Review Article 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Psychopharmacological Interventions for Autism Spectrum Disorder: A 5-Year Review
*Corresponding author:Yulu Han, Qingdao Medical College, Qingdao University, Qingdao, Shandong, China.
Received:March 08, 2026; Published:March 13, 2026
DOI: 10.34297/AJBSR.2026.30.003938
Abstract
While behavioural therapies remain the primary approach for managing Autism Spectrum Disorder (ASD), there has been a notable increase in specialized interventions that address the neurophysiological aspects of ASD in recent years. Although no medications have been officially approved for the direct treatment of the core symptoms of autism, numerous studies have investigated the efficacy of various substances, including cannabinoids and other pharmacological agents. This review provides an overview and comparison of the current psychopharmacological treatments available for ASD, focusing on traditional psychotropic drugs such as risperidone and aripiprazole, alongside newer agents like cannabidiol and bumetanide. The study examines the mechanism of action, clinical application status, efficacy evaluation, and related challenges while offering references to support the advancement of clinical treatment.
Keywords: Atypical Antipsychotics, Autism Spectrum Disorder, Mood Stabilizers, Pervasive Developmental Disorders, Psychopharmacology
Introduction
Autism Spectrum Disorder (ASD) constitutes a multifaceted neurodevelopmental condition rooted in biological factors, impacting approximately 1 in 44 individuals [1]. It affects multiple aspects of development, including behaviour, problem-solving abilities, self-care skills, and social communication. The symptoms of ASD vary widely in type and severity among individuals, influenced by factors such as age, cognitive abilities, language skills, and co-occurring conditions. The most recent update of the Diagnostic and Statistical Manual (DSM-5) characterizes ASD by deficits in social communication and interaction, as well as restricted, repetitive patterns of behaviour. Social communication deficits include difficulties in social-emotional reciprocity, challenges in nonverbal communication, and problems in forming and maintaining rela tionships [2]. Additionally, restricted, repetitive, and stereotypical patterns of behavior are exhibited through unusual repetitive actions or behaviors, limited interests, a strong insistence on consistency, and a rigid commitment to routines [3, 4]. ASD is often accompanied by co-occurring mental health and medical conditions, including Attention Deficit Hyperactivity Disorder (ADHD), anxiety, depression, intellectual disabilities, speech and language impairments, sleep disturbances, and sensory processing difficulties [5, 6]. These co-occurring conditions can significantly impact the overall functioning and quality of life of individuals with ASD, complicating the identification, diagnosis, and clinical treatment [7].
Traditional approaches to managing medical conditions primarily focus on pharmacological therapies. However, despite years of research into ASD, the prevailing evidence supports behavioral (non-pharmacological) interventions as the cornerstone of treatment for core ASD symptoms. One factor contributing to the limited effectiveness observed in numerous treatment studies is the diverse etiology encompassed by the broad classification of ASD. Some investigations have addressed this complexity by categorizing participants based on genetic backgrounds or phenotypic characteristics. the objective of this paper is to provide an updated overview and comparison of the current psychopharmacological options available for ASD, alongside various therapeutic subtypes, including both established and novel treatments that may become standard care in the coming years based on scientific findings.
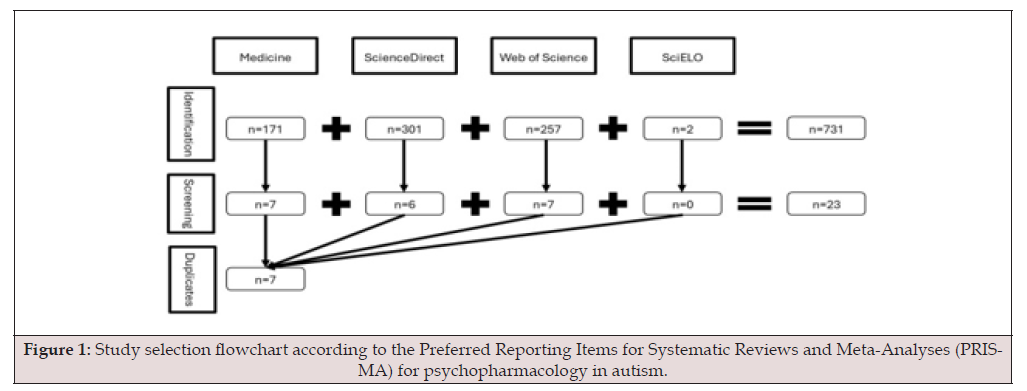
According to system evaluation and management analysis instructions (PRISMA), systematic literature access was applied in January 2025. When the registration number 42024617873, this research is recorded in the initial database of the original system (flowering) international assessment. The search was conducted across four databases: MEDLINE/PubMed, ScienceDirect, Scientific Electronic Library Online (SciELO), and Web of Science. Using keywords such as “autism,” “autism spectrum disorder,” and “psychopharmacology”, we searched 731 papers published between 2019 and January 2025. Subsequently, we performed an additional screening of reserved double-blind placebo-controlled clinical trials, which yielded 7 studies (Figure 1). In this analysis, we focus on psychopharmacological treatments such as risperidone, aripiprazole, bumetanide, and cannabidiol, all of which exhibit significant efficacy and are generally available to clinicians either currently or shortly (Figure 1).
\Established Psychopharmacological Treatments
Risperidone
Risperidone is classified as an atypical antipsychotic and is commonly used to address irritability and related behavioral issues in children and adolescents diagnosed with ASD. The therapeutic benefits of the drug primarily arise from its effects on neurotransmitter systems, specifically targeting serotonin and dopamine pathways. The primary mechanism of action of risperidone involves the antagonism of dopamine D2 and serotonin 5-HT2A receptors. This combined action is particularly significant as it helps to normalize the neurotransmitter systems that often exhibit dysregulation in individuals with ASD [8,9]. Risperidone is one of the few medications approved by the U.S. Food and Drug Administration (FDA) for the treatment of irritability associated with ASD in children and adolescents aged 5 to 16. Clinical trials have demonstrated its efficacy, indicating that risperidone significantly reduces disruptive behaviors compared to a placebo, with notable improvements in irritability, aggressive behavior, and self-harming tendencies [9-11]. A meta-analysis investigating pharmacological interventions for ASD found that both risperidone and aripiprazole significantly impacted core symptoms, which include difficulties in social communication and repetitive behaviors [12]. Additionally, the pharmacokinetic properties of risperidone, as well as its active metabolite 9-hydroxy risperidone, are essential for its effectiveness. Research on therapeutic drug monitoring indicates that keeping plasma concentrations within a specific range can improve treatment results and reduce adverse effects [10]. It is essential to recognize that individual responses to risperidone can differ markedly, possibly due to genetic factors that affect drug metabolism and receptor sensitivity [9,13].
Notwithstanding its endorsement and proven effectiveness, administering risperidone to children with ASD raises certain concerns. This medication may result in side effects such as weight gain, drowsiness, and elevated prolactin levels, potentially requiring thorough monitoring and management [10]. Nonetheless, the proof regarding its effectiveness in alleviating core symptoms remains regarded as initial, and the regular prescription of risperidone for these symptoms is not consistently advised [12].
Aripiprazole
Aripiprazole, classified as an atypical antipsychotic, has garnered attention for its potential therapeutic benefits in managing behavioral dysregulation associated with ASD in younger populations. The drug operates through a multifaceted mechanism, primarily interacting with various neurotransmitter systems, particularly the dopamine and serotonin pathways [9,14]. Furthermore, studies indicate that aripiprazole may enhance the levels of Brain-Derived Neurotrophic Factor (BDNF) and phosphorylated cAMP Response Element-Binding Protein (p-CREB) in neuronal cells. These factors play a critical role in neuronal survival, synaptic plasticity, and overall cognitive performance [14,15].
Aripiprazole, FDA-approved 2009, shown effective/safe via trials, preferred for ASD behavioral issues [9,15]. Recent randomized controlled trials indicate that aripiprazole effectively reduces symptoms such as irritability, hyperactivity, inappropriate verbal communication, and repetitive behaviors within this population [16]. A clinical research study compared the effects of aripiprazole and risperidone over 10 weeks, revealing that both medications led to substantial improvements in irritability. Notably, the aripiprazole group experienced considerably less weight gain, which is a critical consideration given the potential side effects associated with weight gain in younger patients [16-18].
Aripiprazole’s benefits require noting hypertension risk, as in an ASD child’s case [19]. This underscores the necessity for careful monitoring of blood pressure in pediatric patients receiving treatment with aripiprazole. Research has associated aripiprazole with a variety of adverse effects, including weight gain, sedation, excessive salivation, and tremors [20]. Additionally, it has been observed that aripiprazole may increase appetite, potentially exacerbating this negative outcome, whose effect could involve alterations in glucose and lipid metabolism, thereby increasing the risk of developing diabetes and cardiovascular issues over time [13]. Consistent evaluation of metabolic indicators is crucial for children undergoing long-term treatment with aripiprazole. Parents and caregivers must work closely with healthcare professionals to ensure that any adverse effects are promptly identified and managed.
Off-label use of risperidone/aripiprazole beyond FDA-approved ASD irritability raises ethics concerns due to untested safety/ efficacy [19]. The ethical implications of the pharmaceutical industry’s role in promoting medications like risperidone cannot be overlooked. Marketing practices may prioritize profit over patient welfare, leading to the over-prescription of medications without adequate consideration of their long-term effects on children with ASD [21].
Emerging Targeted Treatments with a Possible Role in ASD
Bumetanide
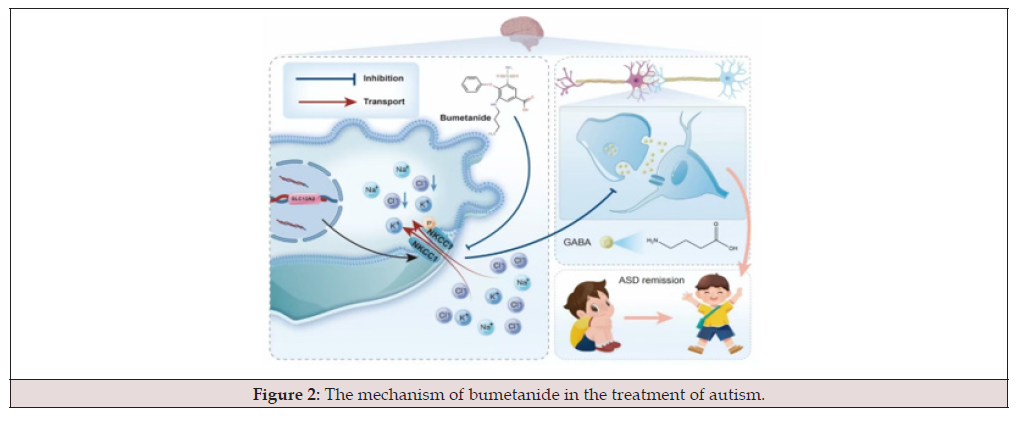
Bumetanide, a loop diuretic, has been explored for its potential therapeutic benefits in ASD due to its capacity to influence the Excitatory-Inhibitory (E-I) balance in the brain. As illustrated in Figure 2, the primary mechanism by which bumetanide exerts its effects involves the inhibition of the Sodium-Potassium-Chloride Cotransporter 1 (NKCC1), a transporter that plays a crucial role in regulating intracellular chloride levels, which subsequently impacts GABAergic signaling, a key inhibitory neurotransmitter system in the brain [22,23]. By inhibiting NKCC1, bumetanide reduces intracellular chloride concentrations, which shifts the effect of GABA from excitatory to inhibitory. This alteration may alleviate certain symptoms associated with ASD, including social impairments and repetitive behaviors [24].
Recent clinical research has emphasized the effectiveness and safety of bumetanide in addressing core symptoms of ASD, especially among children aged 3-6 years, with tablet formulations and interventions lasting three months. For instance, a clinical trial demonstrated that individuals treated with bumetanide experienced significant reductions in symptom severity, along with changes in GABA/glutamate ratios in the brain, suggesting a neurobiological basis for the observed behavioral changes [22]. A case report described a child diagnosed with ASD who exhibited significant improvement in symptoms following bumetanide therapy, while prior treatment with Vitamin D had not produced positive results. This case underscores the potential of bumetanide to target various mechanisms involved in the pathophysiology of ASD [25]. Currently, two Phase III trials are underway to evaluate the efficacy and safety of bumetanide in children and adolescents diagnosed with ASD, the results of which may pave the way for the first pharmacological intervention specifically aimed at enhancing social reciprocity and reducing repetitive behaviors in children affected by ASD [26]. A meta-analysis of nine randomized controlled trials (RCTs) involving 1,036 participants showed that bumetanide significantly improved overall core symptoms of ASD, particularly in sub-domains including relation to inanimate objects, adaptation to environmental changes, auditory response, and anxiety [27].
Despite these encouraging results, the application of bumetanide in clinical settings encounters several challenges. Some studies have indicated that bumetanide treatment could improve factors such as irritability and social behavior [28-30]. However, in a recent study, bumetanide did not significantly affect the core symptoms of ASD, including repetitive behaviors and challenges in social communication. This finding suggests that bumetanide may not address all facets of ASD [31]. Clinical trials generally indicate that participants tolerate bumetanide well. However, it is important to recognize that the existing studies predominantly consist of open-label trials with relatively small sample sizes, which may not adequately represent the full spectrum of side effects that could arise in a larger population [29,32]. Bumetanide may cause common side effects associated with diuretics, such as electrolyte imbalances, dehydration, and potential alterations in renal function. [33]. Moreover, the pharmacological characteristics of bumetanide indicate potential systemic effects that extend beyond its functions in the central nervous system. For instance, it has been proposed that bumetanide might engage with the immune system, which may introduce further considerations about its safety and tolerability in children diagnosed with ASD [28]. More about the clinical studies are described in Table 1. Bumetanide shows promise as a potential treatment for ASD, particularly in improving social behaviors and reducing symptom severity. However, its application in clinical settings faces several challenges, including inconsistent results across studies and potential side effects. Future research, particularly large-scale, well-controlled trials, is needed to fully understand the efficacy and safety profile of bumetanide in ASD. Additionally, further investigation into the mechanisms underlying its therapeutic effects and potential interactions with other biological systems is warranted.
Cannabidiol
Cannabis, particularly its non-intoxicating component cannabidiol (CBD), has garnered interest for its potential effects on ASD and associated neurotransmitter pathways. The endocannabinoid system (ECS), which consists of cannabinoid receptors and naturally occurring cannabinoids, plays a vital role in regulating numerous brain functions, including synaptic plasticity and neuromodulation [34]. Research indicates that cannabis oil with a high concentration of CBD may alleviate symptoms such as anxiety, hyperactivity, and sleep disturbances in children diagnosed with ASD. This effect is likely mediated through interactions with the endocannabinoid system, which may enhance the levels of endogenous cannabinoids, such as anandamide (AEA) and 2-Arachidonoylglycerol (2-AG) [35,36,41]. Furthermore, the influence of cannabinoids on serotonin receptors may contribute to improved management of anxiety and mood disorders commonly observed in individuals diagnosed with ASD [37,38,42].
Numerous observational studies have indicated that cannabis extracts containing CBD can result in significant improvements across various symptom domains, including attention deficit/ hyperactivity disorder, a range of behavioral disorders, and communication challenges, particularly among individuals exhibiting severe self-injurious behaviors [39]. For instance, a study involving 188 individuals with ASD who received medical cannabis treatment found that a significant proportion reported improvements in symptoms after six months, with 30.1% indicating substantial progress in their condition [8]. Notably, the results were particularly promising among patients without epilepsy, indicating a potentially significant therapeutic benefit of CBD for this population [43]. In addition to alleviating symptoms, the use of cannabis has been associated with improvements in the overall quality of life for both patients and their families. A study involving 20 patients who received full-spectrum cannabis extracts found that 90% reported enhancements in primary and related symptoms, with many participants successfully reducing or discontinuing other medications [38].
While the results seem promising, it is crucial to approach the consideration of cannabis as a treatment option with caution. Research has observed that treatment with cannabis may result in changes in behavior, such as heightened agitation, increased irritability, and reduced appetite [39,44-46]. Individual responses to cannabis can vary considerably, and there is a notable lack of standardized dosing protocols. Caregivers and parents must weigh the potential benefits against the risks and uncertainties associated with cannabis therapy [47]. More about the clinical studies is described in Table 1. Future research, particularly large-scale, well-controlled trials, is needed to fully understand the efficacy and safety profile of CBD in ASD. Additionally, further investigation into the mechanisms underlying their therapeutic effects and potential interactions with other biological systems is warranted.
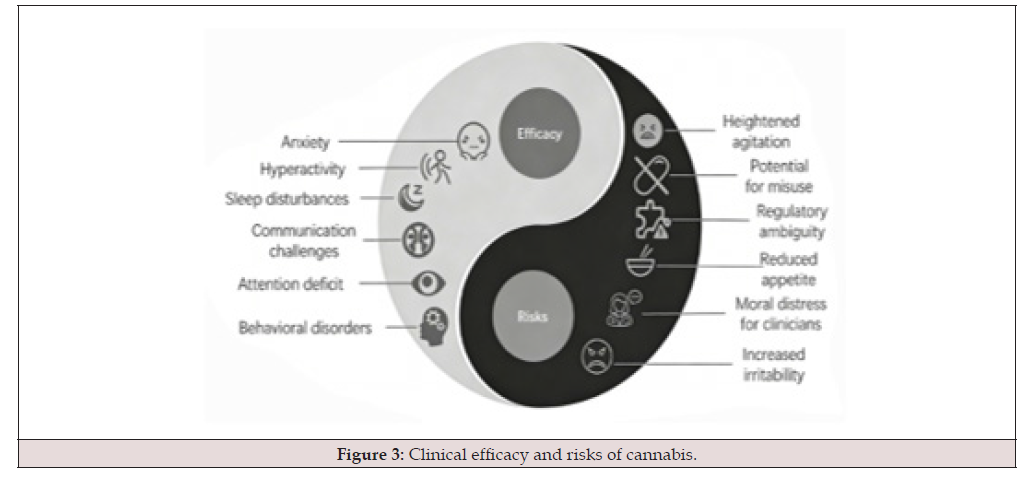
As cannabis becomes increasingly accepted and legalized in various jurisdictions, disparities may emerge in access to quality cannabis products and treatment options [47]. This situation raises ethical questions regarding fairness and justice in healthcare, especially for marginalized communities [48]. Additionally, the role of healthcare providers in advocating for or against cannabis treatment for ASD presents an ethical dilemma that necessitates careful consideration. This scenario can result in moral distress for clinicians, who may find themselves conflicted between their ethical obligations and the wishes of the families they serve [43]. Figure 3 summarizes the therapeutic efficacy and risks of cannabidiol in treating.
The Potential of Combined Psychopharmacological Therapy
Currently, only two drugs specifically approved by the FDA for the treatment of core symptoms of ASD are aripiprazole and risperidone. These medications are primarily utilized to manage irritability and aggression associated with ASD. However, the symptoms exhibited by individuals with ASD are highly complex and diverse, making a singular pharmacological approach often inadequate to comprehensively address and effectively ameliorate all facets of these symptoms [48]. Given the unique mechanisms of action of various pharmacological agents, combination therapy has emerged as a novel and promising treatment strategy for ASD. For example, the combination of atypical antipsychotics with other medications, such as Selective Serotonin Reuptake Inhibitors (SSRIs), has been investigated to target both core and associated symptoms of ASD. SSRIs are recognized for their potential to alleviate anxiety and depressive symptoms, which are frequently observed in individuals with ASD [49]. Furthermore, the combination of memantine and aripiprazole has been investigated for its potential to manage cognitive deficits and autistic-like behaviors in animal models. This combination therapy has demonstrated improvements in cognitive performance and the restoration of neurotransmitter balance, which is frequently disrupted in individuals with ASD [15]. These findings suggest that targeting multiple pathways through drug combinations may offer a more comprehensive approach to treatment.
It is essential to recognize that combined treatment is not without risk. One drug may enhance or inhibit the metabolism of another, thereby altering the concentration of active drugs in the body and potentially triggering previously unreported adverse reactions. For instance, the concurrent use of antipsychotics with other medications that inhibit cytochrome P450 enzymes can lead to increased plasma concentrations of the antipsychotic, heightening the risk of adverse effects [50]. Consequently, prior to implementing combined treatment, a thorough evaluation of potential drug interactions and risks is imperative, which should include comprehensive in vitro studies and animal experiments. If feasible, smallscale clinical trials should also be conducted.
The Integration of Intervention Training with Pharmacological Approaches
Evidence-based ABA and TEACCH, first-choice ASD treatments, target core symptoms via reinforcement/structure; ABA, individualized, aids early skills with data tracking [51]. For example, Early Intensive Behavioral Intervention (EIBI), a form of ABA, has demonstrated substantial gains in cognitive and social skills [52]. ABA is often considered the first choice for ASD treatment due to its evidence-based effectiveness in addressing core symptoms and promoting long-term skill development [53]. EACCH emphasizes creating a structured, supportive environment tailored to the unique needs of individuals with ASD. TEACCH, on the other hand, uses visual support and predictable routines to help individuals navigate their daily activities independently. This approach is highly effective for individuals who struggle with sensory processing or require a predictable environment to feel secure. Recent studies have shown that TEACCH can improve cognitive and language skills, as well as reduce caregiver stress [54]. However, the effectiveness of TEACCH in autism interventions is still being investigated, with some studies reporting mixed results [55].
Compared to pharmacotherapy, ABA and TEACCH offer several distinct advantages in the treatment of ASD. While medications can provide relief for specific symptoms, they often do not address the core deficits of the disorder. ABA and TEACCH, however, directly target social communication deficits and restricted, repetitive behaviors, leading to long-term improvements in skill development and independence. These behavioral interventions also promote a more favorable psychological and physical foundation for individuals undergoing training, enhancing the overall effectiveness of treatment. Recent research has shown that combining pharmacological approaches with ABA or TEACCH can yield improved outcomes, leveraging the strengths of both interventions to provide comprehensive support [56]. For instance, a feasibility study proposes examining the effects of propranolol, a medication recognized for its efficacy in reducing situational anxiety, in conjunction with EIBI grounded in ABA principles [57]. The rationale behind this approach is that pharmacological agents can potentially target underlying neurobiological mechanisms, thereby enhancing the effectiveness of behavioral therapies. Furthermore, Medications influence the neurotransmitter system, modulating the brain’s physiological functions and establishing a more favorable physical and psychological foundation for patients undergoing training. For instance, risperidone alleviates symptoms of irritability, facilitating patient cooperation during intervention training and enhancing the effectiveness of the training [9]. This constructive collaboration between pharmacological and intervention strategies achieves comprehensive rehabilitation support, addressing both physiological and behavioral aspects.
Discussion
Individual Differences in Psychopharmacological Treatments
While risperidone, aripiprazole, bumetanide, and cannabis have all demonstrated varying degrees of efficacy in the medical treatment of ASD, which are illustrated in Table 2, it is crucial to acknowledge the significant differences in individual patient responses to these medications, which are influenced by numerous complicating factors, including gene polymorphisms. Given the heterogeneous nature of ASD, where symptoms and their severity can vary significantly among individuals, a one-size-fits-all approach is frequently inadequate. By customizing combinations of medications and behavioral interventions, clinicians can more effectively address the unique needs of each patient, potentially resulting in improved outcomes in social skills, communication, and overall quality of life [57, 58]. Factors such as cognitive abilities, age, and initial symptom severity have been identified as predictors of treatment response. For example, children with higher cognitive abilities and lower symptom severity tend to exhibit better outcomes from interventions [59,60]. This suggests that tailoring interventions, regardless of whether they include pharmacological components, could lead to more effective treatment strategies.

Table 2:Mechanism of action, main efficacy, common side effects, indications, and limitations of commonly used drugs in ASD.
Different medications target distinct subgroups of symptoms in patients with autism, necessitating that clinicians accurately select medications based on the specific symptoms presented by the patient. Risperidone and aripiprazole primarily address irritability- related symptoms. Risperidone encompasses various manifestations, including aggression, self-injury, and loss of temper. Aripiprazole is derived from risperidone and is recommended for patients who exhibit intolerance to risperidone or have a poor response, as well as for those with comorbid tic disorders. Cannabidiol primarily targets severe anxiety and sleep disorders, while Bumetanide focuses on social behavior abnormalities and delayed language skills. Different individuals may be more suited to specific medications due to their unique conditions and physical health. In terms of usage restrictions, risperidone and aripiprazole have limitations for patients with certain medical conditions due to the characteristics of the drugs; cannabidiol is constrained by legal and safety considerations, limiting its scope of use; and Bumetanide is still in the research phase, requiring further clarification regarding its applicable population and safety. In practical applications, physicians must comprehensively consider the patient’s symptoms, age, physical health, drug safety, and accessibility, weighing the pros and cons to formulate personalized treatment plans (Table 2).
Application of Artificial Intelligence (AI) to Autism Psychopharmacology
The application of Artificial Intelligence (AI) holds significant potential for advancing psychopharmacological interventions for ASD, aligning with the study’s focus on evaluating and optimizing pharmacological treatments. A novel predictive framework based on Drug-Target Interaction (DTP) networks has been developed to analyze associations between drugs and their targets, which helps identify potential effective drug combinations for ASD-directly supporting the study’s comparison of established and emerging pharmacological agents. Additionally, AI-driven platforms like Panda Omics analyze gene expression profiles from patient samples to identify dysregulated pathways and therapeutic targets, providing a data-driven basis for understanding the efficacy of drugs such as risperidone, aripiprazole, bumetanide, and cannabidiol evaluated in this review. AI also assists in optimizing combination therapies by analyzing drug-drug interactions and their effects on biological targets, addressing the complexity of ASD symptom heterogeneity that single-agent therapies often fail to resolve. By streamlining drug screening and reducing development time, AI accelerates the translation of promising pharmacological interventions from research to clinical practice, which is crucial for addressing the unmet need for effective ASD treatments highlighted in this study.
Ethical Challenges in The Psychopharmacological Treatment of Autism
Psychopharmacology for ASD raises ethical issues, including regulatory ambiguity, public health impacts, misuse risk, and informed consent challenges, especially for children [20]. The issue of access to treatment also presents ethical dilemmas. The ethical principle of “do no harm” is paramount in medical treatment, and the potential for adverse effects must be weighed against the expected benefits. Clinicians and researchers must ensure that the risks are minimized and that patients are monitored closely during treatment. This raises questions about the adequacy of safety protocols and the responsibility of healthcare providers to protect vulnerable populations, such as children with ASD [33].
The potential for stigmatization associated with the use of antipsychotic medications in children with ASD is another ethical concern. The label of being on a psychiatric medication can lead to social stigma, which may affect the child’s self-esteem and social interactions. This stigma can also extend to families, who may feel judged for their child’s treatment choices [61]. There is a growing body of evidence suggesting that children from lower socioeconomic backgrounds may have less access to effective treatments for ASD, including pharmacological options like risperidone. This disparity raises questions about equity in healthcare and the ethical obligation to ensure that all children, regardless of their background, have access to appropriate and effective treatment [39].
Conclusion
The current pharmacological management of ASD is multifaceted. Traditional medications like risperidone and aripiprazole are effective in reducing behavioral symptoms, but their side effects can impact patients’ quality of life. The emerging drug bumetanide shows promise in treating core symptoms, but further validation through large-scale studies is needed. Cannabis is being explored as a treatment for ASD, but psychoactive risks, legal constraints, and quality control issues limit its clinical use. Overcoming these challenges requires scientific advancements in quality control and safety research, along with collaborative efforts across society. Combination therapies offer potential for ASD treatment, but the complexity of drug interactions necessitates careful evaluation by researchers and clinicians. Precision medicine is crucial in managing ASD, given the variability in individual responses to drug treatment. In-depth studies on pathophysiological mechanisms, along with the integration of artificial intelligence and big data technologies, can lead to the development of more targeted and safer therapeutic agents. Long-term follow-up studies on existing medications will provide valuable insights for clinical practice, benefiting ASD patients and their families by improving their quality of life and integration into society. The seven included double-blind placebo- controlled clinical trials provide foundational evidence for the discussed psychopharmacological interventions, with consistent findings supporting the efficacy of traditional and emerging agents in specific symptom domains of ASD. These studies collectively highlight the need for personalized treatment strategies, as well as further research to validate the long-term safety and effectiveness of these interventions across diverse patient populations.sponsible use, scientific innovation, and international cooperation.
Ethical Approval and Consent to participate
This study did not involve human or animal subjects, and thus, no ethical approval was required. The study protocol adhered to the guidelines established by the journal.
Human Ethics
No ethical approval was required. The study protocol adhered to the guidelines established by the journal.
Consent for Publication
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor- in-Chief of this journal.
Availability of Supporting Data
No relevant data was used in the article.
References
- Maenner MJ, Shaw KA, Bakian AV, Bilder DA, Durkin MS, et al. (2021) Prevalence and Characteristics of Autism Spectrum Disorder Among Children Aged 8 Years - Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2018. MMWR Surveill Summ 70 (11): 1-16.
- Nazeer A, Ghaziuddin M (2012) Autism spectrum disorders: clinical features and diagnosis. Pediatr Clin North Am 59(1): 19-25.
- Baird G, Cass H, Slonims V (2003) Diagnosis of autism. BMJ 327(7413): 488-493.
- Hyman SL, Levy SE, Myers SM (2020) Identification, Evaluation, and Management of Children with Autism Spectrum Disorder. Pediatr 145(1): e20193447.
- Parmeggiani A, Corinaldesi A, Posar A (2019) Early features of autism spectrum disorder: a cross-sectional study. Ital j Pediatr 45(1): 144.
- Waizbard Bartov E, Fein D, Lord C, Amaral DG (2023) Autism severity and its relationship to disability. Autism Res 16(4): 685-696.
- Underwood JFG, DelPozo Banos M, Frizzati A, Rai D, John A, et al. (2023) Neurological and psychiatric disorders among autistic adults: a population healthcare record study. Psychol Med 53(12): 5663-5673.
- Shrader SH, Mellen N, Cai J, Barnes GN, Song ZH (2024) Cannabidiol is a behavioural modulator in BTBR mouse model of idiopathic autism. Front Neurosci 18: 1359810.
- Choi H, Kim JH, Yang HS, Kim JY, Cortese S, et al. (2024) Pharmacological and non-pharmacological interventions for irritability in autism spectrum disorder: a systematic review and meta-analysis with the GRADE assessment. Mol Autism 15(1): 7.
- Hongkaew Y, Gaedigk A, Wilffert B, Gaedigk R, Kittitharaphan W, et al. (2021) Pharmacogenomics Factors Influencing the Effect of Risperidone on Prolactin Levels in Thai Pediatric Patients with Autism Spectrum Disorder. Front Pharmacol 12: 743494.
- Hacohen M, Stolar OE, Berkovitch M, Elkana O, Kohn E, et al. (2022) Children and adolescents with ASD treated with CBD-rich cannabis exhibit significant improvements particularly in social symptoms: an open label study. Transl Psychiatry 12(1): 375.
- Siafis S, Çıray O, Wu H, Schneider Thoma J, Bighelli I, et al. (2022) Pharmacological and dietary-supplement treatments for autism spectrum disorder: a systematic review and network meta-analysis. Mol Autism 13(1): 10.
- Chen S, Lian J, Su Y, Deng C (2022) Effects of Risperidone and Prenatal Poly I:C Exposure on GABA (A) Receptors and AKT-GSK3β Pathway in the Ventral Tegmental Area of Female Juvenile Rats. Biomolecules 12(5): 732.
- Jeong YJ, Son Y, Park HJ, Oh SJ, Choi JY, et al. (2021) Therapeutic Effects of Aripiprazole in the 5xFAD Alzheimer's Disease Mouse Model. Int J Mol Sci 22(17): 9374.
- Zohny SM, Habib MZ, Mohamad MI, Elayat WM, Elhossiny RM, et al. (2023) Memantine/Aripiprazole Combination Alleviates Cognitive Dysfunction in Valproic Acid Rat Model of Autism: Hippocampal CREB/BDNF Signaling and Glutamate Homeostasis. Neurotherapeutics 20(2): 464-483.
- Maneeton N, Maneeton B, Putthisri S, Suttajit S, Likhitsathian S, et al. (2018) Aripiprazole in acute treatment of children and adolescents with autism spectrum disorder: a systematic review and meta-analysis. Neuropsychiatr Dis Treat 14: 3063-3072.
- DeVane CL, Charles JM, Abramson RK, Williams JE, Carpenter LA, et al. (2019) Pharmacotherapy of Autism Spectrum Disorder: Results from the Randomized BAART Clinical Trial. Pharmacotherapy 39(6): 626-635.
- Maneeton N, Maneeton B, Putthisri S, Woottiluk P, Narkpongphun A, et al. (2018) Risperidone for children and adolescents with autism spectrum disorder: a systematic review. Neuropsychiatr Dis Treat 14: 1811-1820.
- Uzun AD, Sapmaz ŞY, Öztürk M, Kandemir H (2019) Hypertension Induced by Aripiprazole Use in an Autistic Child Patient. Clin Psychopharmacol Neurosci 17(4): 556-558.
- Lopes LPN, de Oliveira JC, Bergamaschi CC, Fulone I, Lima EDC, et al. (2023) Use of second-generation antipsychotics in autism spectrum disorder: a systematic review and meta-analysis protocol. BMJ Open 13(6): e069114.
- Alaqel SI, Abdullah O, Alharbi A, Althobaiti YS, Alturki MS, Ramzy S, et al. (2023) Guava-fruit based synthesis of carbon quantum dots for spectrofluorometric quantitative analysis of risperidone in spiked human plasma and pharmaceutical dosage forms. RSC Adv 13(26): 17765-17774.
- Zhang L, Huang CC, Dai Y, Luo Q, Ji Y, et al. (2020) Symptom improvement in children with autism spectrum disorder following bumetanide administration is associated with decreased GABA/glutamate ratios. Transl Psychiatry 10(1): 9.
- Kourdougli N, Nomura T, Wu MW, Heuvelmans A, Dobler Z, et al. (2024) The NKCC1 Inhibitor Bumetanide Restores Cortical Feedforward Inhibition and Lessens Sensory Hypersensitivity in Early Postnatal Fragile X Mice. Biological Psychiatry.
- Kourdougli N, Nomura T, Wu MW, Heuvelmans A, Dobler Z, et al. (2025) The NKCC1 Inhibitor Bumetanide Restores Cortical Feedforward Inhibition and Lessens Sensory Hypersensitivity in Early Postnatal Fragile X Mice. Biological Psychiatry 97(5): 507-516.
- Feng JY, Li HH, Wang B, Shan L, Jia FY (2020) Successive clinical application of vitamin D and bumetanide in children with autism spectrum disorder: A case report. Med 99(2): e18661.
- Crutel V, Lambert E, Penelaud PF, Albarrán Severo C, Fuentes J, et al. (2021) Bumetanide Oral Liquid Formulation for the Treatment of Children and Adolescents with Autism Spectrum Disorder: Design of Two Phase III Studies (SIGN Trials). J Autism Dev Disord 51(8): 2959-2972.
- Xiao HL, Zhu H, Jing JQ, Jia SJ, Yu SH, et al. (2024) Can bumetanide be a miraculous medicine for autism spectrum disorder: Meta-analysis evidence from randomized controlled trials. Res Autism Spectrum Disor 114: 102363.
- Li Q, Zhang L, Shan H, Yu J, Dai Y, et al. (2022) The immuno-behavioural covariation associated with the treatment response to bumetanide in young children with autism spectrum disorder. Transl Psychiatry 12(1): 228.
- Van Andel DM, Sprengers JJ, Oranje B, Scheepers FE, Jansen FE, et al. (2020) Effects of bumetanide on neurodevelopmental impairments in patients with tuberous sclerosis complex: an open-label pilot study. Mol Autism 11(1): 30.
- Lemonnier E, Rabiei H, Makowski D, Hadjikhani N, Ben Ari Y (2021) Treating Autism with Bumetanide: Are Large Multicentric and Monocentric Trials on Selected Populations Complementary? J Am Acad Child Adolesc Psychiatry 60(8): 937-938.
- Jan J Sprengers, Dorinde M van Andel, Nicolaas P A Zuithoff, Mandy G Keijzer Veen, Annelien J A Schulp, et al. (2021) Bumetanide for Core Symptoms of Autism Spectrum Disorder (BAMBI): A Single Center, Double-Blinded, Participant-Randomized, Placebo-Controlled, Phase-2 Superiority Trial. Journal of the American Academy of Child and Adolescent Psychiatry 60 (7): 865-876.
- Dorinde M van Andel, Jan J Sprengers, Mandy G Keijzer Veen, Annelien J A Schulp, Marc R Lillien, et al. (2022) Bumetanide for Irritability in Children with Senwith Processing Problems Across Neurodevelopmental Disorders: A Pilot Randomized Controlled Trial. Frontiers in psychiatry 13: 780281.
- Hui Lv, Xiao Gu, Xingyue Shan, Tailin Zhu, Bingke Ma, et al. (2022) Nanoformulated Bumetanide Ameliorates Social Deficiency in BTBR Mice Model of Autism Spectrum Disorder. Frontiers in immunology 13: 870577.
- Adi Aran, Maya Eylon, Moria Harel, Lola Polianski, Alina Nemirovski, et al. (2019) Lower circulating endocannabinoid levels in children with autism spectrum disorder. Molecular autism 10: 2.
- Orit Stolar, Ariela Hazan, Roni Enten Vissoker, Ibrahim Abu Kishk, Dana Barchel, et al. (2022) Medical cannabis for the treatment of comorbid symptoms in children with autism spectrum disorder: An interim analysis of biochemical safety. Frontiers in pharmacology 13: 977484.
- Bar Lev Schleider L, Mechoulam R, Saban N, Meiri G, Novack V, et al. (2019) Real life Experience of Medical Cannabis Treatment in Autism: Analysis of Safety and Efficacy. Scientific reports 9 (1): 200.
- Charlotte M Pretzsch, Bogdan Voinescu, Maria A Mendez, Robert Wichers, Laura Ajram, et al. (2019) The effect of cannabidiol (CBD) on low-frequency activity and functional connectivity in the brain of adults with and without autism spectrum disorder (ASD). Journal of psychopharmacology 33 (9): 1141-1148.
- Jenika Staben, Megan Koch, Keelee Reid, Jessica Muckerheide, Lauren Gilman, et al. (2023) Cannabidiol and cannabis-inspired terpene blends have acute prosocial effects in the BTBR mouse model of autism spectrum disorder. Frontiers in neuroscience 17: 1185737.
- Susanne W Duvall, Olivia Lindly, Katharine Zuckerman, Michael E Msall, Melissa Weddle, et al. (2019) Ethical Implications for Providers Regarding Cannabis Use in Children with Autism Spectrum Disorders. Pediatrics 143 (2): e20180558.
- Micha Hacohen, Orit E Stolar, Matitiahu Berkovitch, Odelia Elkana, Elkana Kohn, et al. (2022) Children and adolescents with ASD treated with CBD-rich cannabis exhibit significant improvements particularly in social symptoms: an open label study. Transl Psychiatry 12 (1): 375.
- Swenson K (2025) Beyond the hype: a comprehensive exploration of CBD’s biological impacts and mechanisms of action. Journal of Cannabis Research 7 (1): 24.
- Hamid Abbasi, Mohammad Mehdi Abbasi, Mohammadjavad Pasand, Mahshad Mohtadi, Farnush Bakhshimoghaddam, et al. (2024) Exploring the efficacy and safety of cannabidiol in individuals with epilepsy: an umbrella review of meta-analyses and systematic reviews. Inflammopharmacology 32 (5): 2987-3005.
- Paulo Fleury Teixeira, Fabio Viegas Caixeta, Leandro Cruz Ramires da Silva, Joaquim Pereira Brasil Neto, Renato Malcher-Lopes, et al. (2019) Effects of CBD-Enriched Cannabis sativa Extract on Autism Spectrum Disorder Symptoms: An Observational Study of 18 Participants Undergoing Compassionate Use. Frontiers in neurology 10: 1145.
- Adi Aran, Moria Harel, Hanoch Cassuto, Lola Polyansky, Aviad Schnapp, et al. (2021) Cannabinoid treatment for autism: a proof-of-concept randomized trial. Molecular autism 12 (1): 6.
- Shannon Inglet, Bradly Winter, Sarah E Yost, Sophia Entringer, Anh Lian, et al. (2020) Clinical Data for the Use of Cannabis-Based Treatments: A Comprehensive Review of the Literature. The Annals of pharmacotherapy 54 (11): 1109-1143.
- Kinga Borowicz Reutt, Julia Czernia, Marlena Krawczyk (2024) CBD in the Treatment of Epilepsy. Molecules (Basel, Switzerland) 29 (9): 1981.
- Patrícia Soares Silva Montagner, Wesley Medeiros, Leandro Cruz Ramires da Silva, Clarissa Nogueira Borges, Joaquim Brasil-Neto, et al. (2023) Individually tailored dosage regimen of full-spectrum Cannabis extracts for autistic core and comorbid symptoms: a real-life report of multi-symptomatic benefits. Frontiers in psychiatry 14: 1210155.
- R Ashish Jain, Ajay Prakash, Bikash Medhi (2019) Newer potential pharmacological targets for autism spectrum disorder. Indian journal of pharmacology 51 (4): 284-286.
- Shun Chin Liang, Cheuk Kwan Sun, Hsin Yi Fan, Weilun Chung, Ruu Fen Tzang, et al. (2022) Therapeutic effects of antidepressants for global improvement and subdomain symptoms of autism spectrum disorder: a systematic review and meta-analysis. Journal of psychiatry & neuroscience : JPN 47 (4): E299-e310.
- Pura Ballester, Cristina Espadas, Susana Almenara, Jordi Barrachina, Javier Muriel, et al. (2023) CYP2D6 Genotype and Pharmacovigilance Impact on Autism Spectrum Disorder: A Naturalistic Study with Extreme Phenotype Analysis. Pharmaceuticals (Basel, Switzerland) 16 (7): 954.
- Kyong Mee Chung, Eunsun Chung, Hoomyung Lee (2024) Behavioral Interventions for Autism Spectrum Disorder: A Brief Review and Guidelines with a Specific Focus on Applied Behavior Analysis. Journal of the Korean Academy of Child and Adolescent Psychiatry 35 (1): 29-38.
- Elizabeth A Fuller, Kelsey Oliver, Sarah F Vejnoska, Sally J Rogers (2020) The Effects of the Early Start Denver Model for Children with Autism Spectrum Disorder: A Meta-Analysis. Brain sciences 10 (6): 368.
- Theresa Eckes, Ulrike Buhlmann, Heinz Dieter Holling, Anne Möllmann (2023) Comprehensive ABA-based interventions in the treatment of children with autism spectrum disorder - a meta-analysis. BMC psychiatry 23 (1): 133.
- Qian Yu, Enyao Li, Liguo Li, Weiyi Liang (2020) Efficacy of Interventions Based on Applied Behavior Analysis for Autism Spectrum Disorder: A Meta-Analysis. Psychiatry investigation 17 (5): 432-443.
- Hongling Zeng, Shuo Liu, Run Huang, Yi Zhou, Jun Tang, et al. (2021) Effect of the TEACCH program on the rehabilitation of preschool children with autistic spectrum disorder: A randomized controlled trial. Journal of psychiatric research 138: 420-427.
- Hellings J (2023) Pharmacotherapy in autism spectrum disorders, including promising older drugs warranting trials. World journal of psychiatry 13 (6): 262-277.
- Hirst K, Zamzow RM, Stichter JP, Beversdorf DQ (2023) A Pilot Feasibility Study Assessing the Combined Effects of Early Behavioral Intervention and Propranolol on Autism Spectrum Disorder (ASD). Children (Basel) 10 (10): 1639.
- Devahuti Chaliha, John C Mamo, Matthew Albrecht, Virginie Lam, Ryu Takechi, et al. (2021) A Systematic Review of the MDMA Model to Address Social Impairment in Autism. Current neuropharmacology 19 (7): 1101-1154.
- Yana Sinai Gavrilov, Tali Gev, Irit Mor Snir, Giacomo Vivanti, Ofer Golan, et al. (2020) Integrating the Early Start Denver Model into Israeli community autism spectrum disorder preschools: Effectiveness and treatment response predictors. Autism : the international journal of research and practice 24 (8): 2081-2093.
- Daniela Bordini, Ana Cláudia Moya, Graccielle Rodrigues Cunha Asevedo, Cristiane Silvestre Paula, Décio Brunoni, et al. (2024) Exploring the Acquisition of Social Communication Skills in Children with Autism: Preliminary Findings from Applied Behavior Analysis (ABA), Parent Training, and Video Modeling. Brain sciences 14 (2): 172.
- Vincent Chin Hung Chen, Tsai Ching Hsu, Chiao Fan Lin, Jing Yu Huang, Yi Lung Chen, et al. (2022) Association of Risperidone with Gastric Cancer: Triangulation Method from Cell Study, Animal Study, and Cohort Study. Frontiers in pharmacology 13: 846455.




 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.