Research Article 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Current Practice of Warfarin Anticoagulation Therapy in China
*Corresponding author:Gislaine Aurelie Kemegne, Centre for Research on Medicinal Plants and Traditional Medicine, Institute of Medical Research and Medicinal Plants Studies, Yaoundé, Cameroon.
Received: February 12, 2024; Published: February 19, 2024
DOI: 10.34297/AJBSR.2024.21.002864
Abstract
Typhoid fever is a systematic infectious disease affecting sub-Saharan Africa and most especially Cameroon. A good number of her indigens turn towards alternative treatments (herbs/plants and their decoctions as a means of treatment) rather than modern treatments (standardized chemical molecules as means of treatment) against this infection due to their low economic statuses, which may be a leading cause of bacteria resistance for many. By reason of that, we aimed to determine the prevalence of bacterial pathogens from stools of typhoid fever patients under modern treatments on one hand and/or alternative treatment on the other hand in Yaoundé as well as assess their antibiogram profile. Stool samples were examined macroscopically and microscopically and subjected to culture. The suspected Salmonella spp on Salmonella-Shigella Agar were isolated and cultured in Triple Sugar Iron Agar and Motility Indole Urea Agar. After culture, biochemical testing and characterization was performed and the isolates were assessed for antimicrobial susceptibility. The therapeutic habits indicated that 61.4% of the participants used modern treatment, 3.4% used alternative treatment and 34.1% used both. Among pathogens identified, Citrobacter freundii was found in 71.4% of the samples, followed by 14.3% of Salmonella spp and Proteus mirabilis. The sensitivity of all tested isolates to cefixime, meropenem, ertapenem, azithromycin was 100% and they were resistant to ampicillin. More than 58% of patients under modern treatment declared to recover within weeks while only 33% under alternative treatment recovered in the same period. Nevertheless, no isolates positive in SS agar, TSI and MIU media were detected in stools from patients under alternative treatment.
Keywords: Antibiotics susceptibility, prevalence of bacterial population, Stool culture, Therapeutic habits, Typhoid fever
Introduction
Typhoid fever is a communicable disease and is still a major health concern worldwide, especially in Africa, Latin America, and South or Southeast Asia, where the disease is endemic. The fecal-oral route spreads the disease through contaminated food and water sources [1]. Typhoid fever is caused by the Gram-negative bacterium Salmonella enteric serovar typhi. The disease is mainly associated with low socioeconomic status and poor hygiene, with human beings the only known natural hosts and reservoir of infection [2]. According to WHO in 2018, there are between 11 to 21 million cases of typhoid fever and approximately 128 000 to 161 000 deaths registered annually worldwide with most cases occurring in South and South-East Asia and sub-Saharan Africa [3]. Therapeutic agents commonly used in the treatment of typhoid include ciprofloxacin, ceftriaxone, cefixime chloramphenicol, trimethoprim, sulfamethoxazole or ampicillin. In Cameroon, 3 out of 20 patients can afford to buy these drugs in hospitals and one out of every 1 000 patients may be able to see a specialist [4]. Treatment of typhoid fever may cost between US$4 and US$40 depending on the antibiotic used [5], hence the most considered primary healthcare system for 60% of children with high fever is the use of herbal medicines at home. Traditional healing plays an important role in African culture as it provides primary health care needs for the population [6].
Some studies have demonstrated the antimicrobial activity of some plant extracts against enterobacteria while others showed potentiation effects of the activity of some antibiotics on the same pathogen [7,8,9]. Again, some studies demonstrated that plant extracts reduce the antimicrobial activity of ciprofloxacin, the last therapeutic line against S. Typhimurium and S. Typhi [10]. Another study indicated that continuous exposure of Salmonella Serotype Typhi to some plant extracts traditionally used in Cameroon reduces the sensitivity of the strain to some antibiotics resulting to the development of antibiotic resistance [7]. The present study was hence undertaken to determine the prevalence of bacterial pathogens from stools of typhoid fever patients under modern and/or alternative treatment in Yaoundé and assess their antibiogram profile.
Materials and Methods
Study Design
This was a hospital based cross-sectional study conducted from April 2022 to August 2022 aiming to isolate Salmonella enteric serovar Typhi from stools of typhoid fever patients attending selected hospitals. Structured questionnaires were administered to patients and stool samples were collected.
Study Sites
The recruitment of patients was carried out using a stratified random sampling technique whereby, District Hospitals, District Medical Centres and Health Centres in Yaoundé were randomly sampled in areas with high frequency of potable water shortage and the willingness of the hospital management authorities to participate in the study. Among these were a total of three District Hospitals (Djoungolo, Efoulan, Biyem Assi), three District Medical Centres (Mvog-Ada, Mendong, Elig-Essono) and four Health Centres (Apsad, Djamtabo, Top Santé, Espoir). Fresh fecal samples were collected in sterile box and transferred to the laboratory of Pharmacology and Drug Discovery immediately.
Study Participants
Patients attending the selected hospitals that tested positive for typhoid fever based on clinical symptoms or Widal agglutination tests were recruited for the study.
Inclusion Criteria
Patients visiting the hospital at the time of the data collection, typhoid fever positive, of whom a stool culture was requested and who did give their consent.
Exclusion Criteria
Patients who did not give their consent and typhoid fever negative.
Sample size Calculation
The sample size of the study was calculated using the Lorentz formula [11] and the estimate (13.2%) of the prevalence (p) of Salmonella in Cameroon [12]. Normal distribution was anticipated with a 95%, confidence interval of statistical significance (a=0.05) and the degree (D) of precision level set to 0.05 for a 95% confidence interval.
Sampling
Fresh stool samples were collected (before treatment was administered) in sufficient amount in universal sterile (plastic) disposable box with screw cap, kept at 4°C until transported (as soon as collected) at the same temperature to the Laboratory of Pharmacology and Drug Discovery at the Institute of Medical Research and Medicinal Plant Studies, Yaoundé for analysis. Structured questionnaires were also administered to obtain socio-demographic information of each patient. To assess their recovery time, patients who agreed to follow-up were called once a month for 3 months to discuss symptom resolution.
Stool Culture, Isolation and Identification of Bacteria
All the samples were collected and processed within 2 h of collection. Macroscopic examination of the stool was carried out to record characteristics such as the appearance (presence of mucus, presence of blood, bloody mucus), the consistency (liquid, soft, pasty, hard) and the color (greenish, blackish, brownish). Isolation of Salmonella from stool samples was performed as recommended in the guidelines of the bacteriology analytic manual [13]. Briefly, from each fecal sample, 1 g was pre-enriched in 10 mL (1:10) of sterile buffered peptone water (Liofilchem®, Italy) and incubated at 37°C for 24 h. After that, for selective enrichment, 100 µL of a pre-enriched sample was transferred into 10 mL of Rappaport-Vassiliadis Soy (Liofilchem®, Italy) broth and incubated at 41.5±0.5°C for 24 h. Further, the culture was inoculated onto Salmonella-Shigella (TM MEDIA®, India) Agar plate and incubated for 24 h at 37°C. Three suspected Salmonella species colonies with black center were isolated and confirmed by biochemical examinations using Triple Sugar Iron (TSI) Agar (Liofilchem®, Italy) and Motility Indole Urea (MIU) Agar (Rapid Labs®, UK). Based on their biochemical characteristic on TSI (red slant, yellow butt, negative gas production and high production of H2S) and MIU (non-motile, negative indole production and negative urease activity) media, representative Salmonella isolates from each sample were selected for identification with the EnteroSystem 18R reagent (Liofilchem®, Italy).
Antibiotic Susceptibility Test
Antimicrobial susceptibility was performed by disc diffusion on Mueller Hinton agar (Rapid Labs®, UK) following the European Committee on Antimicrobial Susceptibility Testing and the Clinical Laboratory Standards Institute guidelines [14,15]. Escherichia coli ATCC 25922 was used as the quality control strain. Antibiotic discs were purchased from Rapid Labs®, UK. The antimicrobial agents used were Amoxicillin (AMC, 30 µg), Azithromycin (AT, 15 µg), Ciprofloxacin (CIP, 5 µg), Ofloxacin (OF, 5 µg), Co-trimoxazole (COT, 25 µg), Gentamicin (GEN, 10 µg), Cefixime (CFM, 5 µg), Ampicillin (AMP, 10 µg), Meropenem (MER, 10 µg), and Ertapenem (ETP, 10 µg).
Determination of MAR Index
Multiple Antibiotic Resistance (MAR) index was determined for the isolates against the tested antibiotics and calculated based on the formula MAR index= a/b where 'a' represent the number of antibiotics to which the isolate is resistant and 'b' the number of antibiotics tested [16].
Statistical Analysis
The data obtained in this study were entered in duplicates and analysed using Microsoft excel 2010 to conduct descriptive statistics, frequencies, and bivariate analyses (cross tabulation).
Results
Socio-Demographic and of Clinical Sign Presentations of the Participants
A hundred and eighty participants (typhoid fever patients) were enrolled in the study and interviewed. 24, 17 and 11 patients were included in the Djoungolo, Efoulan, and Biyem Assi district hospitals respectively. 17, 10 and 44 patients were included in the Mvog-Ada, Mendong, and Elig-Essono Disctrict Medical Centres respectively. 28, 20, 3 and 6 patients were included in the Apsad, Djamtabo, Top Santé, and Espoir Health Centres respectively. Among the participants, 119 patients (66.1%) were diagnosed according to clinical signs and symptoms as having typhoid fever while only 61 (33.8%) patients were selected because they were positive to the Widal test. Table 1 below represents the socio-demographic characteristics of typhoid fever patients interviewed from hospital in the city of Yaoundé. From this table, it can be observed that those consulting with fever and typhoid suspicion are mainly in the age range from 21-30 years. Those above 60 years were the least affected. Gender had no impact on the studied samples while among those interviewed, 58.9% had not receive primary education.
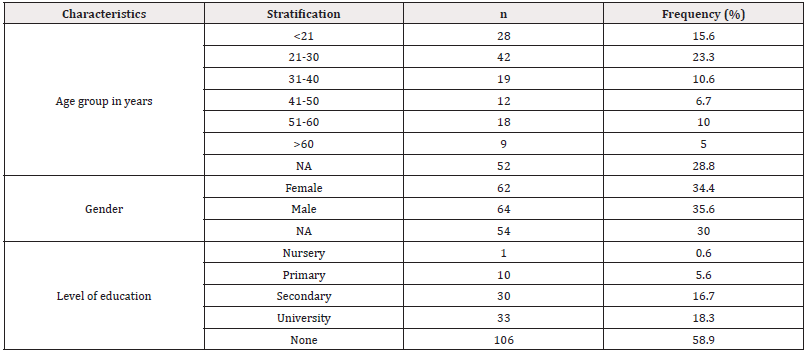
Table 1: Socio-demographic characteristic of participant in the city of Yaoundé.
Note*: NA: Not applicable, None: Prefer not to say, n: Number of participants.
Clinical signs and symptoms among study participants are reported in Figure 1. Among them, the most cited were fever (69, 38.3%) and headache (63, 35%) while the less cited were vomiting/diarrhea (23, 12.8%) and sweating (20, 11.1%). Other symptoms not related to typhoid fever were also cited allergy (1, 0.6%), gastric illness (1, 0.6%) and nausea (1, 0.6%) (Figure 1)
Table 2 shows the consistency and the color of collected stools. Approximately 35% were hard or pasty, 9% were liquid while 45.6 % were brownish, and 10% blackish.
Therapeutic Habits and Recovery Time of The Participants
Therapeutic habits and recovery time are presented in Table 3. The therapeutic habits indicate that most of the participants used modern treatments. Among 180 participants included, only 61 agreed to be follow-up for 3 months to record their recovering time. Of the 61 participants, the majority recovered within weeks of treatment.
When we cross the recovery time of the 61 patients with information on the therapeutic habits (Table 4), we globally notice that the only patient to have followed the parallel treatment was cured in one to three months. 59% of patients under modern treatment declared to recover within weeks while 33% under alternative treatment recovered in the same period.
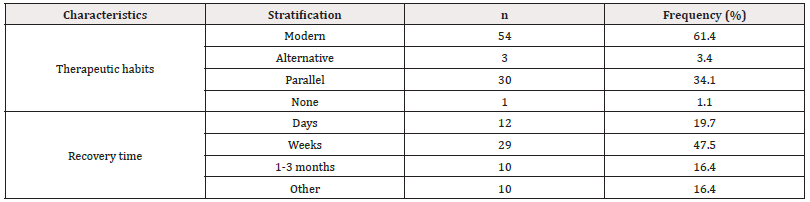
Table 3: Therapeutic habits and recovery time of participants in the city of Yaoundé.
Note*: n: Number of participants.

Table 4: Distribution of recovery time within each therapeutic habit.
Note*: NA: Not applicable, n: Number of participants.
Prevalence of Pathogens Isolates from Stools of Typhoid Fever Patients
All 180 stools collected in the hospitals were subjected to microbiological and biochemical analysis. Microbiologically, the samples were analyzed according to the reference protocol and selective medium used was SS medium. Out of the total samples, Shigella was detected in 92 samples (51%) and both Salmonella and Shigella were detected in 86 samples (47%). For each sample on Salmonella-Shigella medium, where there were black colonies, a maximum of 3 colonies, if possible, were recovered for further purification and identification.
Stools culture with salmonella shigella medium allowed storing 233 suspected Salmonella spp and 178 suspected Shigella spp isolates. Tables 5 and 6 show the reaction of the suspected Salmonella spp isolates on TSI (Triple Sugar Iron) and MIU (Motility Indole Urea) respectively.

Table 5: Number of isolates with positive (+) and negative (-) reactions on the Triple Sugar Iron Agar (TSI) medium.
Note*: S: Slant, B: Butt, Y: Yellow, R: Red, -: Negative, +: Positive, G: Gas, /: Unknow

Table 6: Number of isolates with positive (+) and negative (-) reactions on the Motility Indole Urea Agar (MIU) medium.
Note*: M: Motility, I: Indole production, U: Urease activity, -: Negative, +: Positive, pink: overall tube color change.
The EnteroSystem 18R identification gallery was used for the biochemical identification of 20 suspected Salmonella Typhi isolates (Table 7). The 20 isolates were selected based on TSI and MIU reactions for known Salmonella Typhi. The EnteroSystem 28R allowed the identification of 10 Citrobacter freundii, 2 Salmonella spp, 2 Proteus mirabilis, 1 Serratia liquefaciens, 1 Salmonella arizonae and 4 none completely identified isolates. Of the isolates obtained from the 61 patients agreed to be follow-up for 3 months, only 2 were identified from two patients with modern treatment as therapeutic habits; Proteus mirabilis (AP11/Sal2) and Citrobacter freundii (EF10/Sal2). Those patients were found cured within a few weeks.

Table 7: Isolated identified by using EnteroSystem 18R identification gallery.
Note*: /: Not identified.
In the same Table 7, it is noted that of the 20 isolates identified, 55% and 45% of them were from patients included in the study based on positive Widal test and clinical signs respectively. Only two Salmonella spp were identified and each belonged to participants diagnosed by the positive Widal test and by clinical signs.
Antimicrobial Susceptibility
The antibiotic susceptibility profiles of the isolates were classified as "Resistant", "Intermediate", and "Susceptible" (Table 8). The test of the 14 isolates against 10 different antibiotics was done by disc diffusion and underwent standardization and evaluation by the methods of the National Committee for the Clinical Laboratory Standards. Different classes of antibiotics were used, including penicillin (amoxicillin, ampicillin), sulfamides (co-trimoxazole), cephalosporine (cefixime), fluoroquinolones (ciprofloxacin, ofloxacin), carbapenem (meropenem, ertapenem), aminoglycoside (gentamicin), and macrolide (azithromycin). Escherichia coli ATCC 25922 was used as control.

Table 8: Antibiogram values of inhibition diameter (mm) for identified isolates.
Note*: AMC: Amoxicillin 30μg; COT: Co-trimoxazole 25μg; CFM: Cefixime 5μg; CIP: Ciprofloxacin 5μg; OF: Ofloxacine 5μg; MER: Meropenem 10μg; ETP: Ertapenem 10μg; GEN: Gentamicin 10μg; AMP: Ampicillin 10μg; AT: Azithromycin 15μg; E. coli: Escherichia coli ATCC 25922; MAR index: Multi-Antibiotic Resistant index; NA: Not applicable.
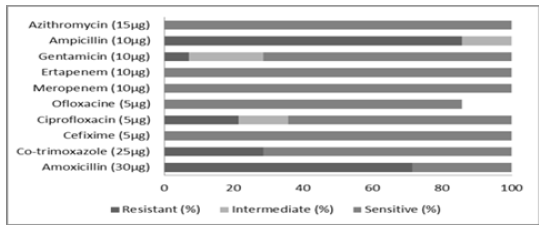
Figure 2 compares the sensitivities of isolates to the various antibiotics. The sensitivity of all tested isolates was most sensitive to cefixime, meropenem, ertapenem, and azithromycin followed by ofloxacine, co-trimoxazole, gentamicine, ciprofloxacin and lastly to amoxicillin. All the isolates were highly resistant to ampicillin and amoxicillin.
Discussion
Our study presented the prevalence of bacterial pathogens from stools of typhoid fever patients under modern treatments on one hand and/or alternative treatment on the other hand in Yaoundé as well as assess their antibiogram profile. After Salmonella Typhi, the pathogen causing the disease has been ingested and multiplied, the main clinical symptoms are fever and diarrhea or constipation [17]. Fluoroquinolones, especially ciprofloxacin, are commonly used as modern treatment resulting in the emergence and spread of multidrug-resistant strains [17]. In addition to modern therapy, patients also use alternative therapy based on medicinal plants with antibacterial effects. At the present, a study illustrated that the antidiarrheal activity of a Chinese herbal formula (composed of rhizomes of Panax ginseng and rhizomes of Atractylodes macrocephala) was associated with the gut microbial modulation [18].
Our data indicated that most of the participants were between 21 to 30 years old (23.33%). Gender did not impact the study population, but most patients were not educated. Most participants presented fever and headache as clinical signs and the stools are mostly brownish and hard or pasty. These results are slightly like that obtained in Douala, Littoral Region in Cameroon where the gender is quiet the same (50.6% females, 49.4% males) and 66.9% of fever is cited followed by headache [12]. The obtained results are different from that reported by numerous studies. Indeed, most patients enrolled in these studies had ages in the range of 10-29 [19], 11-20 [20] and above 20 [5]. Of the total participants, the majority were students [20] and had attained secondary education [5,12]. Abdominal pain is the clinical symptom cited by the participants [20,21]. Stool samples were mostly soft in a retrospective and prospective study [22]. The age range in our population, combined with their low education level may suggest they are independent street workers or unemployed and are more exposed to in between meals and street food as indicated in a study [23].
The bacteria were isolated from adults between 21 to 60 years old. Concerning stool analysis, based on their morphological and biochemical characteristic, 14 presumptive isolates were identified with a prevalence of 71% for Citrobacter freundii and 14% for Salmonella spp and Proteus mirabilis. These results are lower than that obtained in Douala Cameroon (49.4%) [12], in the Northwest Region of Cameroon (42.9%) [19], in Saint Elisabeth General Hospital Shisong Cameroon (30.2%) [11] but higher to that obtained in Bamenda Cameroon (8.70%) and in the Menoua Division of Cameroon (10.4% and 12.90%) [20,21].
The overall sensitivities of isolates to the various antibiotics showed that the isolates were highly susceptible to cefixime, meropenem, ertapenem and azithromycin. With regards to ciprofloxacin, the fluoroquinolones used as the first line drugs against typhoid fever, 21.43% and 14.23% of isolates tested were classified respectively as resistant and intermediate. This differs from the findings who had a susceptible percentage to ciprofloxacin of 52.38% and a high resistance percentage with co-trimoxazole and amoxicillin (100%) [24]. The loss of sensitivity of pathogenic bacteria to the most used fluoroquinolones in general and ciprofloxacin in particular for the treatment of bacterial diseases could be the cause of the treatment failure in suffering patients.
The therapeutic habits indicate that of the total participants, 61.36% used modern treatment, 3.41% used alternative treatment and 34.09% used both (parallel treatment). This is like earlier studies where patients in Ngaoundéré (Cameroon) used modern or traditional medicine or both in regards of factors like accessibility, cost, ability to treat, accuracy of diagnosis and treatment, and the capacity of understanding the patients’ culture [25]. The obtained percentage is a little higher than that reported where among patients in three Cameroonian villages (Nyambaka in Adamaoua, Zoetele in the South and Massaka in the Southwest Region), 55% used modern treatment, 33% consulted healers for traditional treatment and 12% stayed at home taking herbs or waiting for symptoms to stop [26]. In an ethnobotanical survey, numerous plant species were found to be traditionally used against typhoid fever in the Bamboutos Division of the West Region of Cameroon [27]. Elderly persons and traditional healers cited 59 medicinal plants species mostly harvested from the wild. Leaves were the most frequently medicinal plant parts used, shrubs the primary source of plants and decoction the most common drug preparation method. A study conducted within the population of the Bamenda Grassfield in Cameroon revealed that this population used its own traditional medicinal treatment methods. Surprisingly, the coming of the Hausa-Faulani traditional healing methods has had a huge impact on the treatment methods of the local population and is gradually replacing the indigenous methods of treatment in the Bamenda Grassfields [28]. To compare the interaction gap between traditional healers and western medicine with their patients, a study performed in the semi-rural health district of Mfou in Cameroon contradicts the idea that rural Africans do not have access to western medicine due to lack of money. In fact, the use of more patient-centred communication style is a relevant factor that drives population to traditional healers compare to western providers in Cameroon [29].
Conclusion
This work has revealed that among the typhoid fever patients aged between 21 to 60 years old, the modern treatment is the most therapeutic habits and resulted in health recovery within days. Among the identified bacteria, Citrobacter freundii, Salmonella spp and Proteus mirabilis had the highest prevalence. The isolates presented low and zero susceptibility to amoxicillin and ampicillin belonging to the class of penicillin. Despite typhoid fever epidemic, low prevalence of Salmonella Typhi was showed independently of the recruitment criteria for the participants in the study, being based on clinical symptoms or Widal agglutination tests. Finally, more than 58% of patients under modern treatment declared to recover within weeks while only 33% under alternative treatment recovered in the same period. Nevertheless, no isolates positive in SS agar, TSI and MIU media were detected in stools from patients under alternative treatment.
Author Contribution Statement
This work was carried out in collaboration among all authors. GAK and SLSK conceptualized the study design. GAK, FKM and GNN carried out the data and stool samples collection in the field. GAK did the bench work and wrote the manuscript draft. GAK, SLSK and GAA performed the analysis and the interpretation of data. GAK, SLSK and GAA contributed to writing and editing the manuscript. All the authors read and approved the final version of the manuscript.
Acknowledgments
The authors are grateful to all participants who sacrificed their time and donated stool samples for this study. They also wish to thank doctors and laboratory technicians of different hospitals for their contributions.
Funding
This work was supported by The Royal Society of Tropical Medicine and Hygiene, UK (id. 19974380) through the RSTMH Small Grants 2021. The contents are solely the responsibility of the authors and do not necessarily represent the official views of the funders. The funders had no role in study design, data collection and analysis, or decision to publish, or preparation of the manuscript.
Conflict of Interest Statement
The authors declare no conflicts of interest.
Ethical Approval
All the procedures have been carried out in accordance with the guidelines laid down by the National Committee on Ethics in Research for Human Health (N°2022/02/1437/CE/CNERSH/SP) and in accordance with local laws and regulations (Centre Regional Delegation of the Ministry of Public Health, N°E-0246/AAR/MINSANTE/SG/DRSPC). The administrative authorization was obtained from the selected Hospitals administration. Issues about the autonomy, protection, and confidentiality of the subjects were fully respected.
Data Availability Statement
The data supporting this study’s findings are available on request from the corresponding author.
References
- Akwa T, Nguimbous S (2021) Investigation of Typhoid Fever and their Associated Risk Factors in Children Attending “Deo Gratias” Hospital in Douala. Eur J Med Edu Technol 14 (2): 2107-2107.
- Mweu E, English M (2008) Typhoid fever in children in Africa. Trop Med Int Health 13(4): 532-40.
- World Health Organization (2018) Typhoid and other invasive salmonellosis. Available from https://www.who.int/immunization/monitoring_surveillance/burden/vpd/WHO_SurveillanceVaccinePreventable_21_Typhoidua=1.
- Kuete V, Efferth T (2010) Cameroonian medicinal plants: pharmacology and derived natural products. Front Pharmacol 1(123): 1-19.
- Nsutebu EF, Martins P, Adiogo D (2003) Prevalence of typhoid fever in febrile patients with symptoms clinically compatible with typhoid fever in Cameroon. Trop Med Int Health 8(6): 575-8.
- Tsobou R, Mapongmetsem P-M, Voukeng I, Van DP (2015) Phytochemical screening and antibacterial activity of medicinal plants used to treat typhoid fever in Bamboutos division, west Cameroon. J Appl Pharm Sci 5(06): 34-49.
- Ezo’o MF, Tchonang SC, Kemaleu HL, Sado KSL, Essia NJJ (2018) Exposure to plant extract causes the variation of antibiotic susceptibility of two bacterial strains (Salmonella Serotype Typhi and Staphylococcus aureus). J Adv Microbiol 12(2): 1-14.
- Kemegne GA, Nyegue MA, Sado SLK, Etoa F-X, Menut Chantal (2018) Mangifera indica bark essential oil: chemical composition and biological activities in comparison with aqueous and ethanol extracts. Nat Prod Commun 13(7): 903-6.
- Kemegne GA, Sado SLK, Nyegue MA, Menut C, Etoa F-X (2021) Comparing checkerboard, isobologram and CCD methods for drug combination: a case study of ciprofloxacin and plant extracts on Escherichia coli and Shigella. J Med Plant Res 5(10): 479-89.
- Marathe SA, Kumar R, Ajitkumar P, Nagaraja V, Chakravortty D (2013) Curcumin reduces the antimicrobial activity of ciprofloxacin against Salomonella Typhimurium and Salmonella Typhi. J Antimicrob Chemother 68(1): 139-52.
- Njoya HF, Awolu MM, Christopher TB, Duclerc JF, Ateudjieu J et al. (2021) Prevalence and awareness of mode of transmission of typhoid fever in patients diagnosed with Salmonella typhi and paratyphi infections at the Saint Elisabeth General Hospital Shisong, Bui Division, Cameroon. Pan Afr Med J 40: 83.
- Njolle AB, Tientche B, Asaah S, Forfuet FD, Kamga FHL, et al. (2020) The prevalence of salmonellosis in patients with malaria attending an urban hospital in Douala, Littoral Region, Cameroon. J Adv Med Med Res 32(2): 32-45.
- Andrews WH, Jacobson A, Hammack T (2007) Salmonella. In: Bacteriological Analytical Manual. 8th ed., Ch. 4. Revision A. Available from https://www.fda.gov/files/food/published/BAM-Chapter-5-Salmonella-and-Appendix-1--Rapid-Methods-ù28June-2009%29-Incorporated--by-Reference-in-in-21-CFR-parts-16-and-118.
- European Commitee for Antimicrobial Susceptibility Testing (CASFM/EUCAST). Société Française de Microbiologie (2020) Tableau des concentrations critiques pour l’interprétation des CMI et des diamètres critiques des zones d’inhibition. In : CASFM / EUCAST : Société Française de Microbiologie Ed p.36-45.
- Clinical and Laboratory Standards Institute (2007) Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. 9thed. Approved standard 32: 1-68.
- Krumperman PH (1983) Multiple antibiotic resistance indexing Escherichia coli to identify risk sources of faecal contamination of foods. Appl Environ Microbiol 46: 165-70.
- Anwar E, Goldberg E, Fraser A, Acosta CJ, Paul M, et al. (2014) Vaccines for preventing typhoid fever. Cochrane Database Syst Rev 1: CD001261.
- Wang J, Feng W, Zhang S, Chen L, Tang F, et al. (2019) Gut microbial modulation in the treatment of chemotherapy-induced diarrhea with Shenzhu Capsule. BMC Complement Altern Med 19(1):126.
- Wam EC, Arrey CN, Sama LF, Agyingi LA, Wam AN, et al. (2019) Comparative study on the use of Widal test to stool culture in the laboratory diagnosis of typhoid fever in Holy Family Hospital Akum, Northwest Region of Cameroon. Open Microbiol J 13: 73-80.
- Ousenu K, Ali IM, Sama LF, Ndam MN, Tchouangueu TF, et al. (2021) A cross-sectional comparative study of the performance of the Widal test and the Typhidot immunoassay for typhoid fever diagnosis in the West Region of Cameroon. Can J Infect Dis Med Microbial 2021:8279122.
- Ousenu K, Sama LF, Ali IM, Fonbah JL, Nadine OS, et al. (2021) Aetiology and risk factors of bacterial gastroenteritis among febrile outpatients at the Dschang District Hospital, West Region of Cameroon: a cross-sectional study. BMJ Open 11(9): e045965.
- Fodouop SPC, Gingir B, Nodem SFS, Yaou FM, Roselyne TN, et al (2021) Water-related diseases in the Adamawa Region, Cameroon: a prospective and retrospective case study and the susceptibility of isolates bacteria to common antibiotics. J Adv Microbiol 21(7): 75-85.
- Dapi LN, Nouedoui C, Janlert U, Haglin L (2005) Adolescents’ food habits and nutritional status in urban and rural areas in Cameroon, Africa. Scand J Nutr 49: 151-158.
- Awung N, Kengne M, Nwobegahay MJ, Ter GD, Njukeng PA, et al. (2016) The susceptibility pattern of Salmonella species to commonly used antibiotics in the Bamenda District Health area, Cameroon. Afr J Path Microbiol 5: 4.
- Hardy A (2008) The integration of traditional and western medicine in Ngaoundéré, Cameroon. SIT Cameroon Fall, Wesleyan University ISP Topic Codes: 701, 708, 722.
- Nkwi PN (1994) Perceptions and treatment of diarrhoeal diseases in Cameroon. J Diarrhoel Dis Res 12(1): 35-41.
- Tsobou R, Mapongmetsem PM, Van DP (2023) Medicinal plants used against typhoid fever in Bamboutos Division, Western Cameroon. Ethnobot Res Appl 11: 163-74.
- Amadou JM (2019) The impact of Hausa-Fulani traditional medicine and healing method on the indigenous peoples of the Bamenda grassfields. Int J Adv Sci Res Eng 5(7): 86-94.
- Labhardt ND, Aboa SM, Manga E, Bensing JM, Langewitz W, et al. (2020) Bridging the gap: how traditional healers interact with their patients. A comparative study in Cameroon. Trop Med Int Health 15(9): 1099-1108.





 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.