS (2002) Selectively reduced glycerol in skin of aquaporin-3-deficient mice may account for impaired skin hydration, elasticity, and barrier recovery. J Biol Chem 277(48): 46616-46621.
- Hara M, Verkman AS (2003) Glycerol replacement corrects defective skin hydration, elasticity, and barrier function in aquaporin-3-deficient mice. Proc Natl Acad Sci USA 100(12): 7360-7365.
- Cao C, Sun Y, Healey S, Bi Z, Hu G, et al. (2006) EGFR-mediated expression of aquaporin-3 is involved in human skin fibroblast migration. Biochem J 400(2): 225-234.
- Wang C, Rong YH, Ning FG, Zhang GA (2011) The content and ratio of type I and III collagen in skin differ with age and injury. African Journal of Biotechnology 10(13): 2524-2529.




 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.