Research Article 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Towards Cardiac Regeneration: The Promising Future of The Unique Translational Tool to Manage Cardiac Self-Renewal and Regeneration to Secure a Safety of The Post-Infarction Period
*Corresponding author:Sergey Suchkov, The Russian Academy of Natural Sciences (RANS), Moscow, Russia.
Received: January 22, 2024; Published: February 05, 2024
DOI: 10.34297/AJBSR.2024.21.002848
Abstract
Cardiovascular disease is the leading cause of death, with 80% of cases occurring in developing countries. Innovative therapies are required to reduce mortality and limit or abolish the necessity for cardiac transplantation. Over the last decade, stem cells have been a promise for the cure of several diseases not only due to their plasticity but also to their capacity to act in a paracrine manner and influence the affected tissue. Human SC-based therapy derivatives are extremely attractive for therapeutic development because they have direct pharmacologic utility in clinical applications, unlike any other adult cells. Moreover, stem cell-derived paracrine factors have been shown to suppress inflammation and apoptosis, stimulate angiogenesis, and amplify the proliferation and differentiation of resident Cardiac Stem Cells (CSCs). And SC therapies are thus viable alternatives to conventional treatments with substantial therapeutic potential; market opportunities are huge, as multiple product candidates are expected to be approved over the coming decade.
Keywords: Cardiac regeneration, Cardiac stem cells, Pluripotent stem cells, Cardiac Scs, Personalized and Precision Medicine (PPM)
Introduction
The ultimate goal for regenerative medicine is to channel multipotent human cells with high proliferative capacity into pre-defined scenarios and specified differentiation programs within the human body. Current therapies do not address the underlying pathophysiology of this disease, namely, the progressive loss of functional cardiomyocytes. The notion of repairing or regenerating lost myocardium via cell-based therapies remains highly appealing.
The recent identification of adult stem cells, including both cardiac stem/progenitor cells, bone marrow stem cells and others (Figure 1),
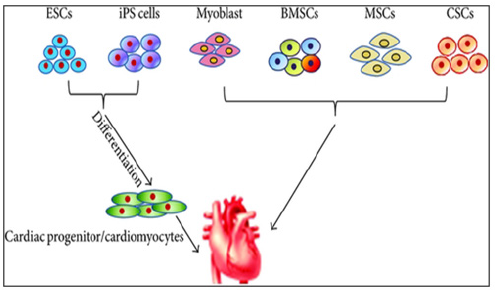
Figure 1: Various Types of Scs Used in Cardiac Regenerative Therapy
PSCs, including the ESCs and iPSCs, are generally differentiated into CMs for heart failure therapy
HSCs, hematopoietic stem cell; iPSCs, induced pluripotent stem cells; BMSC, bone marrow stem cells; MSC, mesenchymal stem cell; CSC, cardiac stem cell; CM, cardiac cell.
Source: Afjeh-Dana, Elham & Naserzadeh, Parvaneh & Moradi, Elham & Hosseini, Nasrin & Seifalian, Alexander & Ashtari, Behnaz. (2022). Stem Cell Differentiation into Cardiomyocytes: Current Methods and Emerging Approaches. Stem Cell Reviews and Reports. 18. 1-27.
has triggered an explosive interest in using these cells for physiologically relevant cardio myogenesis.
Unfortunately, the efficacy of cell-based therapies is somewhat limited by their poor long-term viability, homing, and engraftment to the myocardium. In response, a range of novel SC-based technologies are in development to provide additional cellular modalities, bringing CTs a step closer to the clinic. Enthusiasm for cardiac regeneration via cell therapy has further been fueled by the many encouraging reports in both animals and human studies. Further intensive re-search in basic science, Biodesigndriven translational applications and clinical arenas are needed to make this next great frontier in cardiovascular regenerative medicine a reality.
Meanwhile, Stem Cell (SC)-based therapy has been considered as a promising option in the treatment of ischemic heart disease (Figure 2).
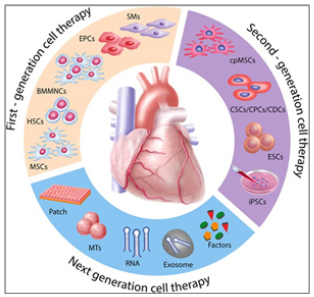
Figure 2: Evolution of Translational Cell-Driven Cardiac Regenerative Therapies
First-generation cell types such as SMs, BMMNCs, HSCs, EPCs, and MSCs demonstrated feasibility and safety with, however, heterogeneous outcomes and limited efficacy in the clinical setting. In order to better match the target organ, second-generation cell therapies propose the use of cpMSCs, CSCs/CPCs, and CDCs, and pluripotent stem cells such as ESCs and iPSCs. Next-generation therapies for cardiac repair are directed toward cell en-hancement (e.g., biomaterials, 3D cell constructs, cytokines, miRNAs) and cell-free concepts (e.g., growth factors, non-coding RNAs, extracellular vesi-cles, and direct reprograming
SMs, skeletal myoblasts; BMMNCs, bone marrow mononuclear cells; HSCs, hematopoietic stem cell; EPCs, endothelial progenitor cell; MSCs, mesenchy-mal stem cells; cpMSCs, chorionic plate derived mesenchymal stem cells; CSCs/CPCs, cardiac stem cell:cardiac progenitor cell ration; CDCs, cardiac dendritic cell; ESCs, embryonic stem cells; iPSCs, induced pluripotent stem cell; RNA, ribonucleic acid;
Source: Cambria, E., Pasqualini, F.S., Wolint, P. et al. Translational cardiac stem cell therapy: advancing from first-generation to next-generation cell types. npj Regen Med 2, 17 (2017). https://doi.org/10.1038/s41536-017-0024-1.
Originally, SC transplantation has emerged as a promising therapeutic strategy for acute and chronic ischemic cardiomyopathy. Regarding the current status of SC therapies for patients with myocardial infarction is critically being discussed now, describing:
the current status of clinical trials of human pluripotent SCs (hPSCs) compared with clinical trials of human adult or fetal stem cells;
the gap between fundamental research and translational application of human SCs;
the use of biomaterials in clinical and pre-clinical studies of SCs; and
trends in design-driven bioengineering to promote SC therapies for patients with myocardial infarction [1,2].
Skeletal Myoblasts (Sms): The Pioneering Steps
Due to the outcome results and findings summarized, initial studies were encouraging, with SMs participating at heart muscle formation, and then were rapidly translated into the clinics with Phase I trials. However, because of the in-creased risk of ventricular arrhythmias potentially due to missing junctional proteins, the trials were stopped. The risk of ventricular arrhythmias is relevant now that pluripotent cell-derived cardiomyocytes aim at re-attempting heart remuscularization [3,4].
Bone Marrow (BM)-Derived Cells in Trials
Differentiation of BM Mononuclear Cells (BMMNCs) into cardiomyocytes was observed but was criticized later [5]. Regarding HSCs, the researchers found no evidence of myocardial differentiation of CD34+ HSCs in preclinical models [6]. The clinical trials did not find significant beneficial effects of cell therapy [7,8].
Mesenchymal Stem Cells (Mscs) In Trials
MSCs fulfill many of the criteria for an ideal adult SC for regenerative therapy for a variety of conditions. Derived from the stromal fraction of bone marrow, MSCs can be readily isolated and identified on the basis of growth and expansion on plastic surfaces. MSCs are easily grown and expanded having multi-lineage potential and appearing to exhibit immunological tolerance.
MSCs have repeatedly been suggested as an immuno privileged and therefore valuable clinical cell source for cardiac repair. MSCs are multipotent stromal cells that are mainly characterized by their adherence properties, a distinct surface marker profile composed of specific markers, paracrine signaling, and the ability to differentiate into the adaptogenic, chondrogenic and osteogenic lineage.
A subset of MSCs can also differentiate into cardiomyocytes under specific conditions in vitro, and thus due to the preclinical trials, adipose tissue-derived and BM-derived MSCs represent an auspicious cell source with therapeutic potential for cardiac repair, and were promising in numerous preclinical trials [9,10]. Instead of the latter, approaches using MSCs in clinical trials, are studied with promising results, but their efficacy needs to be further validated [11-13]. Second-generation therapies were aiming at orienting non-resident SCs, such as MSCs and PSCs (Pluripotent Stem Cells), toward cardiac differentiation. Only few studies compared first-generation and second-generation cell types, which found that cardiac-committed cells dis-played an improved therapeutic effect as assessed by improved engraftment, cardiac function, angiogenesis, and scar size [14-16]. Due to their enormous expansion rate and immunomodulatory properties, MSCs are con-sidered as a potential candidate for both autologous and allogeneic SC-driven therapy with pre-ventive, prophylactic and rehabilitative effects.
Cardiopoietic Mscs (Cpmscs) In Trials
The use of cpMSCs in preclinical trials with chronic ischemic disorders has shown therapeutic benefit, but the latter needs to be validated in long-term studies [17,18]. To date, clinical success of cardiac cell-therapies (including cpMSCs-driven one) remains limited. To enhance the cardio-reparative properties of SCs, the concept of lineage-specification through cardiopoietic-guidance has been recently suggested. However, so far, only results from preclinical studies and from a clinical pilot-trial in chronic heart-failure are available, while sys-tematic evidence of its therapeutic-efficacy is still lacking [19].
Cardiac Stem/Progenitor Cells (Cscs/Cpcs) For the Trials
Being multipotent, clonogenic, and expressing SC markers, are usually derived directly from biopsies of the target organ, and therefore ensure a perfect match and can inhibit cardiomyocytes apoptosis [20]. Due to the pre-clinical studies, c-kit+ CSCs/CPCs could differentiate into cardiomyocytes, suggesting that this phenomenon occurs at a purported functionally insignificant rate [21-24].
Pluripotent Scs (Pscs) In Trials
PSCs, including Embryonic Stem Cells (ESCs) and InDuced Pluripotent Stem Cells (iPSCs), constitute another source for guided cardiac differentiation (Figure 3).
The most primitive of all SC populations are the ESCs that develop as the inner cell mass at day 5 after fertilization in the human blastocyst. When isolated and transferred to appropriate culture media, ESCs can undergo an undetermined number of cell doublings while retaining the capacity to differentiate into specific cell types, including cardiomyocytes. Common teaching suggests that SCs emerging during late embryonic and fetal development are restricted to the production of tissue-specific cell types. And although SCs continue to self-renew in adult life, their ability to differentiate is limited to the tissue in which they reside, retaining a high degree of developmental plasticity. The latter would open a way to enter a revolutionary period in SC biology and regenerative medicine.

Figure 3: Types Of Adults (Multipotent) And Pluripo-Tent Stem Cells For Heart Regeneration
Strategies that improve therapeutic efficacy as well as cellular survival and engraftment, such as modified stem cells, mixed stem cells and cardiac tissue engineer-ing, are under development. Techniques that can induce trans-differentiation of somatic cells directly into func-tional cardiomyocytes in vitro and in vivo have also been developed recently, and may be proven to be a more effective strategy for promoting endogenous heart regeneration rather than transplanting exogenous SCs in the future.
PSCs, pluripotent stem cells; iPSCs, induced pluripotent stem cells; ESC, embryonic stem cell; HSCs, hemato-poietic stem cells; EPCs, endothelial progenitor cells; MSCs, mesenchymal stem cells; CPSs, cardiac progeni-tor cells.
The source: Liao, SY., Tse, HF. Multipotent (adult) and pluripotent stem cells for heart regeneration: what are the pros and cons?. Stem Cell Res Ther 4, 151 (2013).
Meanwhile, Human Pscs (hPSCs) are abundant sources of cardiomyocytes for cell re-placement therapy and other applications such as disease modeling, drug discovery and cardio-toxicity screening [25]. And hPSC-derived cardiomyocytes have thus attracted attention as an unlimited source of cells for cardiac therapies to attract the strategy for cardiomyocyte production via the biphasic, involving hPSC expansion to generate adequate cell numbers followed by differentiation to cardiomyocytes for specific applications [26]. Preclinical studies with PSCs yielded mixed results depending on the animal model. But in most cases, human ESC-derived CPCs have been transplanted into animal models of MI, showing improved cardiac function, thus leading to clinical trials.
PSCs represent an appealing source from which to develop cell replacement therapies (Figure 4).
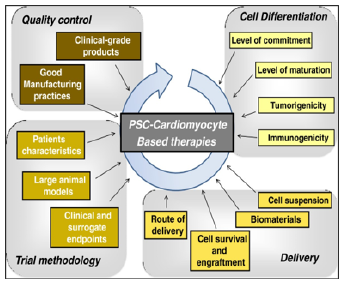
Figure 4: Considerations For Pre-Clinical Models and Clinical Tri-Als of Pluripotent Stem Cell-Derived Cardiomyocytes
The main questions and issues that should be considered before translating PSC-derived cardiomyocytes into clinical investigations. ESC/iPSC technologies have several specific issues that remain to be overcome, including the development of efficient protocols to generate pure populations of cardiomyo-cytes, and the development of techniques to improve the retention and survival rate of transplanted cells. Once these are overcome, the first clinical trials will then be on the horizon.
PSCs, pluripotent stem cells; iPSCs, induced pluripotent stem cells; ESC, embryonic stem cell.
The source: Hulot, JS., Stillitano, F., Salem, J.E. et al. Considera-tions for pre-clinical models and clinical trials of pluripotent stem cell-derived cardiomyocytes. Stem Cell Res Ther 5, 1 (2014).
been launched to promote their development toward clinical applications including stages before translating PSC-derived cardiomyocytes into clinical investigations, including the development of good manufacturing practice-level PSC lines, the development of efficient protocols to generate pure populations of cardiac myocytes, and the development of techniques to improve the retention and survival rate of transplanted cells [27].
SC-based therapies represent a possible paradigm shift for cardiac repair. However, most of the first-generation approaches displayed heterogeneous clinical outcomes regarding efficacy. Stemming from the desire to closely match the target organ, second-generation cell types were introduced and rapidly moved from bench to bedside. Unfortunately, debates remain around the benefit of SC therapy, optimal trial design parameters, and the ideal cell type [17].
Having the above-mentioned summarized and assessed, we might mention that cell (SC-driven) therapy holds potential to tackle myocardial infarction and heart failure. But the key is-sues such as the cell type, cell number, delivery route, timing, follow-up periods, and endpoints remain unsolved. Meanwhile, the field has rapidly evolved to address in particular the ideal cell type and does require to get the problems be surmounted to secure the full potential of cell therapy be realized in the near future to come.
Toward Cardiac Regeneration: Combination of PSC-Based Therapies and Design-driven Bioengineering Strategies
The potential to repair cardiac tissue by cell grafting has attracted the attention of the field, due to the scarce capacity of CMs to proliferate and replace the damaged tissue. The dispute over the existence of cardiac progenitors in the adult heart is still unsolved; on the same trail, the ability of immature cardiac progenitor cells to engraft with pre-existing CMs in vivo has not been irrefutably determined. Due to their unique capacity to produce functional CMs, a wide variety of cells have been evaluated for therapeutic delivery, including BMCs, MSCs, and endogenous CSCs as the most promising sources for cardiac regenerative medicine application. But despite the encouraging results from these and other in vivo studies on the potential beneficial effects of iPSC-based cell therapy for heart failure, preclinical experiments, using CMs derived from human iPSCs, have given controversial results [28,29]. Meanwhile, despite the fact that many different types of SCs and derivatives have been proposed as valuable candidates for regeneration of the myocardium, a general consensus on which cell type should be considered the gold standard for cell replacement therapy of the heart, remains elusive.
The problem is that the majority of preclinical studies have been conducted in 2-D culture systems, a condition that does not take into account the multiple interactions occurring in a 3-D structure, like the heart: this is among the major issues still limiting the application of cell therapy approaches into the clinics. Thus, in the last years, many productive studies have directed their efforts for the development of a 3D structure able to functionally resemble the cardiac tissue and suitable for heart transplantation.
3D Culture Systems as Applicable to Manage Cardiac Regeneration
In particular, the cardiac tissue is composed by contractile and non-contractile cells, that are organized as a complex 3-D structure (spheroids and organoids both referring to 3D culture systems with a specialized architecture and cell organization that typically form through self-assembling processes), which acting as tightly regulated interaction among these diverse cell types, plays a central role in the heart’s function. Thus, 3D cell cultures are emerging as a new tool for both targeted drug discovery and regenerative medicine applications, prompting and boosting the development of 3D cellular systems, as applicable to the functional features of cardiac tissue. However, those systems were found to be not ideal for developing cardiac cells for cell therapy.
The genuine true is that engineered cardiac tissue models have recently emerged as prom-ising approaches to repair damaged cardiac tissue as well as suitable platforms for drug/toxicity testing and disease modeling (MacNeil, 2007). Meanwhile, the improvement of protocols for differentiation of PSCs toward the cardiac lineage made the production of large amounts of human CMs feasible, bursting the development of human heart surrogates for disease modeling, drug testing and autologous cell therapy applications. On this regard, embedding CMs derived from PSCs into engineered structures has been shown to positively impact cell maturation to different extents, depending on the specific characteristics of the generated 3D-constructs. In fact, use of both human iPSCs and ESCs deals with an important issue of cell maturation following cardiac induction in vitro [30-33]. Thus, altogether these studies strongly indicate that the above-mentioned constructs are excellent supports for maturation of CMs derived from human PSCs, since they provide a physiological-like environment and stimuli to cells. And, moreover, altogether those studies showed that cardiac engineered constructs enhance intercellular organization and cross-talk of iPSC-CMs, improving their maturation in vitro, and ameliorate cardiac performance after the transplantation in vivo, thus providing encouraging evidences for their future use to achieve cardiac regeneration in humans.
A significant number of different cell therapy strategies that employ iPSC-CMs have been proposed to restore cardiac function, ranging from injection of cells alone, to their combination with ECM-like biomaterials, and to more complicated engineered-based strategies, which aim to faithfully recreate the myocardium. Although different types of stem cells have been proposed as potential candidates for cardiac regenerative medicine, there is no consensus about which represents the best choice. Given the complexity of cardiac tissue, which is composed by diverse cell types, human PSCs – mostly iPSCs – are certainly among the most relevant sources. In particular, CMs differentiated from iPSC may represent a realistic option for the regeneration of the injured heart (Figure 5).
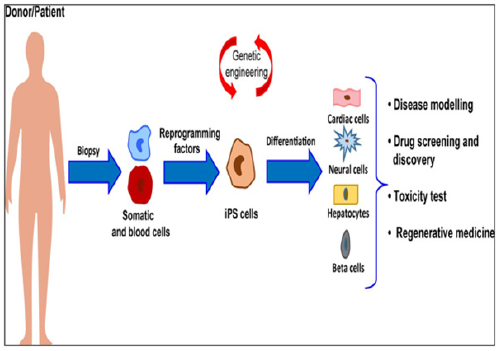
Figure 5: Schematic Representation of The Workflow for Ipsc Generation and Differentiation From Patients-Related Somatic Cells and Major Applications to Human Health
The ability of the heart to recover after ischemic injury depends on several molecular and cellular pathways, and the imbalance between them results into adverse remodeling, culminating in heart failure. In this complex scenario, regenerative medicine has been opened with the discovery of iPSCs, which share the same characteristic of ESCs, but are generated from patient-specific somatic cells, providing an autologous source of human cells. Similarly, iPSCs are able to efficiently differentiate into cardiomyo-cytes (CMs), and thus hold a real regenerative potential for future clinical applications. However, cell-based therapies are subject-ed to poor grafting and may cause adverse effects in the failing heart. Thus, design-inspired bioengineering technologies focused their attention on the improvement of both survival and functionality of iPSC-derived CMs. The combination of these two fields of study has burst the development of cell-based three-dimensional (3D) structures and organoids which mimic, more realistical-ly, the in vivo cell behavior. Toward the same path, the possibility to directly induce conversion of fibroblasts into CMs has recently emerged as a promising area for in situ cardiac regeneration. So, the field would strongly need the advancements in the application of iPSCs and tissue-engineering for therapeutically relevant cardiac regenerative approaches, aiming to highlight out-comes and future perspectives for their clinical translation.
iPSCs, induced pluripotent stem cells; ESCs, embryonic stem cells; CMs, cardiomyocytes.
The source: Mazzola M and Di Pasquale E (2020) Toward Cardiac Regeneration: Combination of Pluripotent Stem Cell-Based Therapies and Bioengineering Strategies. Front. Bioeng. Biotechnol. 8:455. doi: 10.3389/fbioe.2020.00455.
The majority of these approaches have shown - at least to some extent - success in mimicking heart cells and improving cardiac function, providing new hope for the replacement of CMs after the irreversible loss of heart tissue occurring during myocardial infarction (Figure 6).
The progress in the field made by tissue engineering technologies has been encouraging and thus opened ways to create bio-supports able to incorporate the cells needed for the regeneration of cardiac tissue. However, significant improvements are still mandatory before we could consider their routine use in the clinical field for treatment of patients suffering from myocardial diseases. These improvements mainly regard scaffold production, the interactions between cells and the ECM and vascular integration with the host, hopefully opening new doors for cardiac regeneration therapy via tissue engineering strategies. However, despite the promising results obtained by preclinical research, the approach is still rather immature for its clinical translation and further investigations are needed to develop effective and more efficient strategies to achieve cardiac regeneration in vivo.
Engineered Cardiac Tissue Constructs to Be Translated into Clinical Applications
No comments, but design-driven tissue engineering strategies are probably a more suitable option. And given the limited regenerative capacity of the heart, the design-driven bio-engineered scaffolds and tissue engineering approaches are thus ideal for cardiovascular regenerative medicine applications. The main goal is to create a cardiac graft which can be implanted and restore the functionality of the myocardium without major side effects. To this aim, tissue engineering intends to recreate the microenvironment of the cardiac tissue, in terms of cell composition, stiffness, geometry, physical and electrical stimuli and, extracellular matrix, and in order to generate constructs with an actual translational value, keeping in mind the native characteristics of the cardiac tissue [34]. And although SC therapy is a promising treatment for myocardial infarction, the minimal functional improvements observed clinically limit its widespread application. Where a need exists to maximize the therapeutic potential of those SCs by first understanding what factors within the infarct microenvironment affect their ability to regenerate the necrotic tissue, and to define a novel mechanism by which the extracellular environment of the infarction regulates the therapeutic potential of MSCs.

Figure 6: Hpsc-Based Bioengineering Strategies for Car-Diac Regeneration
The panel (A) shows a schematic representation of the different hPSC-based methodologies used for regenerative purposes in the cardiac field. The panel (B) provides a timeline that summa-rizes the key milestones reached in the field, starting from the simple injection of hPSC-CMs into the heart to the development of tissue-like structures with enhanced hPSC-CM maturation, more complex perfusable and personalized constructs and in-jectable hydrogels
SMs, skeletal myoblasts; hPSC-CM, cardiomyocytes from hu-man pluripotent stem cells; PSCs, pluripotent stem cells; iPSCs, induced pluripotent stem cells; ESC, embryonic stem cell; EPCs, endothelial progenitor cells; CPSs, cardiac progenitor cells; ECM, extracellular matrix; dECM, decellularized extracellular matrix
Source: Marta Mazzola, Elisa Di Pasquale. Toward Cardiac Regeneration: Combination of Pluripotent Stem Cell-Based Therapies and Bioengineering Strategies. Front. Bioeng. Biotechnol., 2020, Sec. Tissue Engineering and Regenerative Medicine, Vol 8, PP 1-23 https://doi.org/10.3389/fbioe.2020.00455.
Moreover, within the last years, many biomaterials have emerged as good candidates for cardiac tissue engineering to fill the existing gap for their application to cardiac regeneration and treatment of myocardial infarction, and to increase cell retention, improving cell survival and coupling with the host. In general, cell homing and adhesion to cardiac tissue can occur via specific receptor-mediated interactions with the extracellular matrix (ECM) proteins or can be initiated via physical/chemical adhesion to cardiac cells via hydrophobic and electrostatic interactions. It is widely accepted that SCs target inflammation and injury sites, including infarcted myocardium. Accordingly, many SC membrane reengineering approaches aim to promote these endogenous processes, whilst describing the development of new targeting strategies to improve homing efficiency (Figure 7).
The fast-growing development of new technologies to reengineer the membrane of SCs or provide a supporting biocompatible matrix may alleviate these limitations. It is clear that genetic approaches are extremely exciting, as they can be implemented through reliable protocols that are easy to track in the preclinical phase and have a low risk of triggering unwanted immune responses. With respect to the developments within the biomaterial scaffold space, although they of-fer an effective solution to myocardium cell retention and cell number, the transplant process is generally more invasive, and challenges with effective electromechanical integration still remain. Meanwhile, the scaffolds could be modified or implemented in combination with molecules that activate the recruited cells (e.g., statins) and amplify their therapeutic potential. In conclusion, it is likely that no single approach to SC membrane reengineering will provide the “magic bullet” for cardiac cell therapies, and that the next generation of therapies will likely utilize combinations of these technologies to fully harness the therapeutic potential of transplanted SCs.
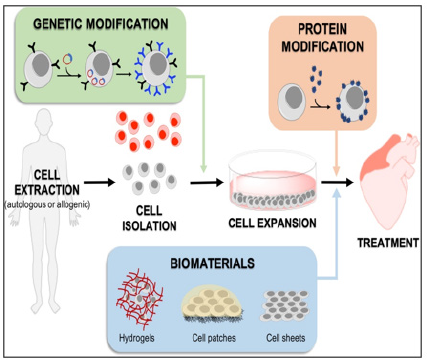
Figure 7: Methods For Augmenting Stem Cell (SC) To Improve Homing and Retention in Cardiac Therapies
SCs are extracted from the source, purified, and expanded to achieve the desired numbers for treatment. Their cell membrane can be modified to improve homing to cardiac tissue genetically, or by using homing proteins or soft biomaterials. SCs can be genetically modified prior to the expansion phase to overexpress membrane receptors or ad-hesion markers. They can also be treated with proteins that are prone to stick to cardiac tissue after expansion or they can be built into scaffolds that provide them with new functionalities and properties
SCs, stem cells
Source: Raquel Cruz-Samperio, Millie Jordan, Adam Perriman, Cell augmentation strategies for cardiac stem cell therapies, Stem Cells Translational Medicine, Volume 10, Issue 6, June 2021, Pages 855–866.
However, regardless of the employed strategy, generation of tissue-like engineered structures suitable for regenerative cardiology applications needs further considerations, specifically in relation to some aspects that may be critical for their efficacy and potential undesired effects in vivo.
Cardiac Scs (Cscs) As Promising Cell Types for Cardiac Regeneration
Myocardial infarction results in an irreversible loss of cardiomyocytes due to aging or pathophysiological conditions, which is generally considered irreversible, and can lead to lethal conditions with subsequent adverse remodelling and heart failure. But human PSCs, including ESCs and iPSCs, can self-renew while maintaining their pluripotency to differentiate into all cell types, including cardiomyocytes. So, identifying new sources for cardiomyocytes and promoting their formation represents a goal of cardiac biology and regenerative medicine.
Meanwhile, because of the lack of strong evidence supporting the existence of resident CSCs, efforts are focused on how to mobilize and promote those few cardiomyocytes that have potential to proliferate in mammalian hearts. Whereas endogenous cardiomyocytes can proliferate at a low frequency, methods that promote cardiomyocyte proliferation may be a vital research field in the future. The aforementioned pathways could be manipulated, optimized, and synergized to achieve better functional improvement after cardiac injury. In addition to inducing cardiomyocyte proliferation, other alternative approaches could be considered (Figure 8).
In situ reprogramming of fibroblasts to cardiomyocytes by overexpression of specific transcription factors represents a promising direction for future study [35]. Recent studies have also suggested that small molecules can induce reprogramming of fibroblasts to cardiomyocytes in vivo, which offers a dual pharmaceutical approach to regenerate cardiomyocytes and reduce scar formation [36].
Human ESCs or iPSCs-derived cardiac progenitors and cardiomyocytes have been successfully transplanted and confirmed the survival of transplanted cells within the host myocardium and an improvement in cardiac function after injury. Furthermore, dual SC therapy, such as human iPSC-derived cardiomyocytes and MSCs, or human ESC-derived epicardium and cardiomyocytes, synergistically improves cardiac function and augments vascularization in the injured myocardium. Moreover, recent advances in engineered epicardial patch containing multiple cardiac cell types are improved heart function and neovascularization after myocardial infarction [37-39].
In this context, CSCs are considered to be very promising cell types for cardiac regeneration. And within the past decade, among cardiac myocytes many types of putative CSCs have been reported to regenerate the injured myocardium by differentiating into new cardiomyocytes. Some of these CSCs have been translated from bench to bed with reported therapeutic effective-ness. However, recent basic research studies on stem cell tracing have begun to question their fundamental biology and mechanisms of action, raising serious concerns over the myogenic potential of CSCs [40].
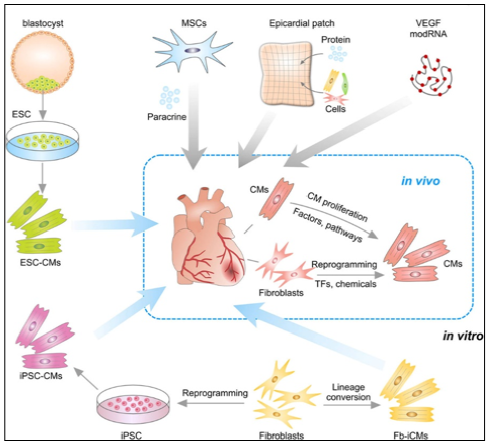
Figure 8: Strategies for Treating Cardiac Repair and Regeneration
Transplantation of embryonic stem cell (ESC)-derived CMs or iPSC-derived CMs into myocardial infarction heart. CMs could also be reprogrammed from Fb in vitro or by transcriptional factors (TFs) or chemicals in vivo. Transplantation of MSCs promotes neovascu-larization and cardiomyocyte survival through para-crine mechanism. Epicardial patch containing growth factors or cardiac cells restores heart function after myocardial infarction. Administration of modified RNA (modRNA) of paracrine factors promotes heart function and drives heart progenitor cell fate. CMs could be promoted to proliferation by regulation of factors or signaling pathways
Fb, fibroblasts; ESC, embryonic stem cell; CMs, cardi-ac myocytes; TF, transcriptional factor; iPSC, induced pluripotent stem cell; MSCs, mesenchymal stem cells; modRNA, modified RNA
Source: Lingjuan He, Ngoc B. Nguyen, Reza Ardehali and Bin Zhou. Heart Regeneration by Endogenous Stem Cells and Cardiomyocyte Proliferation. Controversy, Fallacy, and Progress.
Circulation. 2020;142:275–291, https://doi.org/10.1161
In particular, c-Kit+ CSCs represent a promising candidate for cardiac-specific SC line-ages, which is likely heterogenous in nature [41]. The 1-year follow-up results from the SCIPIO trial support that intracoronary infusion of one million autologous c-Kit+ CSCs in patients with heart failure of ischemic etiology undergoing coronary artery bypass grafting is safe and feasible and may promote significant improvements in global heart function, decrease scar size, and induce regeneration of viable myocardium. As you might see, resident CSCs are self-renewing and can give rise to cardiomyocytes. Their multipotentiality allows them to differentiate along the three main cardiac lineages: myocytes, endothelial cells, and smooth muscle cells. After their injection in the ischemic heart, the formation of the above-mentioned cell types contributes to the regeneration of myocardium and the improvement of its contractility. But the approaches securing cardiac regeneration in post-infarction period are not available to be practiced. And the key problem is the identity of cells be born to generate functionally active cardiac myocytes replenishing those being lost during ischemia.
Previous research made by several groups has identified an extensive population of CSCs in the adult heart. Through the view of the latter, there are four ongoing clinical studies to test the benefits of autologous CSCs:
ALCADIA will use cardiac-derived SCs to treat ischemic cardiomyopathy;
CADUCEUS: a prospective, randomised phase 1 trial [42].
TICAP will take advantage of the cardiosphere-derived stem/progenitor cells (de-rived from cell outgrowth of autologous cardiac biopsies) for patients with a re-cent MI or heart failure separately;
SCIPIO trial, where patients with ischemic cardiomyopathy are treated with c-kit+ Lin– cardiac progenitor cells derived from the right atrial appendage [43].
Meanwhile, with identification of Resident CSCs (rCSCs), it has been supposed that the latter may be a crucial source to initiate and prompt myocardial self-renewal and regeneration. But along with the latter, endogenous Cardiomyogenic SCs (CMSCs) might be better choice for cardiac repair. Ideally, those cells should be directly stimulated in situ, avoiding extraction, purification, culture and reinjection. It is therefore of uttermost importance to understand the identity and function of the cells that constitute the natural environment of cardiac progenitors and support their quiescence, self-renewal and activation. Additionally, further studies are needed to develop a deeper understanding about the properties of pericytes, as these cells have the potential to migrate to different tissues away from their perivascular location and play an active role in the activation of cardiac repair after ischaemia. This would involve a modern interpretation of the pericyte’s role as a cell type involved in reducing the threshold for the activation of an angiogenic program in cardiac repair [44].
CSC therapy holds great potential to prompt myocardial regeneration in patients with is-chemic heart disease. The selection of the most suitable cell type is pivotal for its successful ap-plication. Various cell types, including crude bone marrow mononuclear cells, skeletal myoblast, and hematopoietic and endothelial progenitors, have already advanced into the clinical arena based on promising results from different experimental and preclinical studies. However, most of these so-called first-generation cell types have failed to fully emulate the promising preclinical data in clinical trials, resulting in heterogeneous outcomes and a critical lack of translation. There-fore, different next-generation cell types are currently under investigation for the treatment of the diseased myocardium [45].
Drug (including SC-driven) development continues to move in the direction of Personalized and Precision Medicine (PPM), - where ideally the most effective therapy or treatment is determined by the genetic makeup of the patient and thus by a spectrum of biomarkers to be targeted by the therapeutics (including those being based on SCs). Regarding PPM-based cardiology practice and the innovations made in the field of SC-driven technologies, the focus in the last years has been moved towards a concept of the new wave of cardiac myocyte (CM) formation via a scenario of dedifferentiation and proliferation of mature CMs. The observation that CSCs can be developed inside a pool of immature cardiac cells by formation of “cell-in-cell structures” (CICSs) has enabled us to conclude that CICSs being encapsulated are implicated into mammali-an cardiac myogenesis over the entire lifespan (Figure 9A, B).
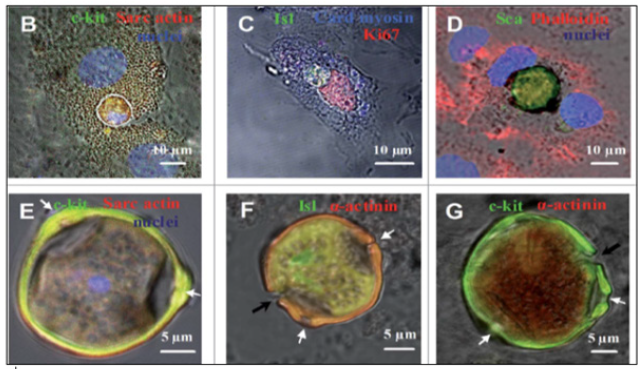
Figure 9A: The CSCs inside CMCs and the formation of CSC-containing CICSs in the cultures ob-tained from newborn and 20- and 40-day-old rats
(A) Experimental design. The cells were plated and cultured for up to 30 days, followed by immunostaining or time-lapse microscopy. (B–G) Immunocytochemistry. The nuclei of the cells have been stained with Hoechst. Transmitted light and fluorescent images are merged. (B) c-kitC CSC inside a CM obtained from a newborn rat (day in vitro). (C) Isl1C CSC inside a CM obtained from a newborn rat (day in vitro). As docu-mented by the expression of Ki67, both the CSC and the host cell exhibit proliferative ability. (D) ScaC CSC encapsulated between the nuclei of the host cell (20-day-old rat, day in vitro). (E) A mature c-kitC CSC-containing CICS with a prominent coating (“capsule”) with 3 pores (white arrows, 40-day-old rat, day in vitro 6). Optical sectioning shows the host cell nucleus (blue) just above the CICS. (F-G) The CICS cap-sule in detail. (F) Erosion of the Isl1C CSC-containing CICS capsule (black arrow) obtained from a 40-day-old rat, day in vitro. The pores are also visualized (white arrows). The capsule interior is positive for sarcomeric a-actinin, also observed in (G). (G) Erosion of the c-kitC CSC-containing CICS capsule (black arrow) obtained from a newborn rat, day in vitro 20. The pores are seen (white arrows).
CMs, cardiac myocytes; CSC, cardiac stem cell; CICSs, cell-in-cell structures”.
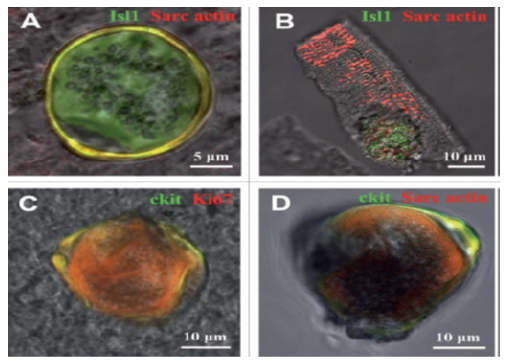
Figure 9B: Cell-In-Cell Structures (Cicss) Identified in The Suspension of Freshly Isolated Myocardial Cells (Ex Vivo) Of 20- And 40-Day-Old Rats
Transmitted light and fluorescent images are merged. (A and B) Isl1C CSCs inside cardiomyocytes of 20-day-old rats (Isl1, green), a-Sarcomeric actin, red). (C) c-kitC CICS. (40-day-old rat, c-kit, green; Ki67, red). (D) c-kitC CICS. (40-day-old rat, c-kit, green; a-Sarcomeric actin, red).
It had been demonstrated before that new CMs are generated through formation of CSC-derived transitory amplifying cells (TACs) either in the CM colonies or in a process of intracellu-lar development of CICSs being encapsulated. The analysis of adult rat cardiac cell suspension 1-5, 10, and 14 days after permanent coronary occlusion and ischemia/reperfusion has gifted a researcher a unique phenomenon of TAC release from mature CMs with clear sarcomeric structure. In this case a development of the intra-cellular CSC occurs within the vacuole pre-formed by CSC-driven sarcolemma-induced invagination. The comparison of TACs exiting the CICS with capsule with TACs just released from non-encapsulated CICS showed that non-encapsulated CICS-derived TACs are characterized by in-creased expression of cardiac markers and decreased expression of stemness-related markers. Earlier on syngeneic animals using the label - GFP was shown that a local laser apoptotic effect on tissues causes an intensive transition of mesenchymal BMSCs from the bloodstream to the zone of programmed cell death (Figure 10).
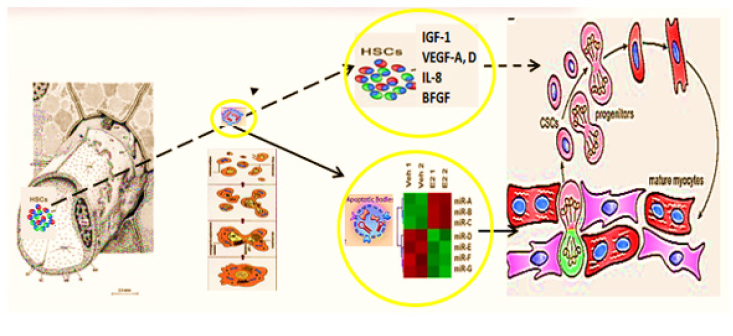
Figure 10: Hypothesis of pathogenesis of restoration of myocardial integrity after injury involving apoptotic bodies of cardiomyocytes (ApBc), resident myocar-dial stem cells (CSCs), and mesenchymal bone marrow stem cells (HSCs).
Apoptotic Bodies (ApB) of cardiomyocytes combine the functions of CSC and HSCs in the areas of myocardial regeneration. Signalling molecules are located on the surface of ApB, which mediate the homing and chemotaxis of HSCs to the area of the damaged myocardium. HSCs provides targeted delivery of growth factors and cytokines required to maintain CSC proliferation. ApB contains a complex of molecules, carriers of "epigenomic memory" about the tissue be-longing of a dead cell. It is likely that simultaneously with the triggering of the effector link of apoptosis, which ends with the formation of ApB, RNA is expressed, the new profile of which is the “code” of the tissue belonging to the dead cell. When ApB enters the CSC via endocytosis, a specific set of long and short non-coding RNAs express genes that determine the direction of differentiation of resident myocardial stem cells. It can be assumed that this hypothesis is true not only for the heart, but also for other organs and tissues [46].
Those data for the first time suggest that the development of CSCs inside the encapsulated CICSs is important for cardiac self-renewal and maintenance of CSC-based pool. At the same time, the development of CSCs inside a population of mature CMs is resulting in the formation of pre-cardiac myocytes, which are able to substitute for irreversibly injured CMs, representing the major mechanism of myocardial regeneration. And that, in turn, would open up a green light to secure the targeted management of regenerative cardiac myogenesis. So, the formation of new cardiomyocytes within the injured myocardium has not been conclusively demonstrated. Meanwhile, SC therapies are viable alternatives to conventional treatments with substantial therapeutic potential and market opportunities. And the lessons learned from our studies would yield fundamental biological insights in the repair of ischemic and other myocardial dis-eases whilst securing the therapeutic resources with the unique future.
Conclusion
To stress the above-mentioned, Biodesign inspired and biotech-driven translational re-search & applications are keeping success in the field elusive in terms of return on investment and in terms of attractiveness to investors within and outside of biopharma and SC-related market as well. The SC market itself is predicted to grow to around $12.1 billion by 2024, whilst the development of the SC therapy into further applications has not yet become common practice, and the true potential of regenerative medicine has yet to be demonstrated fully. For instance, the allogeneic or ‘off-the-shelf’ business model for SC-based therapies is far more akin to current biopharmaceuticals, where the product maintains long-term stability. Meanwhile the global SC therapy market for cardiovascular disorders is likely to grow to $3.3 billion by 2030, driven by the rising incidence of those disorders.
However, the translation of SC therapy for heart diseases from bench to bed is still hampered by several limitations, covering some barriers dictated by the clinical application of SC-based therapies, the investigation of mechanisms behind SC based cardiac regeneration and also, what bioengineers can do and have been doing on the translational stage of SC therapies for heart repair. In this context, the biotech company CardioCell announced effective results in the application of SCs for the treatment of chronic heart failure indications at the European Society of Cardiology Congress, presenting the outcome of preclinical and clinical trials to study the effects of intravenous ischemic tolerance to MSCs in the treatment of chronic heart failure [47].
During the same year, at the annual meeting of the Society for Cardiovascular Angiography and Interventions (SCAI), a number of experts announced promising results for the RENEW trial and the ATHENA (Autologous adipose-derived regenerative cells for refractory chronic myocardial ischemia with left ventricular dysfunction) trial. Although the results of these trials did not sufficiently show significant efficiency due to the early termination and limited sample size, they could still be promising development demonstrating the potential for viable SC-based heart therapies. Meanwhile, design-driven engineering methods (Figure 11).
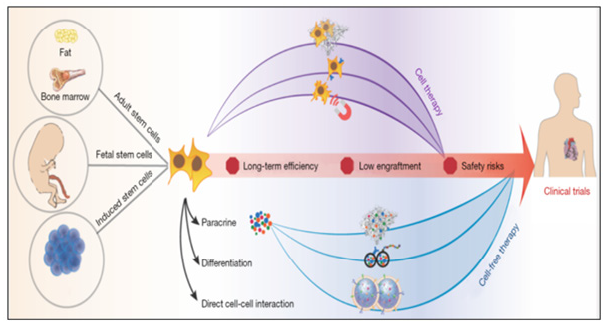
Figure 11:Design-driven bioengineering strategies to accelerate clinical translation of SC therapies
How to overcome the most conspicuous shortcomings with more innovative strategies is a question for every bioengineer, which has been trying to do and needs to be done in the future. In any case, an indispensable premise is a more comprehensive inter-pretation and understanding of the mechanism under which stem cells benefit cardiac regeneration. As we mentioned before, in addition to the paracrine effect, we still believe that direct cell-cell contact plays a vital role in this process. Therefore, the combination of the paracrine effect and the potential activation of the intrinsic program of cardiac cells, which is triggered by cell-cell crosstalk, followed by further observation of cell fate, cell niche, and cell in situ migration, are our top priorities for the next decade of advancing stem cell therapy for heart repair SC, stem cell
The source: Li, Junlang, Hu, Shiqi, Cheng, Ke. Engineering better stem cell therapies for treating heart diseases. Annals of Trans-lational Medicine; 2020, Vol 8, No 8, PP> 1-12.
that aim to realize the multi functionalization of SC therapies have been thriving in the past five years. With the collaboration of physicians, chemists, biodesigners and bioengineers are able to develop SC-based therapies that combine SCs, or their byproducts, with biomaterials in order to enhance therapeutic efficiency and to secure the effective delivery and biosafety [48].
So, SC research and translational applications have made significant progress in the field of cardiovascular regenerative medicine over the past few decades. However, there is still a long way to go before SC therapy can be safely and effectively applied in clinical practice. But moving forward, we must better characterize SC-based therapy and clinical trials, facilitating decision making across the sector. More basic science investigation is required to elucidate the specific mechanism(s) by which SC promote cardiac regeneration and/or repair, and how cells can be optimally delivered and engineered. As our understanding of SC therapy increases, it becomes more likely that clinical trials can produce truly meaningful results with implications for clinical practice.
Based on the new mechanisms and unique phenomenon, we are developing improvement strategies to boost the potency of SC repair and to generate the “next generation” of SC-based and regulatory biomolecules-based (bimodal) therapeutics. Moreover, our strategies should aim at more personalized SC therapies in which individual disease parameters influence the selection of optimal cell type, dosage and delivery approach. And encouraging pre-clinical and clinical studies as one and solid entity reporting significant SC-mediated cardiac regeneration would rapidly pave the way for clinical translation. So, a desire to discover innovative SC-based technologies of the next-step generation would encourage governments and companies to focus directly on re-generative medicine as a future potential economy and social insurance booster.
Thus, prospective research should focus on the development of specific responder scores and the identification of prognostic SC- and cardiac damage-related biomarkers to identify patient cohorts who benefit most from distinct SC treatments. Thereby, a higher standardization of study designs and the establishment of a global open-access database for the registration and publication of pre-clinical and clinical trials would greatly improve the comparability and access of obtained data. In reality, the global (worldwide) SC therapy market is still in an early stage. And the developing translational pipelines for rising applications will build the competition among merchants amid the conjecture time frame.
Consequently, the focus of research in the field has since shifted to SC-derived paracrine factors, including cytokines, growth factors, mRNA, and miRNA. Notably, both mRNA and miRNA can enter into the extracellular space either in soluble form or packed into membrane ves-icles. SC-derived paracrine factors have been shown to suppress inflammation and apoptosis, stimulate angiogenesis, and amplify the proliferation and differentiation of resident cardiac SCs (CSCs). Such features have led to exosomes being considered as potential drug candidates affording myocardial regeneration.
The search for chemical signals capable of stimulating cardio myogenesis is ongoing de-spite continuous debates regarding the ability of mature cardiac myocytes to divide or dedifferentiate, trans-differentiation of other cells into cardiac myocytes, and the ability of CSCs to differentiate into cardiac myocytes. Future research is aimed at identifying novel cell candidates capable of differentiating into cardiac myocytes. The observation that CSCs can undergo intra-cellular development with the formation of “cell-in-cell structure” and subsequent release of transitory amplifying cells with the capacity to differentiate into cardiac myocytes may provide clues for stimulating regenerative cardiomyogenesis.
Indeed, human SC-based therapy derivatives are extremely attractive for therapeutic development because they have direct pharmacologic utility in clinical applications, unlike any other adult cells. The human SC as a special entity is emerging as a new type of potential therapeutic agent of cellular entity in cell-based regenerative medicine, because human SC-based therapy derivatives have the potential for human tissue and function restoration that the conventional drug of molecular entity lacks. In this context, realizing the translational and therapeutic potential of CICSs has been hindered by some obstacles which would be surmounted and brought SC-based therapy of the future towards clinical applications, including establishing defined culture systems for de novo derivation and maintenance of clinical-grade progenitor cells and lineage-specific differentiation of the latter by siRNA-driven modulation. Such milestone advances and medical innovations in CICSs research allow generation of a large supply of clinical-grade cardiac progenitor-based therapy derivatives targeting for cardiac problems, bringing cell-based regenerative medicine to a turning point. We expect that advanced modalities that integrate cellular, bioengineering, and information (IT) technologies via clinical studies and translational applications as new consolidated entities will enhance the efficacy of cardiac cell therapy and further contribute to cardiac re-generative medicine.
Acknowledgments
None.
Conflict of Interest
None.
References
- Malliaras K, Marban E (2011) Cardiac cell therapy: where we've been, where we are, and where we should be headed. Br Med Bull 98(1): 161-185.
- Menasche P, Vanneaux V, Fabreguettes JR, Lucie Toscaet, Julia Pouly, et al. (2015) Towards a clinical use of human embry-onic stem cell-derived cardiac progenitors: a translational experience. Eur Heart J 21;36(12): 743-50.
- Taylor DA, BZ Atkins, P Hungspreugs, TR Jones, MC Reedy, et al. (1998) Regenerating functional myocardium: improved performance after skeletal myoblast transplantation. Nat Med 4(8): 929-33.
- Reinecke, H MacDon-ald, Hauschka GH, Murry SD (2000) Electromechanical coupling between skeletal and cardiac muscle. Implications for infarct repair. J Cell Biol 1;149(3): 731-40.
- Murry, Mark H Soonpaa, Hans Reinecke, Hidehiro Nakajima, Hisako O Nakajima, et al. (2004) Haematopoietic stem cells do not transdifferentiate into cardiac myo-cytes in myocardial infarcts. Nature 8;428(6983) :664-8.
- Zhang S, Junbo Ge, Lan Zhao, Juying Qian, Zheyong Huang, et al. (2007) Host vascular niche contributes to myocardial repair induced by intracor-onary transplantation of bone marrow CD34+progenitor cells in infarcted swine heart. Stem Cells 25(5): 1195-203.
- Tendera M, Wojciech Wojakowski, Lidia Chojnowska, Cezary Kepka, Wiesława Traczet, et al. (2009) Intracoronary infusion of bone marrow-derived selected CD34+CXCR4+cells and non-selected mononuclear cells in patients with acute STEMI and reduced left ventricular ejection fraction: results of randomized, multicentre myocar-dial regeneration by intracoronary infusion of selected population of stem cells in acute myocardial infarction (REGENT) trial. Eur Heart J 30(11): 1313-21.
- Hirsch A, Robin Nijveldt, Pieter A van der Vleuten, Willem J van der Giessen, Jurrien M ten Berg, et al. (2011) Intracoronary infusion of mononuclear cells from bone marrow or periph-eral blood compared with standard therapy in patients after acute myocardial infarction treated by primary percutaneous coronary intervention: results of the randomized con-trolled HEBE trial. Eur Heart J 32, 1736-1747.
- Cambria, E Pasqualini, Petra Wolint, Julia Günter, Julia Steiger, et al. (2017) Translational cardiac stem cell therapy:ad-vancing from first-generation to next-generation cell types. npj Regen Med 13: 2:17.
- Mazo M, Salomón Hernández, Juan José Gavira, Gloria Abizanda, Miriam Araña, et al. (2012) Treatment of reperfused ischemia with adipose-derived stem cells in a preclinical swine model of myocardial infarction. Cell Transplant 21(12): 2723-33.
- Heldman, Darcy L DiFede, Joel E Fishman, Juan P Zambrano, Barry H Trachtenberg, et al. (2014) Transendocardial mesenchymal stem cells and mononuclear bone marrow cells for ischemic cardiomyopathy: the TAC-HFT randomized trial. JAMA 1;311(1) :62-73.
- Lee, Seung-Hwan Lee, Young-Jin Youn, Min-Soo Ahn, Jang-Young Kim, et al. (2014) A randomized, open-label, multicenter trial for the safety and efficacy of adult mesenchymal stem cells after acute myocardial infarction. J Korean Med Sci 29(1): 23-31.
- Mathiasen, Abbas Ali Qayyum, Erik Jørgensen, Steffen Helqvist, Anne Fischer-Nielsen, et al. (2015) Bone marrow-derived mesenchymal stromal cell treatment in patients with severe ischaemic heart failure: a randomized placebo-controlled trial (MSC-HF trial). Eur Heart J 14;36(27): 1744-53.
- Rossini, Caterina Frati, Costanza Lagrasta, Gallia Graiani, Alessandro Scopece, et al. (2011) Human cardiac and bone marrow stromal cells exhibit distinctive proper ties related to their origin. Cardiovasc Res 15;89(3): 650-60.
- Oskouei, Guillaume Lamirault, Chacko Joseph, Adriana V Treuer, Stephanie Landa, et al. (2012) Increased potency of cardiac stem cells compared with bone mar-row mesenchymal stem cells in cardiac repair. Stem Cells Transl Med 1(2): 116-24.
- Li TS, Ke Cheng, Konstantinos Malliaras, Rachel Ruckdeschel Smith, Yiqiang Zhang, et al. (2012) Direct comparison of different stem cell types and subpopulations reveals superior paracrine potency and myocardial repair efficacy with cardiosphere-derived cells. J Am Col Cardiol 6;59(10): 942-53.
- Vagnozzi R J, Maillet M, Sargent MA, Hadi Khalil, Anne Katrine Z Johansen, et al. (2020) An acute immune response underlies the benefit of cardiac stem cell therapy. Nature 577(7790): 405-409.
- Cambria E, Pasqualini FS, Wolint P, Julia Günter, Julia Steiger, et al. (2017) Translational cardiac stem cell therapy: ad-vancing from first-generation to next-generation cell types. npj Regen Med 13: 2:17.
- Emmert, Maximilian Wolint, Petra Jakab, András Sheehy, Sean Pasqualini, et al. (2016) Safety and efficacy of cardiopoietic stem cells in the treatment of post-infarction left-ventricular dysfunction - From cardio-protection to functional repair in a translational pig infarction model. Biomaterials 122: 48-62.
- Behfar A, Crespo Diaz R, Terzic, Gersh BJ (2014) Cell therapy for cardiac repair - les-sons from clinical trials. Nat Rev Cardiol 11(4): 232-46.
- Xiao, Y Pan, X H Li, X Y Yang, Y L Feng, et al. (2016) Cardiac progenitor cell-derived exosomes prevent cardiomyocytes apoptosis through exosomal miR-21 by targeting PDCD4. Cell Death Dis 23;7(6): e2277.
- Ellison GM, Carla Vicinanza, Andrew J Smith, Iolanda Aquila, Angelo Leone, et al. (2013) Adult c-kit (pos) cardiac stem cells are necessary and sufficient for functional cardiac regeneration and repair. Cell 154(4): 827-842.
- Sultana N, Lu Zhang, Jianyun Yan, Jiqiu Chen, Weibin Cai, et al. (2015) Resident c-kit (+) cells in the heart are not cardiac stem cells. Nat Commun 6: 8701.
- Liu Q, Rui Yang, Xiuzhen Huang, Hui Zhang, Lingjuan He, et al. (2016) Genetic lineage tracing identifies in situ kit-expressing cardiomyocytes. Cell Res 26(1): 119-130.
- Keung W, Boheler KR, Li RA (2014) Developmental cues for the maturation of metabol-ic, electrophysiological and calcium handling properties of human pluripotent stem cell-derived cardiomyocytes. Stem Cell Res Ther 5(1): 17.
- Chen A, Ting S, Seow J, Shaul Reuveny, Steve Oh, et al. (2014) Considerations in designing systems for large scale pro-duction of human cardiomyocytes from pluripotent stem cells. Stem Cell Res Ther 5(1): 12.
- Hulot JS, Stillitano F, Salem JE, Jason C Kovacic, Valentin Fuster, et al. (2014) Considerations for pre-clinical models and clin-ical trials of pluripotent stem cell-derived cardiomyocytes. Stem Cell Res Ther 5(1): 1.
- James J H Chong, Xiulan Yang, Creighton W Don, Elina Minami, Yen Wen Liu, et al. (2014) Human embryonic-stem-cell-derived cardiomyocytes regenerate non-human primate hearts. Nature 510(7504): 273-277.
- Zhu K. et al., 2018.
- Roseann C Schaaf, Susan Toth Cohen, Stephanie L Johnson, Gina Outten, Teal W Benevides, et al. (2011) The everyday routines of families of children with autism: examining the impact of sensory processing difficulties on the family. Autism 15(3): 373-389.
- Nathaniel L Tulloch, Veronica Muskheli, Maria V Razumova, F Steven Korte, Michael Regnier, et al. (2011) Growth of engineered human myocardium with mechanical loading and vascular coculture. Circ Res 109(1): 47-59.
- Ingra Mannhardt, Kaja Breckwoldt, David Letuffe Brenière, Sebastian Schaaf, Herbert Schulz, et al. (2016) Human Engineered Heart Tissue: Analysis of Contractile Force. Stem Cell Reports 7(1): 29-42.
- Kacey Ronaldson Bouchard, Stephen P Ma, Keith Yeager, Timothy Chen, LouJin Song, et al. (2018) Advanced maturation of human cardiac tissue grown from pluripotent stem cells. Nature 556(7700): 239-243.
- Higuchi A, Ku NJ, Tseng YC, Chih Hsien Pan, Hsing Fen Li, et al. (2017) Stem cell therapies for myocardial infarction in clinical trials: bioengineering and biomaterial aspects. Lab Invest 97(10): 1167-1179.
- Song K, Nam YJ, Luo X, Qi X, Tan W, et al. (2012) Heart repair by reprogramming non-myocytes with cardiac transcription factors. Nature 485(7400): 599-604.
- Qian L, Huang Y, Spencer CI, Foley A, Vedantham V, et al. (2012) In vivo reprogramming of murine cardiac fibroblasts into induced cardiomyo-cytes. Nature 485(7400): 593-598.
- Park SJ, Kim RY, Park BW, Lee S, Choi SW, et al. (2019) Dual stem cell therapy synergistically improves cardiac function and vascular regeneration following myocardial infarction. Nat Commun 10(1): 3123.
- Bargehr J, Ong LP, Colzani M, Davaapil H, Hofsteen P, et al. (2019) Epicardial cells derived from human embryonic stem cells augment cardiomyocyte-driven heart regeneration. Nat Biotechnol 37(8): 895-906.
- Ye L, Chang YH, Xiong Q, Zhang P, Zhang L, et al. (2014) Cardiac repair in a porcine model of acute myocardial infarction with human induced pluripotent stem cell-derived cardiovascular cells. Cell Stem Cell 15(6): 750-761.
- Lingjuan He, Ngoc B Nguyen, Reza Ardehali, Bin Zhou (2020) Heart Regeneration by En-dogenous Stem Cells and Cardiomyocyte Proliferation. Controversy, Fallacy, and Pro-gress. Circulation 142(3): 275-291.
- Shathiyah Kulandavelu, Vasileios Karantalis, Julia Fritsch, Konstantinos E Hatzistergos, Viky Y Loescher, et al. (2016) Pim1 kinase overexpression enhances c-kit+ cardiac stem cell cardiac repair following myocardial infarction in Swine. J Am Coll Cardiol 68(22): 2454-2464.
- Makkar RR, Smith RR, Cheng K, Malliaras K, Thomson LE, et al. (2012) Intracoronary cardiosphere-derived cells for heart regeneration after myocardi-al infarction (CADUCEUS): a prospective, randomised phase 1 trial. Lancet 379(9819): 895-904.
- Roberto Bolli, Atul R Chugh, Domenico D'Amario, John H Loughran, Marcus F Stoddard, et al. (2011) Cardiac stem cells in patients with ischaemic car-diomyopathy (SCIPIO): initial results of a randomised phase 1 trial. Lancet 378(9806): 1847-1857.
- Lerman DA, Alotti N, Ume KL, Péault B (2016) Cardiac Repair and Regeneration: The Value of Cell Therapies. Eur Cardiol 11(1):43-48.
- Elena Cambria, Julia Steiger, Julia Günter, Annina Bopp, Petra Wolint, et al. (2016) Cardiac Regenerative Medicine: The Potential of a New Generation of Stem Cells. Transfus Med Hemother 43(4): 275-281.
- Tyukavin A, Belostotskaya G Zakharov E, Suchkov S (2019) 4th World Congress & Expo on Pharmaceutics and Drug Delivery Systems.
- Javed Butler, Stephen E Epstein, Stephen J Greene, Arshed A Quyyumi, Sergey Sikora, et al. (2017) Intravenous allogeneic mesenchymal stem cells for nonischemic cardiomyopathy: safety and efficacy results of a phase II-A randomized tri-al. Circ Res 120(2): 332-340.
- Timothy D Henry, Carl J Pepine, Charles R Lambert, Jay H Traverse, Richard Schatz, et al. (2017) The Athena trials: Autologous adipose-derived regenerative cells for refractory chronic myocardial ischemia with left ventricular dysfunction. Catheter Cardiovasc Interv 89(2): 169-177.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.