Research Article 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Review on The Role of Zn2+ Ions in Viral Pathogenesis and the Effect of Zn2+ Ions for Host Cell-Virus Growth Inhibition
*Corresponding author: Tsuneo Ishida, Life and Environment Science Research Division, Saido, Midoriku, Saitama City, Saitama Prefecture, Japan.
Received: March 11, 2019 Published: March 21, 2019
DOI: 10.34297/AJBSR.2019.02.000566
Abstract
Antiviral activities of Zn2+ ions for viral pathogenesis process and inhibition by Zn2+ ions for host cell-virus growth have been investigated. Zn2+ ions inhibit viral entry, local replication, and spread to organ during viral pathogenesis process. ZFNs may become a potential antiviral agent for restricting HIV-1 integration. The ZAP inhibits viral entry and HIV-1 infection. ZMPSTE24 cooperates with IFITM to inhibit viral entry. ZNF502 was confirmed with siRNA knockdown of the host protein levels resulting in reduced RSV virus production in infected cells. Zinc finger-attacking compound can inhibit HIV-1 and MuLV zinc fingers viral replication in vivo, in which anti-retroviral drugs which target the nucleocapsid zinc finger may be clinically useful against HIV-1. Zn2+ ions also inhibit Nidovirus replication that increasing the intracellular Zn2+ concentration can efficiently impair the replication of a variety of RNA viruses. A viral infection spread in the infected cell is involved in direct cell-to-cell transmission and cellfree transmission to spread within a host that the effects of zinc ions (using 15 mM - ZnSO4 solution) against HSV infection. Zinc oxide tetrapods also inhibit viral entry and spread of HSV infection.
Zinc binding motifs such as catalyst, structural, and regulatory ion are found in many proteins encoded by the human genome physiologically and free zinc is many regulated at the single-cell level. Zinc interferes with IFN-λ3 binding to IFNL-receptor 1 (Lambda interferons), resulting in decreased antiviral activity and increased viral replication (HCV, influenza) in vitro. Zn-treated viral capsids are still able to interact with receptor binding sites and Zn-treated HRVs exhibit an increased susceptibility to genomic RNA degradation, a phenomena that may be facilitated by a Zn-mediated cleavage of viral RNA within the viral capsid, in which the mechanism of Zn-mediated inhibition could occur in a cell-independent, extracellular manner to degrade viral RNA and thereby abrogate viral infectivity. Measles virus V protein of an RNA-binding modulatory factor represses genome replication that MV may modulate the immune response, in which these results provide a modular view of common and unique dendritic cells (DC) responses after infection.
ZAP is a host factor that restricts the infection of many viruses mainly through RNA degradation, translation inhibition and innate immune responses that ZAP may act as an intrinsic antiviral factor through specific RNA binding to fight against JEV infection as the first ZAP-sensitive flavivirus. ZMPSTE24 is a virus-specific effector that restricts enveloped RNA and DNA viruses, influenza A, Zika, Ebola, Sindbis, vesicular, cowpox, and vaccinia. Virucidal effect on zinc-coordinated ligand binding in both MMP13 complexes and zinc-finger protein has been indicated in the relative binding free energies of selected inhibitor binding and Zn2+ exerts a strong influence on the relative affinities of the ligands against nucleocapsid protein.
In antiviral therapy, the toxicological effects of zinc oxide nanoparticles (ZnO NPs) are considered as the toxicity of released Zn2+ ion from ZnO NPs aqueous dissolution (ZnO+H2O ⇄ Zn2+ + 2OH-). ZOTEN provides for the protective efficacy of an intravaginal microbicide/vaccine or microbivac platform against genital herpes infections.
Keywords: Zn2+ ion; viral pathogenesis; viral entry/local replication/spread; HIV-1; ZAP; ZOTEN
Abbreviations: AZPS: Artificial Zinc Finger Proteins; BLV: Bovine Leukemia Virus; CNBPS: Cellular Nucleic Acid-Binding Proteins; DCS: Dendritic Cells; Fr-MLV: Friend Murine Leukemia Virus; HAD: HIV-1-Associated Dementia; HBV: Hepatitis B Virus; HCV: Hepatitis C Virus; HDV: Hepatitis D Virus; HIV-1: Human Immune deficiency Virus-1; HMPV: Human Metapneumovirus; HSV=Herpes Simplex Virus; HRVS: Human Rhinoviruses; HTLV- 1: Human T-Cell Lymphotropic Virus Type 1; IAV: Influenza A Virus; IFITM: Interferon-Induced Transmembrane Protein; JAV: Japanese Encephalitis Virus; JUNV: Junin Virus; LASV: Lassa Virus; MDCK: Madin-Darby Canine Kidney; MT: Metallothionein; Mmps: Matrix Metalloproteinases; MHV: Mouse Hepatitis Virus; Mulv: Murine Leukemia Virus; MP: Mononuclear Phagocytes; MT: Metallothionein; MV: Measles Virus; NC: Nucleocapsid; NMDA: N-Methyl-D-Aspartate; Nmdars: NMDA Receptors; OA: Oleic Acid; PDTC: Pyrrolidine Dithiocarbamate; RBD: Receptor-Binding Domain; RSV: Respiratory Syncytial Virus; SFV: Semliki Forest Virus; SIV: Simian Immunodeficiency Virus; SSP: Stable Signal Peptide; TACV: Tacaribe Virions; TM: Transmembrane Protein; Vif: Virion Infectivity Factor; ZAP: Zinc-Finger Antiviral Protein; ZBD: Zinc-Binding Domain; ZIP: Zrt-,Irt-Related Protein; ZMPSTE24: Zinc Metallopeptidase STE24; Znfs: Zinc-Finger Proteins; Zn0 NPs: Zinc Oxide Nanoparticles; ZNT: Zn Transporter; ZOTEN: Zinc Oxide Tetrapod Nanoparticles
Introduction
Zinc element is an essential micronutrient for human health. Zinc controls the enzymes that operate and renew the cells in our bodies, in which zinc play in the maintenance of immune functions including cellular and humoral immunity and changes in the intracellular concentration of zinc ions control immune cell signal transduction by regulating the activity of major signaling molecules including kinases, phosphatases and transcription factors [1]. Zinc is established to be essential for an immune system, which zinc flux and zinc homeostasis control the adequate function of innate as well as adaptive immunity. Dietary Zn may influence the immune system that at low concentrations of zinc, it serves as an essential nutrient and functions as a metal cofactor for several enzymes, and high concentrations are relatively nontoxic, and these concentrations have proven useful as an alternative dietary approach for molt induction [2]. The role of Zn2+ ions in neuropathy, neuropathology, neurophysiology, and neuropathogenesis is an important term for both physiological and pathological processes. Specially, Zn2+ ions in virus neuropathogenesis play significant functions for human immunodeficiency-1 (HIV-1) infection [3] and HIV-induced central nervous system (CNS) inflammation and neurodegeneration [4]. Cellular Zn homeostasis is mediated by three main mechanisms which one is the transportation of Zn into the cytosol through plasma membrane by importers from the ZIP-family, second is exported out of the cytosol by proteins from the ZnT-family, and third is due to Zn-binding proteins such as metallothionein (MT) that maintain Zn homeostasis [5]. In this review, it is described on viral pathology that zinc(Ⅱ) ions inhibit virus pathogenesis and suppress host cell-virus growth.
Zinc Homeostasis, Immune System, and Inflammation
The human body containing 2-3 g zinc and nearly 90% in muscle and bone is found and on the cellular level, 30-40% of zinc is localized in the nucleus, 50% in the cytosol and the remaining part is associated with membrane [6]. In humans, high zinc concentrations are found in retina (3.8μg/g dry weight), choroid of the eye (274μg/g) and in bone(100-250μg/g), while only 1μg/mL zinc is found in plasma, which equals around 0.1% of total body zinc [7]. Zinc ion homeostasis is crucial for an adequate function of the immune system. Zinc deficiency as well as zinc excess result in severe disturbances in immunity, which can result in increased pathogenesis owing to susceptibility to infections and development of inflammatory diseases [7]. Chronic inflammation and oxidative stress are important causative roles in many chronic diseases and a long-term decrease in zinc availability may contribute to pathological processes in conditions of chronic inflammation [8]. Zinc deficiency causes severe impairment of immune function, comprising the adaptive as well as the innate immune system. A balanced zinc homeostasis is critical for either depending against invading pathogens or protecting the human body against an overreactive immune system causing autoimmune diseases [8,9]. On the other hand, high zinc excess provokes an impairment of the immune system and can cause focal neuronal pathology, hence zinc deficiency or excess is significant clinical concern [10]. Zinc can be recognized as potential therapeutic for clinical use to contribute beneficially the stability of patients suffering from immune and inflammatory diseases.
Viral Pathogenesis Process
Viral pathogenesis which viruses cause disease, addresses the capacity of a virus to cause injuries to cells and tissues that injuries are secondary consequences of many fundamental interactions during infection [11]. Viral Pathogenesis is the process by which virus infection leads to disease that pathogenic mechanism of viral disease includes implantation of virus at the portal of entry, local replication, and spread to the organs and the environment [12]. It is therefore important to study the therapeutic potential of virus entry inhibitors, especially when combined with strategies to induce immune-mediated killing of infected hepatocytes that strategic medicines with basic and clinical gastroenterology and hepatology inhibit entry of hepatitis B virus (HBV) and hepatitis D virus (HDV) into hepatocytes [13]. Viral entry is that to infect its host, a virus must first enter cells at a body surface that common sites of entry include the mucosal linings of the respiratory, alimentary, and urogenital tracts, the outer surface of the eye, and the skin [14].
The spikes with coronavirus spike protein function to define viral tropism by their receptor specificity and also by their membrane fusion activity during virus entry into cells that evidence supporting a role for spike protein projections as agents of organ tropism and pathogenesis began with comparative studies of different naturally occurring mouse hepatitis virus (MHV) strains [15], in which the diversity among coronaviruses and the receptors provides models for understanding early events in viral pathogenesis. The host factors control pathogenesis and identified viral entry as a ratelimiting step for infection in Drosophila melanogaster model of pathogenesis that the clathrin-mediated endocytotic pathway is essential for both infection and pathogenesis [16].
Influenza virus human pathogen causes annual epidemics and occasional pandemics. As entry is the first essential step of virus replication and is an ideal target to block infection efficiently, new entry inhibitors for influenza A virus are particularly of importance for anti-influenza drugs [17]. New aspects of interaction between arenaviruses, Lassa virus (LASV) cell and host cell have been appeared or revealed from the entry sites that the molecular mechanism of receptor recognition, endocytosis, and the use of endosomal entry factor [18]. As the entry inhibitors, viral entry is the first essential step of virus replication and is an ideal target to block infection efficiently that the entry process of the virus represents a favorable target for drug development, as inhibition of this first step of virus infection should result in an efficient block of virus propagation. The interaction of influenza viruses with their receptor and sialic acid, receptor-mediated endocytosis, and the subsequent endosomal trafficking of virus are described, however, recently a focus has been on the protein function during entry and established entry inhibitors targeting viral and host factors as well as latest prospects for designing novel anti-entry compounds [17]. Keggin polyoxometalate (PT-1, K6HPTi2W10O40) [19] and EGA [20] may be developed as new anti-HIV-1 [19], and multiple toxins and viruses [20] agents through these entry inhibitions.
Local replication has feature specific to viral replication in the host that viruses infect their hosts in three general patterns; local, “local + systemic,” and systemic infection, in which local infection refers to growth of virus near its site of entry into the host, and systemic infection refers to growth at sites distant from the entry site [11]. Viral replication in brain mononuclear phagocytes (MP) affects leukocyte infiltration, astrocytosis, formation of microglial nodules, and neuronal injury/death, the pathological hallmarks of HIV encephalitis and HIV-1-associated dementia (HAD), in which a mechanism for this antiretroviral activity was by suppression of HIV type 1 Tat-induced viral long terminal repeat promoter activity [21]. Picornaviruses cause several diseases, not only in humans but also in various animal hosts. The virus-host interaction is important for viral replication, virulence and pathogenicity that the interactions of viral capsid proteins with host cell factors are addressed in viral replication, viral translation and the switch from translation to RNA replication [22]. Pyrrolidine dithiocarbamate (PDTC) inhibits picornavirus polyprotein processing and RNA replication of human rhinovirus by transporting Zn2+ ions into cells [23]. Although cell fusion directed by the aminoterminal end of transmembrane protein (TM) is postulated to be essential, some proviruses expressing fusion-deficient envelope proteins unexpectedly replicate at wild-type levels that N-linked glycosylation site of the extracellular envelope protein inhibits cellto- cell transmission [24].
Sequence of virus spread to the organs may be followed by local replication and local spread of viruses that most viruses spread extracellularly, but herpesviruses, paramyxoviruses, and poxviruses may spread through both intracellular and extracellular routes [12]. To study the consequences of the availability of a high-affinity receptor for Measles virus (MV) propagation, in miceα/βinterferon controls MV infection and a high-affinity receptor facilitates but is not strictly required for MV spread and pathogenesis [25]. Viral infections exemplified by various viruses have been described about the enzyme Heparanase (HPSE) in infection, spread, and pathogenesis which HPSE’s role in viral pathogenesis predominantly in the area of virus-host interactions [26]. Herpes simplex virus type-1 (HSV-1) infection upregulates HPSE in a manner dependent on HSV-1 infected cell protein that HPSE relocates to the nucleus to regulate cytokine production, enhances viral spread, and thus generates a toxic local environment [27].
Host Protein-Virus Interactions Control Viral Pathogenesis
Interaction of viral envelope proteins with host cell membrane has been extensively investigated in a number of systems. Influenza A virus (IAV)-host interactions contribute to host defense and viral pathogenesis that the interactions may facilitate virus hijacking of host molecular machinery to fulfill the viral life cycle and trigger host immune defense to eliminate the virus [28]. The pathogenesis of HIV infection is a function of the viral life cycle, host cellular environment, and quantity of viruses in the infected individual which HIV pathogenesis is basically a competition between HIV replication and the immune responses of the subject or patient via the cell-mediated and immune-mediated reaction [29]. A defective RNA virus of hepatitis D virus (HDV) that requires the help of hepatitis B virus (HBV) for its replication and assembly of new virions, in which the processes of viral replication, HBV-HDV interaction, and etio-pathogenesis of the severe course of HDV infection have identified the potential therapeutic targets in the virus life cycle for the prophylaxis and treatment of infection and complications [30]. Zika virus which is a mosquito-borne flavivirus, has identified potential targets for antiviral drugs that the clarification for some mechanisms of the Zika virus pathogenesis has been caused to control replication in the placenta and brain, fetal demise and neural damage, and human disease [31].
Zn2+ Ions Inhibit Virus Pathogenic Process
HIV viral entry into host cells is complicated processes involving an attachment of receptor binding, co-receptor binding, and fusion process [32]. These attachment inhibitors, co-receptor binding inhibitors, and fusion inhibitors are under clinical trials that the susceptibility of treatment-native viral strains to different entry inhibitions targeting viral envelop glycoprotein may vary significantly. Engineered zinc-finger nucleases (ZFNs) in the HIV coreceptor CCR5 confers resistance for viral entry and HIV-1 infection with disrupting endogenous CCR5 [33]. ZFNs also inhibit viral entry and infection that ZFNs is involved in the early steps of the viral replication cycle, thus, ZFNs may become a potential antiviral agent for restricting HIV-1 integration [34]. The zinc-finger antiviral protein (ZAP) inhibits viral entry and HIV-1 infection by recruiting both the 5’ and 3’ mRNA degradation machinery to specifically promote the degradation of multiply spliced HIV-1 mRNA [35]. Zinc inhibition of Semliki forest virus (SFV) fusion and infection has been studied that zinc acts by blocking the fold-backed of DIII via interaction with H333, in which zinc’s stereochemical flexibility allows it to be coordinated by 2 to 8 ligands with 4-, 5- and 6-liganded forms seen most often in nature [36]. Zinc metallopeptidase STE24 (ZMPSTE24) cooperates with interferon-induced transmembrane protein (IFITM) to inhibit viral entry that IFITM protein impede viral entry and ZMPSTE24 expression is necessary for IFITM antiviral activity [37]. Zn2+ binding protease is developed anti-virus agent through its effect on entry inhibition.
The respiratory syncytial virus (RSV) matrix protein is found in the nucleus early in infection in a transcriptional inhibitory role, and later localizing in viral inclusion bodies before coordinating viral assembly and budding at the plasma membrane. Then, Zinc finger protein ZNF502 was confirmed with siRNA knockdown of the host protein levels resulting in reduced RSV virus production in infected cells [38]. Monitored viral spread by measuring the level of viral DNA in the peripheral blood mononuclear cells had been investigated on competitive polymerase chain reaction that a zinc finger-attacking compound can inhibit HIV-1 and murine leukemia virus (MuLV) zinc fingers viral replication in vivo, in which antiretroviral drugs which target the nucleocapsid zinc finger may be clinically useful against HIV-1 [39]. Zn2+ ions also inhibit Nidovirus replication that increasing the intracellular Zn2+ concentration can efficiently impair the replication of a variety of RNA viruses, including poliovirus and influenza virus, in which the inhibitory effect of Zn2+ could be reversed that zinc ionophores block the replication and transcription [40]. Inhibition of HIV-1 replication causes a bis-thisadiazolbenzene-1, 2-diamine to block efficiently the replication of a wide spectrum of HIV-1, HIV-2, and simian immunodeficiency virus (SIV) strain with compound involving in the chelating Zn2+ ion [41]. Artificial zinc finger proteins (AZPs) inhibit virus DNA replication that since the mechanism of viral DNA replication is conserved among plants and mammals which this approach could be applied to the prevention virus infection in human [42]. Virucidally coadministered zinc ions have potential as multi-action novel topical therapeutic agent against HSV infections such as coldsores [43]. The pathogenesis of influenza virus infection involves viral replication in epithelial cells of the respiratory tract that inhibitory effect on influenza virus induced apoptotic death can be determined at an early stage of the infection by zinc ions [44].
A viral infection spread in the infected cell is involved in direct cell-to-cell transmission and cell-free transmission to spread within a host [45]. Spread effects of Zn2+ cause important role that is not well understood. The avoidance of development and spread of virus is of importance for non-severe herpes simplex virus (HSV) in physiologically tolerable drugs, in which the molecular mechanism of the effects of zinc ions (using 15 mM-ZnSO4 solution) against HSV infection was investigated [46]. This result is that the molecular mechanism of therapeutic effect of ZnSO4 in HSV infection treatment is not due to a cytoinvasive intracellular inhibition of virus replication, but to the drastic inactivation of free virus in skin tissues, intercellular vesicles and blisters, in which affects penetration rather than adsorption, corroborates the interpretation that glycoproteins accumulate the metal until the zinc content becomes incompatible with the glycoprotein functions and inhibition of virus growth [46]. Evidence for a specific interaction between Tat and the HIV-1 glycoprotein 120 (gp120) envelope protein that enhances virus attachment and entry into cells, in which the Tat-gp120 interaction represents a critical step in HIV-1 spreading in the course of infection [47] Table 1 is indicated that Zn2+ inhibit viral entry, local replication, and spread into organ on virus pathogenesis process.
The role of Zn ion transporters in the pathophysiology of infection
Zinc element play essentially crucial roles in numerous physiological, but also pathological processes. Dietary zinc is absorbed in the small intestine and then distributed to peripheral tissues that approximately 60% of zinc is stored in skeletal muscle, ~30% in bones, and ~5% is stored in the liver and skin. The remaining percentage is distributed to other tissues such as brain, kidney, and pancreas. Excess zinc is excreted through gastrointestinal secretion, sloughing mucosal cells. Zinc transport proteins are indispensable for the physiology of zinc that Zn transporter (ZnT) and Zrt-, Irt-related protein (ZIP) contribute to a wide array of physiological and cellular functions such as immune, reproductive, and neuronal by tightly controlling zinc homeostasis [48]. The gastrointestinal tract plays a major role in the regulation of zinc levels in the body that zinc absorption takes place mostly in the small intestine, where zinc homeostasis is regulated, in which zinc is absorbed into intestinal cells through Zip4 transporter [49]. Transport of zinc in blood becomes to that zinc is always bound to other particles to be transported: albumin (50%), transferrin and α-2-macroglobulin (40%), and remaining amino acid (10%), the other, transport of zinc in cell becomes to that ZIP and ZnT proteins are transported and completely dedicated to zinc transport [49].
ZnT8 function has been involved in pathogenesis of type2 diabetes, indicating that the precise control of zinc homeostasis is crucial for maintaining health and preventing various diseases, and ZIP13 homeostatic-regulated is adipocyte biogenesis which indicated zinc homeostasis regulation as possible therapeutic target for obesity and metabolic syndrome [50]. The function of zinc ions in proteins with intake and controlling mechanisms has been examined in both physiological and pathological processes that imbalance of abundance and regulation of the intestinal ion transporter proteins occurs and cause viral infections and diseases [51-53]. Zinc-finger proteins (ZNFs) are implicated in transcriptional regulation, ubiquitin-mediated protein degradation, signal transduction, actin targeting, DNA repair, and cell migration [54]. Thus, zinc binding motifs such as catalyst, structural, and regulatory ion are found in many proteins encoded by the human genome physiologically and free zinc is many regulated at the single-cell level [55].
Virus receptors/Cellular receptors
A virus initiates infection by attaching to its specific receptor on the surface of susceptible host cell that the receptor is a major determinant of the route of entry into the host, the pattern of spread in the host, and the resulting pathogenesis [56]. The virus receptor not only can act a point of attachment for the virus but also may be important in virus entry, intracellular targeting, and uncoating, in which understanding virus-receptor interactions are under basic progress. Virus-receptor interaction is a multistep process, multiple attachment receptors may be used sequentially, or in a cell-typespecific manner, and co-receptors may also be involved which four steps of virus entry into target cells are
- the attachment in envelope protein,
- co-receptors and entry mediators at plasma membrane,
- membrane fusion, and
- release of nucleocapsids and initiation of transcription [57].
In infected organs, cell-to-cell spread contributes significantly to the pathogenesis of a viral diseases that a major impact on pathogenesis is due to the immune-suppressive capacity of many viruses which can also be exerted via cell surface receptor interactions [57]. Zinc interferes with IFN-λ3 binding to IFNLreceptor 1 (Lambda interferons), resulting in decreased antiviral activity and increased viral replication (HCV, influenza) in vitro [58].
Entry of bovine leukemia virus (BLV) and human T-cell lymphotropic virus type 1 (HTLV-1) into the host cell is supposed to be mediated by interactions of the extracellular (SU) envelope glycoproteins with cellular receptors that the envelope of the related HTLV-1 was able to bind zinc and zinc ions were associated with the receptor-binding domain (RBD) of Friend murine leukemia virus (Fr-MLV) SU glycoprotein, in which zinc ions could mediate interactions of the SU RBD either with the C-terminal part of SU structural integrity, or with a partners different from the receptor [59]. The mutations in the glucocorticoid receptor including two zinc fingers show mutants with interdigitated DNA binding and second finger selectively impaired transcriptional enhancement activities, in which such positive control mutants may alter proteinprotein contacts required for transcriptional activation [60]. Entry of the virus into the host cell is mediated by the viral envelope glycoprotein (GPC).
In contrast to other class Ⅰ viral envelope glycoproteins, the mature GPC complex contains a cleaved stable signal peptide (SSP) in addition to the canonical receptor-binding (G1) and transmembrane-fusion (G2) subunits. A cleaved SSP is retained in viral envelope GPC through interaction with a zinc-binding domain (ZBD) in the cytoplasmic tail of G2 [61]. Junin virus (JUNV) ZBD displays a novel fold containing two zinc ions that one zinc ion is coordinated by His-447, His-449, Cys-455, and His-485, and the second zinc ion is coordinated by His-459, Cys-467, and Cys-469 and accepts Cys-57 from SSP as the fourth ligand [61]. Picomolar Zn2+ ion concentrations regulate receptor protein-tyrosine phosphatase and MyD88/TRIF activities which zinc modulates signal transduction in endothelial cells affecting angiogenesis [62,63]. Extracellular zincmediated inhibition against human rhinoviruses (HRVs) had been found that Zn-treated viral capsids are still able to interact with receptor binding sites and Zn-treated HRVs exhibit an increased susceptibility to genomic RNA degradation, a phenomena that may be facilitated by a Zn-mediated cleavage of viral RNA within the viral capsid, in which the mechanism of Zn-mediated inhibition could occur in a cell-independent, extracellular manner to degrade viral RNA and thereby abrogate viral infectivity [64].
Synaptic and tonic zinc modulates extrasynaptic N-Methyl- D-aspartate (NMDA) receptors (NMDARs) that synaptic zinc is phasically released during action potentials and inhibit extrasynaptic NMDARs, in which a physiological role for endogenous synaptic as well as tonic zinc in inhibiting extrasynaptic NMDARs and thereby fine turning neuronal excitability and signaling [65]. Zinc and pH are physiological allosteric modulators of NMDARs with GluN2A containing receptors inhibited by nanomolar concentrations of divalent zinc and by excursions to low pH why how zinc binding to the amino terminal domain elicits structural changes that are transduced through the ligand-binding domain and result in constriction of the ion channel gate [66].
Zinc binding activity
Zinc binding studies were performed using the minimal nonstructural (NS) protease domain within the NS proteins (NSPs)/open reading frames (ORFs) that the protease activity can be blocked by both metal ion chelators and metalloprotease inhibitor captopril, in which the rubella virus (RUB) nonstructural protease is actually a novel virus metalloprotease [67]. Zinc binding activity is important as zinc binding motif (CCCH) of human metapneumovirus (hMPV) M2-1, whereas the first two cysteines play only minor or redundant roles in zinc binding [68]. These results showed that zinc binding activity is indispensable for viral replication and pathogenesis in vivo and may serve as a novel approach to rationally attenuate hMPV. The molecular mechanism against the hMPV has been investigated and used by the hMPV to avoid the host immune system, comparing with human respiratory syncytial virus or influenza virus [69]. The direct treatment of Junin virus (JUNV) and Tacaribe (TACV) virions with zinc finger active compounds showed that the aromatic disulphide was a potent virucidal agent and the other two compounds exhibited negligible virus-inactivating properties [70]. The zinc finger domain is the only structured region of the nucleocapsid protein Gn tail that these structural observations provide further insight into the role of the Gn tail during viral assembly as well as its role in pathogenesis [71]. CCCH-type zinc-finger antiviral protein (ZAP) is a host factor that restricts the infection of many viruses mainly through RNA degradation, translation inhibition and innate immune responses that ZAP may act as an intrinsic antiviral factor through specific RNA binding to fight against Japanese encephalitis virus (JEV) infection as the first ZAP-sensitive flavivirus [72]. Zn2+-cysteine complexes have been observed across diverse protein classes and known to facilitate a variety of cellular process that novel zinc-binding cysteines have structural characteristics and diverse functional roles of Zn2+ ions-cysteine complexes in proteins [73]. Zinc metallopeptidase STE24 (ZMPSTE24) is a transmembrane metalloprotease that ZMPSTE24-mediated antiviral action is independent of protease activity, in which ZMPSTE24 is a virusspecific effector that restricts enveloped RNA and DNA viruses, influenza A, Zika, Ebola, Sindbis, vesicular, cowpox, and vaccinia, but not murine leukemia or adenovirus [74]. Zn2+ binding and intracellular Zn2+ levels are critical for ICP0’s biochemical activity and that depletion of intracellular Zn2+ severely attenuate HSV-1 replication [75]. Zn2+ ions inhibit growth of some RNA viruses and DNA virus [76].
Measles virus (MV) is an important human pathogen that induces transient immunosuppression that MV is an enveloped RNA virus that MV V protein represses genome replication and may be an RNA-binding modulatory factor, in which protein-RNA complex formation was dependent upon the unique Cys-rich carboxy terminus, a region also required to induce maximal repression of minireplicon-encoded reporter gene expression in transient assays [77]. Dendritic cells (DCs) are potent antigen-presenting cells that initiate the immune response to pathogens and are postulated to play a role in MV-induced immunosuppression that MV-infected [78]. These results show that a modular view of common and unique DC responses after infection and the mechanism is suggested by which MV may modulate the immune response.
Zn2+ is bound to ligand by a tetrahedral coordination that Zn2+ can serve as a cofactor for the endonucleolytic reaction catalyzed by the full-length protein, a derivative lacking the N-terminal domain, or the isolated catalytic domain of avian sarcoma virus retroviral integrases (IN) [79]. HIV-1 nucleocapsid Zn2+ finger domains have greatly reduced infectivity, even though genome packaging is largely unaffected in certain cases that the infectivity loss in NC mutants appears to result from defective reverse transcription and integration processes stemming from decreased protection of the full-length viral DNA [80]. The HCCH motif of HIV-1 virion infectivity factor (Vif) is a unique zinc-binding domain capable of mediating protein-protein interactions in the presence of zinc and adds to growing list of examples in which zinc ion binding induces protein misfolding and aggregation [81].
Metalloproteinases
The NS5A protein of hepatitis C virus (HCV) is a zinc coordinated ligands metalloprotein corresponding to be integral part of the viral replicase that mutation of any of the four cysteine components of NS5A zinc coordination and led to a lethal phenotype for HCV RNA replication, whereas mutation of other potential metal coordination residues in the N-terminal domain of NS5A [82]. Matrix metalloproteinases (MMPs) are zinc endopeptidases that are required for degradation of extracellular matrix components during normal embryo development, morphogenesis and tissue. Virucidal effect on zinc-coordinated ligand binding in both matrix metalloproteinase13 (MMP13) complexes and zinc-finger protein has been indicated that the relative binding free energies of selected inhibitor binding with MMP against Madin-Darby Canine Kidney (MDCK) [83]. Cellular nucleic acid-binding proteins (CNBPs) Zn2+ fingers are used in antiviral therapeutics and DNA vaccines, and the Zn2+ exerts a strong influence on the relative affinities of the ligands against nucleocapsid (NC) protein [84]. MMPs also are essential as therapeutic MMP inhibitory drugs that balances between MMPs and endogenous MMP inhibitors are disturbed, mostly in favor of active proteolysis in malaria, trypanosomiasis, leishmaniasis and toxoplasmosis infections [85].
Viral growth inhibition of ZnO nanoparticles
Nanotechnology is defined as an application of scientific manipulation and control in nanoscale (1~100 nm) with specific function at the cellular, atomic and molecular levels that the nanostructures for the inhibition of viral infections have a great biological role in veterinary diagnosis and in animal reproductive biotechnologies beside great impact on diagnosis in animal diseases [86]. In antiviral therapy, zinc oxide nanoparticles (ZnO NPs) as nanomaterials offer significant benefits that there are a continuous and vital need for new antiviral therapeutics and approach to confront the emergence of drug resistance and different secondary effects due to long-term treatment. The nanostructures for the inhibition of viral infections are characterized by either enhanced affinities towards different surface viral receptors and the integration of multiple ligands onto nanostructures turned to be a viable alternative, or abbreviation endoplasmic reticulum and potential points of attack of antiviral nanostructures in the viral replication cycle [87]. Thus, receptor-base nanoparticles are one of the ways of achieving a secured administration of nanoparticles as anti-viral agents that the integration of functional groups and units onto the surface of nanoparticles allows for effectively targeting specific sites of viral infections [87]. Physicochemical properties of silane-coated and uncoated ZnO NPs (20 nm) to elucidate their toxic potencies toward three fresh water and three marine microalgae were compared that the coating ZnO NPs provide the particles with a more hydrophilic surface, the uncoating formed larger aggregates and released more Zn2+, in which each of the coating resulted in ZnO NPs acting through different mechanisms of toxic action [88].
Surface modified zinc oxide nanoparticles to freshwater and marine microalgae could modify the HSV-1 infectivity potential via neutralizing the virus, and the physical entrapment exerted by C(chitosan)-zinc nanoparticles and the electrostatic interference of H (Hydroxyl group rich)-zinc nanoparticles are more significant than the hydrophobic interaction using OA (Oleic acid modified)- zinc nanoparticles [89]. The toxicological effects of ZnO NPs are attracting increasing concern that the toxicity of released Zn2+ ion from ZnO NPs aqueous dissolution (ZnO + H2O ⇄ Zn2+ + 2OH-) is considered, but the mechanism is remained unclear. Exposure to ZnO NPs interferes with the homeostasis of Zn2+ ion concentration, and elevated Zn2+ ion concentration results in cell apoptosis [90]. Human oxidative stress and DNA damage could be occurred by dissoluted Zn2+ ions from ZnO NPs solution that the results clarify the intracellular bioaccumulation of ZnO NPs and the possible mechanisms of DNA repair and cell survival [91].
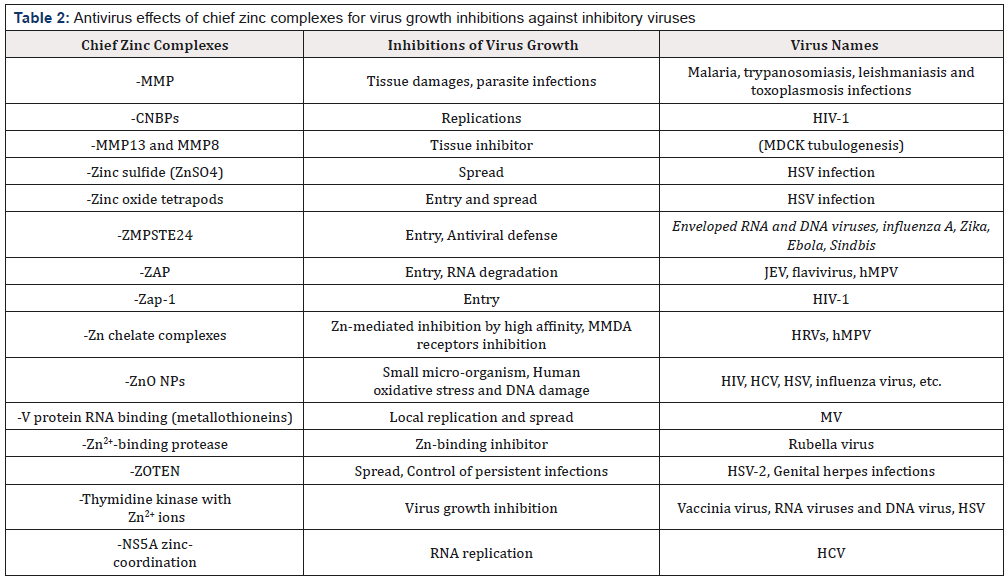
Zinc oxide tetrapods also inhibit viral entry and spread of HSV infection [92]. Zinc oxide tetrapod nanoparticles (ZOTEN) is an effective suppressor of HSV-2 genital infection, the strong HSV-2 trapping ability of ZOTEN reduced the clinical signs of vaginal infection and decreased animal mortality, enhancing T cellmediated and Ab-mediated responses to the infection, and thereby suppressing a reinfection, whereas, ZOTEN will inhibit the spread of newly produced HSV-2, control persistent infections, and be required to the relative usefulness of ZOTEN versus other adjuvants for optimizing the immunization of protective immune response to HSV-2 [93]. Thus, ZOTEN exhibits strong adjuvant-like properties, in which provides for the protective efficacy of an intravaginal microbicide/vaccine or microbivac platform against genital herpes infections, having suppression of persistent infections [93] Table 2 indicate the antivirus effects of chief zinc complexes for virus growth inhibitions against inhibitory viruses.
Conclusion
Zn2+ ions inhibit viral entry, local replication, and spread to organ during viral pathogenesis process. ZFNs may become a potential antiviral agent for restricting HIV-1 integration. The ZAP inhibits viral entry and HIV-1 infection. ZMPSTE24 cooperates with IFITM to inhibit viral entry. Zinc finger protein ZNF502 was confirmed with siRNA knockdown of the host protein levels resulting in reduced RSV virus production in infected cells. Zinc oxide tetrapods also inhibit viral entry and spread of HSV infection.
Zinc finger-attacking compound can inhibit HIV-1 and MuLV zinc fingers viral replication in vivo, in which anti-retroviral drugs which target the nucleocapsid zinc finger may be clinically useful against HIV-1. Zn2+ ions also inhibit Nidovirus replication that increasing the intracellular Zn2+ concentration can efficiently impair the replication of a variety of RNA viruses, including poliovirus and influenza virus, in which the inhibitory effect of Zn2+ could be reversed that zinc ionophores block the replication and transcription.
A viral infection spread in the infected cell is involved in direct cell-to-cell transmission and cell-free transmission to spread within a host that the effects of zinc ions (using 15 mM-ZnSO4 solution) against HSV infection was investigated. The Tat-gp120 interaction represents a critical step in HIV-1 spreading in the course of infection.
Zn transporter (ZnT) and Zrt-, Irt-related protein (ZIP) contribute to a wide array of physiological and cellular functions such as immune, reproductive, and neuronal by tightly controlling zinc homeostasis. Zinc binding motifs such as catalyst, structural, and regulatory ion are found in many proteins encoded by the human genome physiologically and free zinc is many regulated at the single-cell level. Zinc interferes with IFN-λ3 binding to IFNLreceptor 1 (Lambda interferons), resulting in decreased antiviral activity and increased viral replication (HCV, influenza) in vitro. Zntreated viral capsids are still able to interact with receptor binding sites and Zn-treated HRVs exhibit an increased susceptibility to genomic RNA degradation, a phenomena that may be facilitated by a Zn-mediated cleavage of viral RNA within the viral capsid, in which the mechanism of Zn-mediated inhibition could occur in a cell-independent, extracellular manner to degrade viral RNA and thereby abrogate viral infectivity.
CCCH-type ZAP is a host factor that restricts the infection of many viruses mainly through RNA degradation, translation inhibition and innate immune responses that ZAP may act as an intrinsic antiviral factor through specific RNA binding to fight against JEV infection as the first ZAP-sensitive flavivirus. ZMPSTE24 is a virus-specific effector that restricts enveloped RNA and DNA viruses, influenza A, Zika, Ebola, Sindbis, vesicular, cowpox, and vaccinia, but not murine leukemia or adenovirus. MMPs are zinc endopeptidases that are required for degradation of extracellular matrix components during normal embryo development, morphogenesis and tissue.
Virucidal effect on zinc-coordinated ligand binding in both MMP13 complexes and zinc-finger protein has been indicated that the relative binding free energies of selected inhibitor binding with MMP against Madin-Darby Canine Kidney (MDCK) [83], and Zn2+ exerts a strong influence on the relative affinities of the ligands against nucleocapsid protein. In antiviral therapy, ZnO NPs as nanomaterials offer significant benefits and approach to confront the emergence of drug resistance. The toxicological effects of ZnO NPs are attracting increasing concern that the toxicity of released Zn2+ ion from ZnO NPs aqueous dissolution (ZnO + H2O ⇄ Zn2+ + 2OH-) is considered. ZOTEN also exhibits strong adjuvant-like properties, in which provides for the protective efficacy of an intravaginal microbicide/vaccine or microbivac platform against genital herpes infections.
References
- Ch Bimola Devi, Th Nandakishore, Sangeeta N, Gomi Basar, N Omita Devi, et al. (2014) Zinc in human health. IOSR Journal of Dental and Medical Science 13(7): 18-23.
- SY Park, SG Birkhold, LF Kubena, Nisbet DJ, Ricke SC (2004) Review on the role of dietary zinc in poultry nutrition, immunity, and reproduction. Biol Trace Elem Res 101(2): 147-164.
- LA Boven, L Gomes, C Hery, Gray F, Verhoef J, et al. (1999) Increased peroxynitrite activity in AIDS dementia complex: Implications for the neuropathogenesis of HIV-1 infection. J Immunol 162(7): 4319-4327.
- RB Meeker, Lola Hudson (2017) Feline immunodeficiency virus neuropathogenesis: A model for HIV-induced CNS inflammation and neurodegeneration. Vet Sci 4(1).
- Julie Fernando, Shaobo Zhou (2015) The role of zinc in renal pathological changes in Diabetic status, Journal of Nutritional Disorders and Therapy 5(3): 1-8.
- Laura M Plum, Lothar Rink, Hajo Haase (2010) The essential toxin: impact of zinc on human health. Int J Environ Res Public Health 7(4): 1342-1365.
- M Maywald, I Wessels, L Rink (2017) Zinc signals and immunity. Int J Mol Sci 18(10).
- Nour Zahi Gammoh, Lothar Rink (2017) Zinc in infection and inflammation. Nutrients 9(6).
- Inga Wessels, Martina Maywald, Lothar Rink (2017) Zinc as gatekeeper of immune function. Nutrients 9(12).
- Ronald Bartzatt (2017) Neurological impact of zinc excess and deficiency in vivo. European Journal of Nutrition and Safety 7(3): 155-160.
- ML Nibert, DB Furlong, BN Fields (1991) Mechanism of viral pathogenesis, Distinct forms of reoviruses and their roles during replication in cell and host. J Clin Invest 88(3): 727-734.
- S Baron, M Fons, T Albrecht (1996) Viral pathogenesis. In: Baron S (eds.), Medical Microbiology. 4th (Edn), pp 1-7.
- S Urban, R Bartenschlager, R Kubitz, F Zoulim (2014) Review in basic and clinical gastroenterology and hepatology. Gastroenterology 147: 48- 64.
- V Racaniello Viral Pathogenesis Viral Entry. MID 31: 1-19.
- TM Gallagher, MJ Buchmeiert (2001) Coronavirus spike proteins in viral entry and pathogenesis. Virology 279(2): 371-374.
- Sara Cherry, Norbert Perrimon (2004) Entry is a rate-limiting step for viral infection in a Drosophila melanogaster model of pathogenesis. Nat Immunol 5(1): 81-87.
- TO Edingert, MO Pohlt, S Stertz (2014) Entry of influenza A virus: host factors and antiviral targets, Journal of General Virology, 95(2): 263-277.
- G Torriani, C Galan-Navarro, S Kunz (2017) Lassa virus cell entry reveals new aspects of virus-host cell interaction. J Virol 91(4): 1-8.
- X Wang, J Wang, W Zhang, Li B, Zhu Y, et al. (2018) Inhibition of human immunodeficiency virus type 1 entry by a Keggin Polyoxometalate. Viruses 10(5): 1-15.
- EJ Gillespire, Chi-Lee C Ho, K Balaji, Clemens DL, Deng G, et al. (2013) Selective inhibitor of endosomal trafficking pathways exploited by multiple toxins and viruses. Proc Natl Acad Sci USA 110(50): E4904-E4912.
- KA Carlson, G Leisman, J Limoges, Pohlman GD, Horiba M, et al. (2004) Molecular characterization of a putative antiretroviral transcriptional factor. J Immunol 172(1): 381-391.
- Jing-Yi Lin, Tzu-Chun Chen, Kuo-Feng Weng, Chang SC, Chen LL, et al. (2009) Viral and host proteins involved in picornavirus life cycle. J Biomed Sci 16: 103.
- K Lanke, BM Krenn, WJG, Seipelt J, Van Kuppeveld FJ (2007) PDTC inhibits picornavirus polyprotein processing and RNA replication by transporting zinc ions into cells. J Gen Virol 88(4): 1206-1217.
- Alix de Brogniez, Jan Mast, Luc Willems (2016) Determinants of the Bovine Leukemia Virus envelope glycoproteins involved in infectivity, replication and pathogenesis. Viruses 8(4): 88.
- B Mrkic, J Pavlovic, T Rulicke, Volpe P, Buchholz CJ, et al. (1998) Measles virus spread and pathogenesis in genetically modified mice. J Virol 72(9):7420-7427.
- Neel Thakkar, Tejabhiram Yadavalli, Dinesh Jaishankar, Deepak Shukla (2017) Emerging role of heparinase in viral pathogenesis. Pathogen 6(3).
- AM Agelidis, SR Hadigal, D Jaishankar, D Shukla (2017) Viral activation of heparinase drives pathogenesis of herpes simplex virus-1. Pathogens 20(2): 439-450.
- M Zhao, L Wang, S Li (2017) Influenza A virus-host protein interactions control viral pathogenesis. Int J Mol Sci 18(8).
- Odladipo EK, Awoyelu EH (2015) Pathogenesis of HIV: Pathway to eradication. Pelagia Research Library 6(5):81-87.
- Zaigham Abbas, Rafia Afzal (2013) Life cycle and pathogenesis of hematitis D virus: A review. World J Hepatol 5(12): 666-675.
- L Barzon, M Trevisan, A Sinigaglia, et al. (2016) Zika virus: from pathogenesis to disease control, FEMS Microbiol Lett 363(18).
- K Qian, SL Morris-Natschke, K-H Lee (2009) HIV entry inhibitors and their potential in HIV therapy. Med Res Rev 29(2): 369-393.
- EE Perez, J Wang, JC Miller, Jouvenot Y, Kim KA, et al. (2008) Establishment of HIV-1 resistance in CD4+ T cells by genome editing using zinc-finger nucleases. Nat Biotechnol 26(7): 808-816.
- Roger Badia, Eduardo Pauls, Eva Riveira-Munoz, Clotet B, Esté JA, et al. (2014) Zinc finger endonuclease targeting PSIP1 inhibits HIV-1 integration. Antimicrob Agents Chemother 58(8): 4318-4328.
- Y Zhu, G Chen, F Lv, Wang X, Ji X, et al. (2011) Zinc-finger antiviral protein inhibits HIV-1 infection by selectively targeting multiply spliced viral mRNAs for degradation. Proc Natl Acad Sci USA 108(38): 15834-15839.
- CY Liu, M Kielian (2012) Identification of a specific region in the E1 fusion protein involved in zinc inhibition of Semliki Forest virus (SFV) fusion. Journal of Virology 86(7): 3588-3594.
- B Fu, L Wang, S Li, M Dorf (2017) ZMPSTE24 defends against influenza and other pathogenic viruses. J Exp Med 214(4): 919-929.
- S Kipper, S Hamad, L Caly, Avrahami D, Bacharach E, et al. (2015) New host factors important for respiratory syncytial virus (RSV) replication revealed by a novel microfluidics screen for interactors of matrix(M) protein. Mol Cell Proteomics 14(3): 532-543.
- DE Ott, SM Hewes, WG Alvord, Henderson LE, Arthur LO, et al. (1998) Inhibition of friend virus replication by a compound that reacts with the nucleocapsid zinc finger: Anti-retroviral effect demonstrated in vivo. Virology 243(2): 283-292.
- AJW te Velthuis, SHE Van den Worm, AC Sims, Baric RS, Snijder EJ, et al. (2010) Zn2 + inhibits coronavirus and arterivirus RNA polymerase activity in vitro and zinc ionophores block the replication of these viruses in cell culture. PLOS Pathog 6(11).
- C Pannecouque, B Szafarowicz, N Volkova, Bakulev V, Dehaen W, et al. (2010) Inhibition of HIV-1 replication by a bis-thiadiazolbenzen-1, 2-diamine that chelates zinc ions from retroviral nucleocapsid zinc fingers. Antimicrob Agents Chemother 54(4): 1461-1468.
- Takashi Sera (2005) Inhibition of virus DNA replication by artificial zinc finger proteins, J Virol 79(4): 2614-2619.
- DMJ Houston, JJ Bugert, SP Denyer, Heard CM (2017) Potentiated virucidal activity of pomegranate rind extract (PRE) and punicalagin against Herpes simplex virus (HSV) when co-administered with zinc(Ⅱ) ions, and antiviral activity of PRE against HSV and aciclovir-resitant HSV. PLOS one 12(6).
- V Srivastava, S Rawall, VK Vijayan, M Khanna (2009) Influenza a virus induced apoptosis: Inhibition of DNA laddering& caspase-3 activity by zinc supplementation in cultured HeLa cells. Indian J Med Res 129(5): 579-586.
- Frederik Graw, Alan S Perelson (2016) Modeling viral spread, Annu Rev Virol 3(1): 555-572.
- G Kumel, S Schrader, H Zentgraf, et al. (1990) The mechanism of the antiherpetic activity of zinc sulphate. J Gen Virol 71: 2989-2997.
- S Marchio, M Alfano, L Primo, D Gramaglia, Gramaglia D, Butini L, et al. (2005) Cell surface-associated Tat modulates HIV-1 infection and spreading through a specific interaction with gp120 viral envelope protein. Immunology 105(7): 2802-2813.
- T Kambe, T Tsuji, A Hashimoto, N Itsumura (2015) The physiological, biochemical, and molecular roles of zinc transporters in zinc homeostasis and metabolism. Physiol Rev 95: 749-784.
- M Tyszka-Czochara, J Gdula-Argasinska, T Librowski, et al. (2012) Zinc, the trace element essential in living organisms, MIR N-ro 2(25): 55-60.
- A Fukunaka, Y Fujitani (2018) Role of zinc homeostasis in the pathogenesis of diabetes and obesity. Int J Mol Sci 19(2).
- J Molina-Lopez, E Planells del Pozo, DJ Frorea, et al. (2016) Nutritional and methodological perspectives of zinc ions and complexes - Physiological and pathological states. International Journal of Electrochemical Science 11: 4470-4496.
- S Das, R Jayaratne, E Barrett (2018) The role of ion transporters in the pathophysiology of infectious diarrhea. Cell Mol Gastroenterol Hepatol 6(1): 33-45.
- Associate Prof Tsuyoshi Syuto (2017) Defect in zinc supply mechanism affects pathology of intractable pulmonary diseases. 20171-20173.
- M Cassandri, A Smirnov, F Novelli, Pitolli C, Agostini M, et al. (2017) Zincfinger proteins in health and disease. Cell Death Discov 3: 17071.
- CT Chasapis, AC Loutsidou, CA Spiliopoulou, ME Stefanidon (2012) Zinc and human health: an update. Arch Toxicol 86(4): 521-534.
- Leonard C Norkin (1995) Virus receptors: implications for pathogenesis and the design of antiviral agents. Clin Microbiol Rev 8(2): 293-315.
- J Schneider-Schaulies (2000) Cellular receptors for viruses: link to tropism and pathogenesis. J Gen Virol 81(6): 1413-1429.
- SA Read, KS O’Connor, V Suppiah, Ahlenstiel CLE, Obeid S, et al. (2017) Zinc is a potent and specific inhibitor of IFN-λ3 signaling. Nat Commun 8: 15245.
- J-S Gatot, I Callebaut, C Van Lint, et al. (2002) Bovine Leukemia Virus SU protein interacts with zinc, and mutations within two interacting regions differently affect viral fusion and infectivity in vivo. Journal of Virology 76(16): 7956-7967.
- M Schena, LP Freedman, KR Yamamoto (1989) Mutation in the glucocorticoid receptor zinc finger region that distinguish interdigitated DNA binding and transcriptional enhancement activities. Genes Dev 3(10):1590-1603.
- K Briknarova, CJ Thomas, J York, JH Nunberg (2011) Structure of a zincbinding domain in the Junin virus. J Biol Chem 286(2): 1528-1536.
- M Wilson, C Hogstrand, W Maret (2012) Picomolar concentrations of free zinc(Ⅱ) ions regulate receptor protein-tyrosine phosphatase β activity. Journal of Biological Chemistry 287(12).
- A Brieger, L Rink, H Haase (2013) Differential regulation of TLRdependent myD88 and TRIF signaling pathways by free zinc ions. J Immunol 191(4): 1808-1817.
- Ashlee Bennet (2016) Characterization of extracellular zinc-mediated inhibition of human HRVs. Columbia University, Doctor of Philosophy graduate. 2016, ABSTRUCT.
- CT Anderson, RJ Radford, ML Zastrow, Zhang DY, Apfel UP, et al. (2015) Modulation of extrasynaptic NMDA receptor by synaptic and tonic zinc. Proc Natl Acad Sci USA 112(20): E2705-E2714.
- F Jalali-Yaddi, S Chowdhury, Craig Yoshioka, Eric Gouaux (2018) Mechanism for zinc and proton inhibition of the GluN1/GluN2A NMDA receptor. 175(6): 1520-1532.
- X Liu, J Yang, AM Ghazi (2000) Characterization of the zinc binding activity of the rubella virus nonstrutural protease. J Virol 74(13): 5949- 5956.
- H Cai, Yu Zhang, Y Ma, Sun J, Liang X, et al. (2015) Zinc binding activity of human metapneumovirus M2-1 protein is indispensable for viral replication and pathogenesis in vivo. J Virol 89(12): 6391-6405.
- JA Soto, NMS Galver, FM Benavente, Pizarro-Ortega MS, Lay MK, et al (2018) Human metapneumovirus mechanism and molecular targets used by the virus to avoid the immune system. Front Immunol p. 9.
- CC Garcia, NA Candurra and EB Damonte (2000) Antiviral and virucidal activities against arenaviruses of zinc-finger active compounds. Antivir Chem Chemother 11(3): 231-238.
- D Femando Estrada, M Conner, SC St Jeor, Guzman RN (2011) The structure of the hantavirus zinc finger domain is conserved and represents the only natively folded region of the Gn cytoplasmic tail. Front Microbiol 2: 251.
- H-P Chiu, H Chiu, C-F Yang, Lee YL, Chiu FL, et al. (2018) Inhibition of Japanese encephalitis virus infection by the host zinc-finger antiviral protein. PLOS Pathog 14(7): 1-23.
- Nicholas J, PE Weerapana (2014) Zinc-binding cysteines: Diverse functions and structural motifs. Biomolecules 4: 419-434.
- B Fu, L Wang, S Li, M Dorf (2017) ZMPSTE24 defends against influenza and other pathogenic viruses. J Expe Med 214(1): 919-929.
- K Grant, L Grant, L Tong, C Boutell (2012) Depletion of intracellular zinc inhibits the ubiquitin ligase activity of viral regulatory protein ICP0 and restricts herpes simplex virus 1 replication in cell culture. J Virol 86(7): 4029-4033.
- V Zaslavsky (1979) Inhibition of vaccine virus growth by zinc ions: Effect on early RNA and thymidine kinase synthesis 29(1): 405-408.
- CL Parks, SE Witko, C Kotash, Lin SL, Sidhu MS, et al. (2005) Role of V protein RNA binding in inhibition of measles virus minigenome replication. Virology 348(1): 96-106.
- MJ Zilliox, G Parmigiani, DE Griffin (2006) Gene expression patterns in dendritic cells infected with measles virus compared with other pathogens. Proc Natl Acad Sci USA 103(9): 3363-3368.
- G Bujacz, J Alexandratos, A Wlodawer, Merkel G, Andrake M, et al. (1997) Binding of different divalent cations to the active site of avian Sarcoma virus integrase and their effects on enzymatic activity. J Biol Chem 272(29): 18161-18168.
- JS Buckman, WJ Bosche, RJ Gorelick (2003) Human immunodeficiency virus type 1 Nucleocapsid Zn2+ Fingers are required for efficient reverse transcription, initial integration processes, and protection of newly synthesized viral DNA. J Virol 77(2): 1469-1480.
- Indrani Paul, Jian Cui, Ernest L Maynard (2006) Zinc binding to the HCCH motif of HIV-1 virion infectivity factor induces a conformational change that mediates protein-protein interactions. Proc Natl Acad Sci USA 103(49): 18475-18480.
- TL Tellinghuisent, J Marcotrigiano, AE Gorbalenya (2004) The NS5A protein of hematitis C virus is a zinc metalloprotein. J Biol Chem 279(19): 48576-48587.
- NE Hellman, J Spector, J Robinson, Zuo X, Saunier S, et al. (2008) Matrix metalloproteinase 13(MMP13) and Tissue Inhibitor of matrix metalloproteinase 1(TIMP1), regulated by the MAPK pathway, are both necessary for madin-darby canine kidney tubulogenesis. J Biol Chem 283(7): 4272-4282.
- CF McGrath, JS Buckman, TD Gagliardi, Bosche WJ, Coren LV, et al. (32003) Human cellular nucleic acid-binding protein Zn2+ fingers support replication of human immunodeficiency virus type 1 when they are substituted in the nucleocapsid protein. J Virol 77(15): 8524-8531.
- N Geurts, G Opdenakker, PE Van den Steen (2012) Matrix metalloproteinases as therapeutic targets in protozoan parasitic infections. Pharmacol Ther 133: 257-279.
- MM Elalfy, MG El-hadity, MM Abouelmagd (2018) Update of usefulness and adverse effects of nanoparticles on animals and human health. Journal of Veterinary Medicine and Health.
- S Szunerits, A Barras, M Khanal, Q Pagneux (2015) Nanostructures for the inhibition of viral infections. Molecules 20: 15051-14081.
- MMN Yung, P-A Fougeres, Yu H Leung, F Liu, et al. (2017) Physicochemical characteristics and toxicity of surface-modified zinc oxide nanoparticles to freshwater and marine microalgae, Science Reports. 7: 1-14.
- Faten Farouk, Rania Ibrahim Shebl (2018) Comparing surface modifications of zinc oxide nanoparticles for modulating their antiviral activity against Herpes Simplex Virus type-1. International Journal of Nanoparticles and Nanotechnology 4: 1-14.
- Yi-Yun Kao, Yi-Chun Chen, Tsun-Jen Cheng, et al. (2012) Zinc oxide nanoparticles interfere with zinc ion homeostasis to cause cytotoxicity. Toxicol Sci 125(2): 462-472.
- A Scherzad, T Meyer, N Kleinsasser, S Hackenberg (2017) Molecular mechanisms of zinc oxide nanoparticle-induced genotoxicity short running title: Genotoxicity of ZnO NPs. Materials, 10(12).
- N Duggal, D Jaishankar, T Yadavalli, Hadigal S, Mishra YK, et al. (2017) Zinc oxide tetrapods inhibit herpes simplex virus infection of cultured corneas. Mol Vis 23: 26-38.
- TE Antoine, SR Hadigal, AM Yakoub, Mishra YK, Bhattacharya P, et al. (2016) Intravaginal zinc oxide tetrapod nanoparticles as novel immunoprotective agents against genital herpes. J Immunol 196(11): 4566-4575.




 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.