Research Article 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Blood Parameters Predict Single-Dose Methotrexate Treatment Success in Tubal Ectopic Pregnancy
*Corresponding author:Yang Fan, Department of Obstetrics and Gynecology, People’s Hospital of Ningxia Hui Autonomous Region, Ningxia Medical University, Yinchuan, China.
Received:April 13, 2026; Published:April 20, 2026
DOI: 10.34297/AJBSR.2026.30.003985
Abstract
Objective: The availability of reliable and inexpensive markers that can determine the success of Methotrexate (MTX) treatment in tubal Ectopic
Pregnancies (EPs) is considerable. This study investigated the role of blood-derived systemic inflammatory markers, such as Neutrophil-To-Lymphocyte
Ratio (NLR), Derived NLR (dNLR), Monocyte-To-Lymphocyte Ratio (MLR), Neutrophil Monocyte-To-Lymphocyte Ratio (NMLR), Systemic Inflammation
Response Index (SIRI), and Systemic Immune-Inflammation Index (SII) in predicting tubal EP failure under single-dose MTX treatment.
Materials and Methods: A total of 161 patients with tubal EPs, who underwent a single-dose MTX protocol, were retrospectively analyzed.
The control group (83 cases) had patients cured with MTX, while the failure group (78 cases) had patients who were operated on or received
second-dose MTX. Tubal EP features, such as beta-human chorionic gonadotropin levels, sonographic findings, and blood-derived inflammatory
markers were investigated in both groups.
Results: Blood-derived inflammatory markers like NLR, dNLR, NMLR, SIRI, and SII levels were elevated in the single-dose MTX treatment failure
group when compared with the success group.
Conclusion: Blood-derived inflammatory markers can be easily applied to predict tubal rupture risk in EPs, since a complete blood count, an
inexpensive and easy test, is requested from all patients during hospitalization.
Keywords: Ectopic Pregnancy, Blood Parameters, Inflammatory Markers
Introduction
Ectopic Pregnancy (EP) complicates approximately 2% of all pregnancies and is a leading cause of first-trimester maternal morbidity and mortality. Although EP can resolve spontaneously, tubal rupture and associated hemorrhage require emergency surgery. When diagnosed before tubal rupture, therapies include surgery and medical or expectant approaches [1,2,3]. About 25%- 30% of patients with EP are managed medical. Methotrexate (MTX), an anti-folate cytotoxic drug, is an established treatment for unruptured EP. The MTXtherapy allows many hemodynamically stable women to avoid surgery [4,5]. However, treatment failure occurs in about 20% of those treated with single-dose MTX, and they require rescue surgery or a second dose of MTX [6]. Serial monitoring of serum human chorionic gonadotropin (β-hCG) levels can identify those likely to experience treatment success or failure. Current guidelines recommend measuring serum β-hCG on Days 4 and 7 post-MTX treatment [7]. A reduction in β-hCG levels by >15% in this period indicates likely treatment success, while β-hCG level reduction by <15% is considered a failure and such patients need further MTX treatment or surgery. The protocol does not predict response before seven days post-MTX treatment. Early prognostic information has several advantages. First, identifying likely MTX treatment failure early allows for earlier surgical intervention, which may reduce rupture risk and cost. Second, early prognostic information provides early reassurance and reduces the followup burden for those involved in what can be a distressing and uncertain situation. In recent years, these clinical significances have led to the investigation of the predictive value of trends associated with Day 4 serum β-hCG levels, particularly the percentage changes between Day 1 and Day 4 β-hCG levels. A variety of metrics have been investigated using pre-treatment or Day 4 β-hCG, which predicts treatments outcome with varying degrees of utility [8,9,10]. Although several studies have attempted to identify other predictors of MTX treatment failure, risk factors remain poorly understood and findings are often conflicting.
This study aimed to identify the risk factors for single-dose MTX treatment failure in patients with EP to aid clinical decisionmaking and provide evidence for personalized MTX treatment selection. We aimed to determine whether blood parameters can predict single-dose MTX treatment success.
Materials and Methods
Patient Recruitment
Patients diagnosed with EP at Ningxia People Hospital between January 2014 and December 2023 were retrospectively identified. The inclusion criteria were: (1) Patients who received initial treatment with MTX at our hospital, (2) Patients who were diagnosed with tubal EP based on a comprehensive analysis of clinical information, and (3) Patients who received their entire tubal EP treatment at our hospital. Those who received expectant management or chose surgery as the initial treatment were excluded. At our centre, MTX treatment was chosen for EP cases with stable vital signs, low serum β-hCG levels (ideally <5000 U/L), no significant pain, an adnexal mass of <4 cm without a visible fatal heartbeat, information about the risks, the ability to ensure followup, and no absolute MTX treatment contraindications. Those with intrauterine pregnancy, evidence of immunodeficiency, moderate to severe anaemia, leukopenia or thrombocytopenia, MTX sensitivity, active pulmonary disease, active peptic ulcer disease, clinically significant hepatic dysfunction, clinically significant renal dysfunction, active infection, chronic inflammatory diseases (as they affect hematological parameters), or were breastfeeding, were excluded. Additionally, our study included all patients who met the inclusion and exclusion criteria, regardless of whether their β-hCG levels were increasing or spontaneously decreasing before treatment. In this study, all patients initially received intramuscular MTX at a dose of 50 mg/m2. Serum β-hCG levels were measured on days 4 and 7 post-treatment, and then weekly until negative results were obtained. Patients were divided into the success and failure groups based on MTX treatment outcomes. The need for surgical intervention or a second MTX dose for any reason after MTX administration, including unsatisfactory β-hCG decline or suspected rupture, was considered MTX treatment failure. The success group included patients who were treated with MTX only and achieved negative β-hCG results.
The patients were divided into Group 1 (controls, i.e., patients who were successfully treated with MTX) and Group 2 (cases, i.e., patients who experienced rupture during MTX treatment or needed a second MTX dose). Group 2 had 78 patients. Patients’ venous blood is routinely collected in EDTA-coated anticoagulant tubes at the time of hospitalization and a Complete Blood Count (CBC) was performed on a BC-6000 (Mindray, Shenzhen, China) device that detects 23 parameters. In this study, White Blood Cell Counts (WBC), Neutrophil Counts (NEUT), Lymphocyte Counts (LYMPH), Monocyte Counts (MONO), and ratios, including NLR (NEUT/LYMPH), dNLR (NEUT/ (WBC - LYMPH), MLR (MONO/ LYMPH), NMLR ([MONO + NEUT]/LYMPH), SIRI (NEUT×MONO/ LYMPH), and SII (PLT × NEUT/LYMPH) were determined. Age, body mass index (BMI), obstetric history (gravidity, parity, and EP), history of abdominal surgery and pelvic inflammatory disease, β hCG levels and follow-up, EP material diameter and features, and all other features were retrospectively obtained from patient files and the electronic patient registry. Approval from the local ethics committee and patient consent were obtained for the study.
Statistical Analyses
Statistical analyses were done using SPSS Statistics for Windows version 21.0 (IBM Corp., Armonk, NY, United States), and we adopted a confidence level of 95%. If the kurtosis and skewness values obtained from the measurements fell between t3 and-3, they were considered sufficient for normal distribution. P<0.05 indicated statistically significant differences. In two independent groups, the t-test and Mann Whitney test were used to compare parametric and non-parametric variables, respectively. Relationships involving categorical variables were analyzed using the Chi-squared test. Hematologic markers (NLR, dNLR, MLR, NMLR, SIRI, and SII) were expressed as mean and standard deviation.
Results
Treatment flow chart.
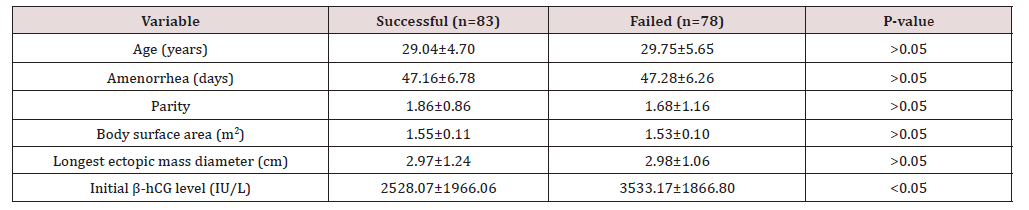
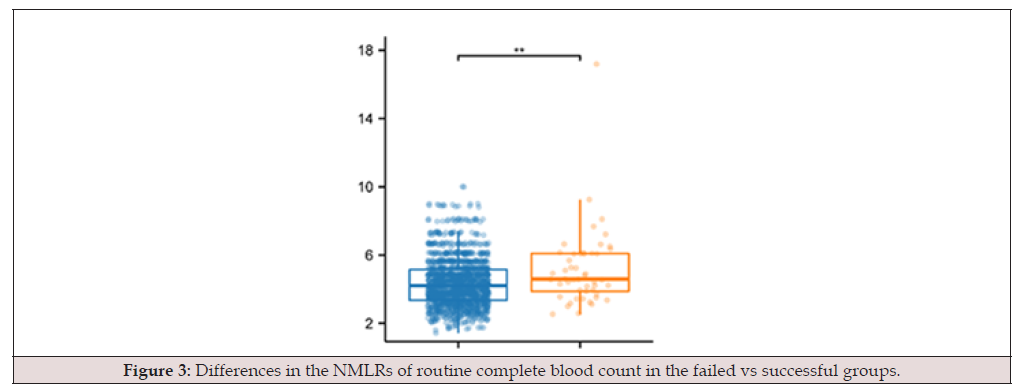
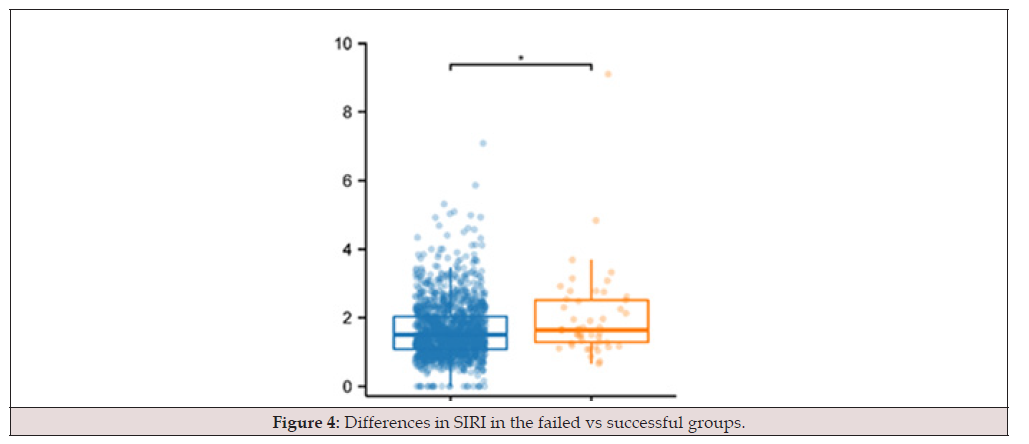
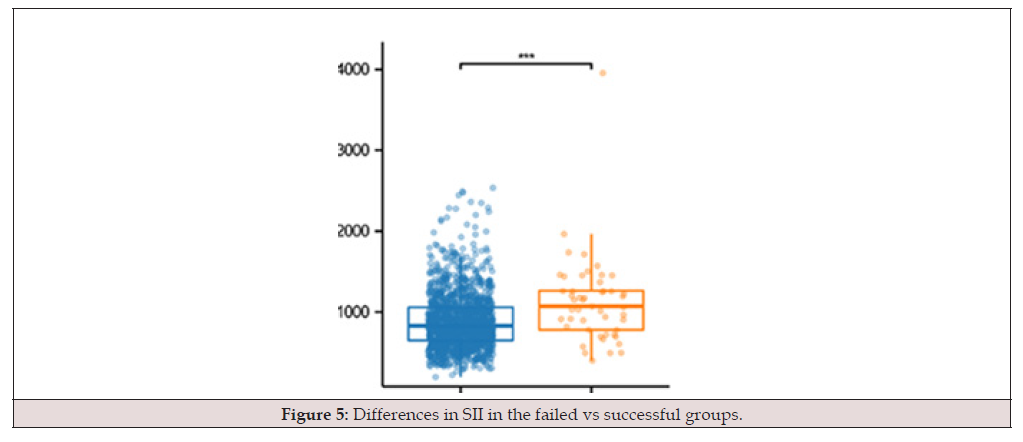
A total of 161 patients were included in the study. Response to treatment was evaluated in 161 patients who received a single dose of MTX and were followed up with β-hCG. Of the 161 patients who received single-dose MTX therapy, 83 (51.6%) were considered successful and 78 (48.4%), who failed single-dose MTX treatment, underwent surgical treatment because of the urgent need for surgery or second-dose MTX treatment (treatment flow chart). Table 1 shows the statistics of numerical variables between groups. The women’s age, the days of amenorrhea, parity, body surface area, and longest ectopic mass diameter did not differ between the groups. However, initial β-hCG levels differed between the groups (2528.07±1966.06 vs 3533.17±1866.80, P<0.05). Table 2 presents the routine blood test parameters of the groups. We determined that the NLR, dNLR, NMLR, SIRI, and SII, but not MLR, differed between the groups. The NLR, dNLR, NMLR, SIRI, and SII are demonstrated in Figures 1-5 (Table 1-2).
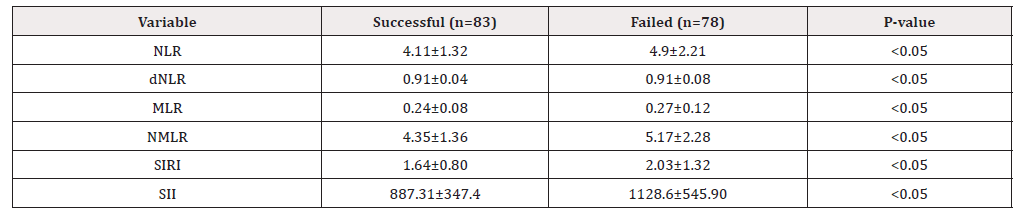
Table 2:Parameters related to routine blood tests in the single-dose methotrexate failure and success groups (mean ± SD).
NLR
Neutrophil-to-lymphocyte ratio, dNLR: derived NLR, MLR: monocyte-to-lymphocyte ratio, NMLR: neutrophil-monocyte-tolymphocyte ratio, SIRI: systemic inflammation response index, SII: systemic immune-inflammation index (Figure 1-5).
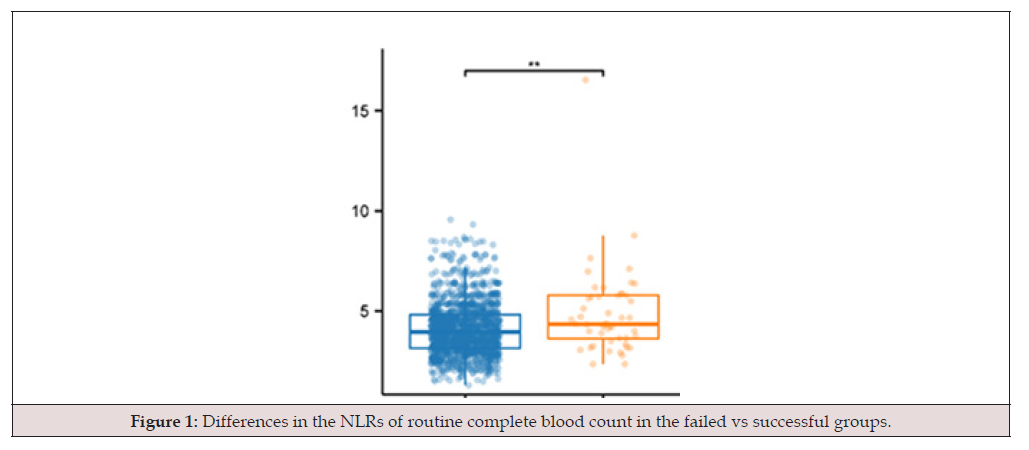
Figure 1:Differences in the NLRs of routine complete blood count in the failed vs successful groups.

Figure 2:Differences in the dNLRs of routine complete blood count on the failed vs successful groups./p>
Discussion
The mechanism of EP is unknown. The conditions that lead to faulty implantation signals because of factors like tubal damage caused by inflammation, altered tubal transport and microenvironment, and increased receptivity in the tubal epithelium, are thought to predispose to EP [11]. Inflammation is an important factor in EP formation and maintenance. Many studies have also shown that the expression of several chemokines and cytokines increases in the tube with the EP [12]. Furthermore, complete blood counts were accepted as serial and basic parameters indicating systemic inflammation. CBC parameters, such as NLR and the platelet-to-lymphocyte ratios are inexpensive and easily calculable inflammatory markers that correlate with systemic inflammatory disease prognosis. Tubal EP rupture may occur during MTX therapy, with serious consequences. Therefore, there is a need for inexpensive, sensitive, and easy-to-apply markers for patients undergoing MTX therapy. This study investigated whether CBC-derived parameters like NLR, dNLR, NMLR, SIRI, and SII can predict EP rupture. Although MTX therapy can be safely administered for tubal EP treatment, it may fail, resulting in ampullary EP tubal rupture. However, many factors affect MTX treatment failure, such as pre-treatment HCG levels, EP size, and gestational age. Additionally, CBC-derived systemic inflammatory markers can affect many parameters. Some studies show that hematological inflammatory markers like NLR may indicate the aging process, and obesity is associated with chronic inflammation. However, in this study, age and BMI were similar in both groups, and patients with systemic comorbidities were excluded. Although we ruled out these variables that may affect inflammatory markers and rupture rate, we determined that one of the two groups with similar characteristics ruptured while the other was cured. In this study, we investigated whether CBC-derived inflammatory markers can predict MTX therapy failure and the resulting life-threatening tubal EP rupture despite the similarities. Pelvic inflammation disease history was significantly higher in the control group but its relationship with rupture is unclear according to literature. Similarly, in these studies, pelvic effusion size was not significant in terms of rupture and in this study, effusion size was higher in the ruptured group. Studies have generally investigated EP properties in an attempt to predict rupture. However, this study investigated the relationship between the CBC parameters routinely obtained from all hospitalized patients and the prediction of tubal EP failure in those undergoing single-dose MTX treatment. Several similar observations have been recently published [13,14]. However, in these studies, EP characteristics, such as baseline HCG levels, differ among the groups. Nonetheless, in these studies, parameters like NLR, dNLR, NMLR, SIRI, and SII are as effective in MTX treatment success as baseline HCG. In the present study, NLR, dNLR, NMLR, SIRI, and SII were higher in the ruptured group than in the control group and other hematological markers, such as NLR, dNLR, NMLR, SIRI, and SII were different in both groups (P<0.05). In this study, NLR increase is an indirect host immune response indicator and was higher in the failure group. However, the underlying mechanisms of the relationship between inflammation and EP treatment outcomes need further study. This study’s strengths are that it was conducted on homogeneous groups (the groups were similar in terms of age, BMI, gestational age, and EP size), with the exclusion of factors influencing systemic inflammation. However, the study is limited by its retrospective and single-centre design.
Conclusion
This study shows that systemic inflammatory markers like NLR, dNLR, NMLR, SIRI, and SII may be suitable tools for detecting MTX treatment failure for tubal EPs. These markers can help physicians estimate which cases will or will not benefit from the treatment right. These systemic inflammatory markers, which are easily obtainable from the CBCs without additional cost, warrant evaluation in larger cohort studies.
Funding
This paper was not supported foundation.
Ethics Approval
This research was approved by the Ethics Committee of Ning Xia Hospital of Medical Sciences.
Consent for Publication
Authors declare no conflict of interest.p>
Author Contributions
All authors contributed to writing and preparing the manuscript.
Consent to Participate
All patients gave written informed consent./p>
References
- Schreiber CA, Sonalkar S (2025) Tubal ectopic pregnancy. N Engl J Med 392(8): 798-805.
- Barnhart KT (2009) Clinical practice. Ectopic pregnancy. N Engl J Med 361(4): 379-387.
- American College of Obstetricians and Gynecologists' Committee on Practice Bulletins—Gynecology (2018) ACOG Practice Bulletin No. 193: Tubal Ectopic Pregnancy. Obstet Gynecol 131(3): e91-e103.
- Lipscomb GH, Stovall TG, Ling FW (2000) Nonsurgical treatment of ectopic pregnancy. N Engl J Med 343(18): 1325-1329.
- Lipscomb GH, McCord ML, Stovall TG, Huff G, Portera SG, et al. (1999) Predictors of success of methotrexate treatment in women with tubal ectopic pregnancies. N Engl J Med 341(26): 1974-1978.
- Barnhart KT, Gosman G, Ashby R, Sammel M (2003) The medical management of ectopic pregnancy: a meta-analysis comparing "single dose" and "multidose" regimens. Obstet Gynecol 101(4): 778-784.
- Kirk E, Condous G, Van Calster B, Haider Z, Van Huffel S, et al. (2007) A validation of the most commonly used protocol to predict the success of single-dose methotrexate in the treatment of ectopic pregnancy. Hum Reprod 22(3): 858-863.
- Mackenzie SC, Moakes CA, Doust AM, Mol BW, Duncan WC, et al. (2023) Early (Days 1-4) post-treatment serum hCG level changes predict single-dose methotrexate treatment success in tubal ectopic pregnancy. Hum Reprod 38(7): 1261-1267.
- Tasgoz FN, Temur M, Dundar B, Kartal E, Ustunyurt E (2020) The role of day 0 and day 4 β-human chorionic gonadotropin values and initial ultrasound findings in predicting the success of methotrexate treatment in ectopic pregnancy. Ginekol Pol 91(7): 389-393.
- Girija S, Manjunath AP, Salahudin A, Jeyaseelan L, Gowri V, et al. (2017) Role of day 4 HCG as an early predictor of success after methotrexate therapy for ectopic pregnancies. Eur J Obstet Gynecol Reprod Biol 215: 230-233.
- Lyons RA, Saridogan E, Djahanbakhch O (2006) The reproductive significance of human fallopian tube cilia. Hum Reprod Update 12(4): 363-372.
- Rajendiran S, Senthil Kumar GP, Nimesh A, Dhiman P, Shivaraman K, et al. (2016) Diagnostic significance of IL-6 and IL-8 in tubal ectopic pregnancy. J Obstet Gynaecol 36(7): 909-911.
- Dereli ML, Savran Üçok B, Özkan S, Sucu S, Topkara S, et al. (2024). The importance of blood-count-derived inflammatory markers in predicting methotrexate success in patients with tubal ectopic pregnancy. Int J Gynaecol Obstet 167(2): 789-796.
- Dinc K, Issın G (2023) Novel marker to predict rupture risk in tubal ectopic pregnancies: the systemic immune-inflammation index. Ginekol Pol 94(4): 320-325.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.