Case Report 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Malignant Peritoneal Mesothelioma: A Case Report and Review of The Literature
*Corresponding author: Khadija Hinaje, Department of Medical Oncology, Hassan II University Hospital, Faculty of Medicine, Pharmacy and Dental Medicine of Fez, University Sidi Mohamed Ben Abdellah, Fez, Morocco.
Received: April 29, 2024; Published: May 1, 2024
DOI: 10.34297/AJBSR.2024.22.002954
Abstract
Background: Malignant peritoneal mesothelioma (MPM) is an uncommon and highly aggressive cancer with a poor prognosis. The rarity of this entity and the challenging differential diagnosis with other ovarian and peritoneal neoplasms make diagnosis difficult. Clinical symptoms are not specific, and the imagery remains little or not contributive. Immunohistochemistry is essential to confirm the diagnosis based on a panel of positive and negative markers. Prognosis remains reserved.
Case Presentation: A 51-year-old female patient, with no significant medical history, presented with a progressively increasing abdominal volume evolving in a context of general deterioration. Clinical and radiological examinations revealed moderate ascites associated with peritoneal thickening, and the diagnosis of primary malignant peritoneal mesothelioma was made based on peritoneal biopsy findings, confirmed by immunohistochemistry. Treatment consisted of cytoreduction surgery followed by chemotherapy. The evolution was marked by clinical and radiological improvement.
Conclusion: Due to the rarity of this disease and its nonspecific signs or symptoms, MPM is difficult to diagnose. The therapeutic arsenal, increasingly vast, improves the prognosis of this aggressive disease.
Keywords: Mesothelioma, Peritoneum, Cytoreduction surgery, Chemotherapy
Introduction
Malignant mesothelioma is a rare tumor, with peritoneal localization being the second most common after the pleura, and it represents the most frequent neoplasm of the peritoneum [1]. The clinical manifestations are nonspecific, stimulating several clinical presentations, which makes the diagnosis difficult. Histological and immunohistochemical studies are necessary to confirm the diagnosis [2]. Its prognosis has improved in recent years, especially due to the development of effective treatments combining cytoreductive surgery with intraperitoneal chemotherapy and with new chemotherapy drugs [3].
Materials and Methods
A 51-year-old female patient, married with two children, without any notable medical history, particularly no history of asbestos exposure, presented two months prior to her admission with a progressive increase in abdominal volume associated with abdominal heaviness and alimentary vomiting, evolving in a context of general deterioration.
Clinical examination revealed a dullness on percussion with abdominal tenderness. Diffuse ascites were observed on abdominal ultrasound. An ascitic fluid tap was performed. An abdominopelvic CT scan revealed peritoneal thickening with tissue-like appearance predominating in the paracolic gutter, suggestive of peritoneal carcinomatosis, associated with a moderate volume ascites. Ca125 value was 42 U/ml. An exploratory laparoscopy was performed, revealing a yellowish serous effusion with several millimeter-sized whitish nodules on the anterior peritoneal layer and the greater omentum, prompting a biopsy of the nodules. The result of the histopathological examination favored a carcinomatous process with tumoral peritoneal fluid. Immunohistochemistry (IHC) revealed that the tumor cells expressed calretinin, cytokeratin CK 5/6, and WT1, Ki-67 proliferation index was estimated at 30%, while they did not express Ber-EP4 or PAX-8. This panel led to the conclusion of a primary epithelioid malignant peritoneal mesothelioma. A cytoreductive surgery was proposed to the patient after surgical exploration revealed the presence of widespread peritoneal tumor involving the entire peritoneal cavity and visceral peritoneum with several large nodules affecting the mesentery, mesocolon, and greater omentum. The procedure involved omentectomy and resection of as many nodules as possible. Subsequently, the patient received six cycles of chemotherapy consisting of cisplatin at a dose of 75 mg/m2 and Pemetrexed at a dose of 500 mg/m2, administered in 21-day cycles. Both clinical and biological tolerance were satisfactory. The evolution was marked by a clinical improvement and a partial radiological response with regression of the peritoneal nodules. Currently, the patient is well-controlled with more than 12 months of follow-up (Figure 1).
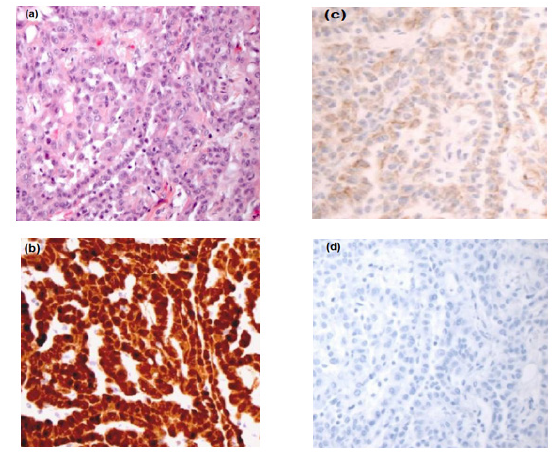
Figure 1: (a) Epithelioid malignant mesothelioma. (b) Immunohistological examination showed nuclear positivity for Calretinine (×25). (c) Immunohistological examination showed nuclear positivity for CK 5/6 (×25), (d) Immunohistological examination showed nuclear negativit for D. Ber - EP 4 :absence of staining (×25).
Discussion
MPM is a rare, aggressive tumor that develops from mesothelial cells lining the pleura, peritoneum, pericardium, and tunica vaginalis of the testis. Pleural malignant mesothelioma is the most common, while peritoneal malignant mesothelioma (PMM) represents only 10 to 30% of all malignant mesotheliomas [4]. MPM is a rare disease, as estimated by the European database Eurocim and the American SEER program, with an annual incidence (per 1,000,000 inhabitants) ranging from 0.5 to 3 cases for men and from 0.2 to 2 cases for women [1]. The average age of onset varies from 47 to 60.5 years, with a median age ranging from 49 to 55.7 years. PMM occurs more frequently in men than in women, although with a less pronounced sex ratio than in pleural mesothelioma [6]. The role of asbestos as a risk factor remains debated in PMM. Some studies suggest that asbestos exposure is a significant etiological factor for PMM, but its implication is believed to be less pronounced than in pleural mesothelioma [5]. Other mineral fibers such as erionite, a mineral fiber belonging to the zeolite group, or mica have also been implicated in PMM tumorigenesis. Many studies have detected the sequence of the SV40 polyomavirus in samples of pleural mesothelioma, and for some, the SV40 virus may act as a co-carcinogen in association with asbestos. Data are limited for PMM. Shivapurkar, et al. analyzed 11 cases of peritoneal mesotheliomas, and the SV40 sequence was detected in 7 of these cases [7]. Finally, the possible etiological role of chronic peritonitis has also been reported [8]. Clinical symptoms are nonspecific and may include abdominal pain, increased abdominal volume, anorexia, weight loss, and/or ascites. General deterioration of health is often present [9], which was the main reason for our patient’s consultation. There are no valuable tumor markers in the diagnosis of MPM. CA-125 is often elevated; however, this marker alone is not specific and is best used to monitor disease recurrence or progression. Radiological assessment comprising ultrasound and abdominopelvic computed tomography (CT) scan is essential to evaluate the peritoneal extension [10]. The definitive diagnosis remains histological and should involve immunohistochemical study. Positive markers for MMP include: calretinin (cytoplasmic staining with nuclear enhancement resembling a “fried egg”), cytokeratin CK 5/6 (cytoplasmic staining), EMA (membranous staining), WT1 (nuclear staining), and D2-40 (membranous staining). Negative markers for MMP, or “epithelial markers,” include: Ber-EP4, B72.3, MOC-31, BG-8, ACE, PAX8 (expressed by Müllerian carcinomas in women), and hormone receptors (expressed in women by ovarian and tubal tumors) [11]. The MPM are classified according to the World Health Organization (WHO) 2004 classification into three main groups: epithelioid mesothelioma (The most common, representing approximately 50% of MPM, and associated with a better prognosis), sarcomatoid mesothelioma, and biphasic mesothelioma [12]. Regarding treatment, there is still no consensus on the best therapeutic approach for MPM. In select patients, the use of cytoreductive surgery combined with hyperthermic intraperitoneal chemotherapy (CRS-HIPEC) is associated with better outcomes and survival. Patients with inoperable MPM or a high risk of early recurrence can be offered several systemic treatments, including chemotherapy, with new approaches emerging. The development of pemetrexed in patients with pleural mesothelioma [13] has justified the implementation of access programs for this molecule in patients with peritoneal mesothelioma. A phase II study evaluating pemetrexed ± cisplatin for patients with peritoneal mesothelioma showed response rates of 23% in previously treated patients and 25% in chemotherapy-naïve patients [14]. Another phase II trial, also for patients with MMP, evaluated pemetrexed ± cisplatin or carboplatin and showed response rates of 13% with a median survival of 10.3 months with pemetrexed alone, and 20% and 24% when pemetrexed was used in combination with cisplatin or carboplatin, with slightly higher median survivals exceeding 13 months [15]. The addition of bevacizumab to the cisplatin + pemetrexed regimen (pemetrexed 500mg/m2; cisplatin 75mg/ m2 and bevacizumab 15mg/kg every 21 days for 6 cycles), followed by maintenance with bevacizumab alone, improved overall survival (median survival increasing from 16.1 to 18.8 months) in patients treated for pleural mesothelioma [16]. In the absence of specific data for MMP and in the absence of contraindications to bevacizumab, the combination of bevacizumab with the cisplatin + pemetrexed regimen is an option for the first-line treatment of unresectable forms of MMP. The prognosis of mesothelioma is bleak. The median survival in untreated patients ranges from 9 to 12 months. With various therapeutic modalities, median survivals range from 29.5 to 94 months depending on the series, with survival rates at 1 year, 3 years, and 5 years ranging respectively from 61% to 88%, from 43% to 68%, and from 17% to 59% [1,4,17].
Conclusion
MPM is a rare condition, and the etiological role of asbestos exposure remains debated. The anatomopathological diagnosis relies on morphological and immunohistochemical features. An international expert group recommends using a panel of antibodies comprising both “positive” and “negative” markers. A double anatomopathological reading is necessary. The implementation of new therapeutic protocols involving maximal cytoreductive surgery and intraperitoneal hyperthermic chemotherapy, as well as systemic treatments, has improved the prognosis of this disease.
References
- Boffetta P (2006) Epidemiology of peritoneal mesothelioma: a review. Ann oncol 18(6): 985-990
- Markaki S, Protopapas A, Milingos S, Lazaris D (2005) Primary malignant mesothelioma of the peritoneum: a clinical and immunohistochemical study. Gynecol Oncol 96(3): 860-864.
- Pocard M (2008) RENAPE (réseau national des tumeurs primitives du péritoine) International workshop on Peritoneal surface malignancy. Lyon.
- Sebbag G, Yan H, Shmookler BM, Chang D, Sugarbaker PH (2000) Results of treatment of 33 patients with peritoneal mesothelioma. Br J Surg 87(11): 1587-1593.
- Loggie BW, Fleming RA, McQuellon RP, Russell GB, Geisinger KR, et al. (2001) Prospective trial for the treatment of malignant peritoneal mesothelioma. Am Surg 67(10): 999-1003.
- Shivapurkar N, Wiethege T, Wistuba II, Milchgrub S, Muller KM, et al. (2000) Presence of simian virus 40 sequences in malignant pleural, peritoneal and noninvasive mesotheliomas. Int J Cancer 85(5): 743-745.
- Peterson JT, Greenberg SD, Buffler PA (1984) Non-asbestos-related malignant mesothelioma. A review. Cancer 54(5): 951-960.
- Mohamed F, Sugarbaker PH (2002) Peritoneal mesothelioma. Curr Treat Options Oncol 3: 75-86
- Krasuski P, Poniecka A, Gal E (2002) The diagnostic challenge of peritoneal mesothelioma. Arch Gynecol Obstet 266: 130-132.
- Husain AN, Colby T, Ordonez N, Krausz T, Attanoos R, et al. (2013) Guidelines for pathologic diagnosis of malignant mesothelioma: 2012 update of the consensus statement from the International Mesothelioma Interest Group. Arch Pathol Lab Med 137: 647-667.
- Travis Wd BE, Brambilla E, Müller Hermelink HK, Harris CC, editors. (2004) Patholgy and genetics of tumors of the lung, pleura, thymus and heart. Lyon: International Agency for Research of Cancer (IARC).
- Vogelzang NJ et al. (2003) Phase III study of pemetrexed in combination with cisplatin versus cisplatin alone in patients with malignant pleural mesothelioma. Journal of Clinical Oncology 21(14): 2636-2644.
- Jänne PA, Anoinette J Wozniak, Chandra P Belani, Mary Louise Keohan, Helen J Ross, et al. (2005) Open-label study of pemetrexed alone or in combination with cisplatin for the treatment of patients with peritoneal mesothelioma: outcomes of an expanded access program. Clinical lung cancer 7(1): 40-46.
- Carteni G, C Manegold, G Martin Garcia, S Siena, C C Zielinski, et al. (2009) Malignant peritoneal mesothelioma-Results from the International Expanded Access Program using pemetrexed alone or in combination with a platinum agent. Lung Cancer 64(2): 211-218.
- Zalcman G, Julien Mazieres, Jacques Margery, Laurent Greillier, Clarisse Audigier Valette, et al. (2016) Bevacizumab for newly diagnosed pleural TNCD - Chapitre 15: Mésothéliome péritonéal - 07/04/2020 59 mesothelioma in the Mesothelioma Avastin Cisplatin Pemetrexed Study (MAPS): a randomised, controlled, open-label, phase 3 trial. The Lancet 387(10026): 1405-1414.
- Chua TC, Yan TD, Morris DL (2009) Outcomes of cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for peritoneal mesothelioma: the Australian experience. J Surg Onco 99(2): 109-113.
- Feldman AL, Libutti SK, Pingpank JF, Bartlett DL, Beresnev TH, et al. (2003) Analysis of factors associated with outcome in patients with malignant peritoneal mesothelioma undergoing surgical debulking and intraperitoneal chemotherapy. J Clin Oncol 21(24): 4560-4507.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.