Research Article 
 Creative Commons, CC-BY
Creative Commons, CC-BY
A Multicentred Phase III Comparative Study between Single Rod Implant Monoplant® and Double Rod Implant Indoplant®
*Corresponding author: Eka Rusdianto Gunardi, Department of Obstetrics and Gynecology, Faculty of Medicine Universitas Indonesia/ Dr Cipto Mangunkusumo Hospital, Jakarta, Indonesia.
Received: August 23, 2019;Published: August 27, 2019
DOI: 10.34297/AJBSR.2019.04.000854
Abstract
Objective: To analyze to determine the effectiveness, safety, and time of both contraceptive methods between single rod implant monoplant® and double rod implant indoplant®.
Design/data identification: Clinical trial, ‘Open’ (‘open label ‘Randomized Clinical Trial).
Materials and methods: This study is a phase III clinical trial, conducted randomized and multicentre, comparing two types of contraceptive implants, namely Monoplant® and Indoplant® performed in Indonesian healthy women. Random blocks of tithe allocation and randomly assigned divided by 5 Monoplants and 5 Indoplants.
Results: Based on the interim results obtained from examining the twenty-fourth month this can be concluded as follows: all centre have completed the number of research subjects, the length of recruitment of subjects extends between 5.5 months to 16.5 months, continuity up to 24 months for contraception implantation of monoplant® or Indoplant® is still good, discontinued use for 24 months totaling 50 subjects, namely 27 subjects for monoplant® and 23 subjects for indoplant®, the main cause of discontinuation for both types of contraception is a disruption of menstrual patterns, there is one case of pregnancy that is most likely due to the initial selection error of the acceptor, there were 7 subjects who had to be removed because they wanted more children even though they had previously agreed to use implants for 3 years, there is no significant difference in levonorgestrel levels in the 24th month between monoplant® contraception and indoplant® contraception, the levonorgestrel levels of the two study groups in the 24th month were mostly located between 200 to 400 picograms which were still actively preventing ovulation.
Conclusion: There is no significant difference the effectiveness, safety, and time in levonorgestrel levels in the 24th month between single rod implant monoplant® and double rod implant indoplant®.
Keywords: Single Rod; Implant; Contraception
Introduction
Levonorgestrel (LNG) implant is the first contraception made in the 1960s. At that time pill contraception containing LNG was widely used, along with a new generation of intrauterine device (IUD) which was also successfully made. Whereas in Indonesia, the form or type of implant first introduced in 1982, Norplant®, which is a 6-stem implant with progesterone LNG active ingredient with 36 mg LNG concentration and effective for 5 (five) years. In 1989, contraception single rod was first introduced through phase II clinical trial research in Indonesia. This contraception has effectiveness for 3 (three) years and is named Implanon® with 3-keto desogestrel active ingredients produced by PT. Organon. Then the implant method was reintroduced which consisted of 2 (two) implants and had 3 years of effectiveness as Jadena® or Jadelle. Besides that, a new implant method, known as Indoplant®, was made up of 2 implants and had effectiveness for 3 years with the active ingredient levonorgestrel [1-3].
The limited variation in the choice of implants in Indonesia and for the implant of the new single rod known only Implanon®, it is very necessary to develop another single rod implant in Indonesia. Seeing the high public interest in the long-term contraceptive implant method compared to the IUD method, PT Triyasa Nagamas Farma tried to develop single rod implants, at affordable prices without reducing the effectiveness and security that was named Monoplant®. Monoplant contains 160 mg of levonorgestrel (LNG), with a tube length of 42.50-44.50 mm and a diameter of 2.52-2.72 mm with effectiveness for 3 (three) years. It is expected that Monoplant® single rod implants can provide effectiveness, the same safety as other implants and easier installation. Phase III clinical trial study on Comparison of contraceptives studies in single rod Monoplant®, with implants of double rod Indoplant®, is a continuation of phase II clinical trials, namely Pharmacokinetic and Pharmacodynamic Study of Monoplant® carried out in 2010. Agency POM RI recommends that before Phase III clinical trials be carried out, phase II clinical trials need to be done first, considering that Monoplant® is a new contraceptive technique or method of single rod contraception that will be developed in Indonesia. If from the results of Pharmacokinetic and Pharmacodynamic Monoplant ® within 6 months LNG levels in the blood show levels of 200 pg / ml or more, then Phase III Clinical Test studies are permitted. From 2007 to 2010 Monoplant® Phase II Clinical Tests were carried out in the Jakarta centre on 30 female subjects with 36 months of observation. The results of this study show that LNG levels for 12 months using Monoplant® averaged above 200 pg / ml (260.81 + 97.25). This figure shows that Monoplant has met the requirements set by BPOM RI, so Phase Clinical Tests III can be implemented. However, LNG levels in the blood fluctuate; in the 18th month LNG levels dropped and the 24th month rose again. Thus BPOM RI recommends, in phase III clinical trials, this study also looks at LNG levels in the blood of subjects using Monoplant® and Indoplant® in 5 (five) observation points, namely before being installed, at 6 months of use, 12 months, 24 months and 36 months of use [4-8].
Materials and Methods
This study is a phase III clinical trial, ‘Open’ (‘open label ‘Randomized Clinical Trial), conducted randomized and multicentre, comparing two types of contraceptive implants, namely Monoplant ® and Indoplant® performed in Indonesian healthy women. Random blocks of tithe allocation and randomly assigned divided by 5 Monoplants and 5 Indoplants.
The sample is calculated statistically, with a sample size formula for comparison of proportions between two sample research groups (Pocock): using the formula above, the number of subjects for each group treatment was obtained by 399. Based on consideration of the drop-out (approximate drop-out) of around 10%, the total sample required was 444 subjects for each group treatment and rounded up to 450 subjects for each group or group.
The total sample observed in this study: 450 subjects of Monoplant ® users and 450 subjects of women using Indoplant® in 5 (five) centre in Indonesia, namely Jakarta, Semarang, Surabaya, Palembang and Makassar. Each centre recruits 180 subjects (90 Monoplant acceptors and 90 Indoplant acceptors).
Subjects included in this study have fulfilled all of the following criteria: women of childbearing age aged 20 - 35 years (sexually active), physically and mentally healthy, not pregnant, still having active sexual relations so that they still have the risk of becoming pregnant, not exposed to hormonal contraception in the last 3 months, already got an explanation and understood the purpose, risks and benefits of the research and signed the informed consent, willing to return to the clinic for a repeat visit, willing to only use implants during the study in the next 3 (three) years.
A woman is excluded as the subject of this study if she experiences one of the following histories or events: has a family history of all types of cancer, abnormal or undiagnosed genital bleeding, severe thromboembolism or cardiovascular, have mental illness, depression, epilepsy, tuberculosis, often severe headaches, having a history of diabetes mellitus, have a history of liver disease or active liver disease, in regular care or just with drugs that induce liver enzymes, such as: barbiturates, phenytoin, carbamazepine or rifampicin, hypertension (systolic> 160 mmHg; diastolic> 100 mmHg), heavy hirsutism, participate in other clinical research activities in the last three months.
Observations on subjects were conducted for 3 years. Blood collection for LNG level examination was carried out in a subsample (33 subjects) for each implant of Monoplant and Indoplant, especially on subjects in the Jakarta centre. Examination of LNG levels in the blood is carried out at PT. Equilab International.
Results
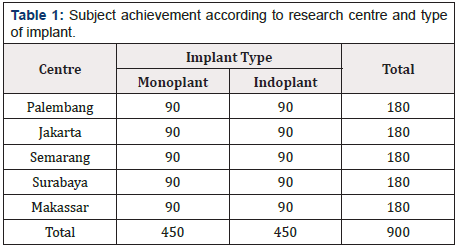
This progress report basically displays the results of phase III clinical trials and results of examination of LNG in the blood for 24 months using implants of Monoplant and Indoplant, because the return visit data that is complete and can be analyzed to date is data up to a 24-month return visit. Phase III clinical trials were carried out on 450 Monoplant and 450 Indoplant subjects, which were conducted in 5 (five) research centre. While LNG levels in the blood are only carried out in one research centre, the Jakarta centre, which was conducted on 33 Monoplant user sub-samples and 33 sub-samples of Indoplant® users Table 1 shows the results of the subject according to the research centre and type of implant. In the table, it can be seen that in approximately 18 months, each centre has collected 180 subjects or 900 total subjects in 5 (five) research centre.
Nearly all centre cannot meet the number of subjects within the set time. Among the 5 centre, which are the fastest and according to the time of the initial recruitment, which for approximately 6 months is the Palembang centre (6 months). The recruitment time for the Jakarta centre is 12 months, Semarang is 15 months, Surabaya is 13 months and Makassar is 17 months. The extension of the subject recruitment time is because implant services are now easily accessible to almost all government and private health facilities that work with BPJS and do not pay, making it difficult to get prospective subjects to participate in this clinical trial. Another reason is, besides the strict exclusion criteria such as having to be hormone free in the last 3 months and the narrow age limit set (Table 1).
There is no difference in systole or diastole between Indoplant and Monoplant groups, as well as between centre. The systole pressure in Semarang centre is the highest compared to other centre with a value between 117.0 + 10.0 mmHg for Monoplant and 117.9 + 10.6 for Indoplant. The lowest sistole was found in Makassar centre namely 109.4 + 11.4 (Monoplant) and 109.3 + 10.3 (Indoplant). The lowest diastole was found in Surabaya centre for Monoplant and Indoplant subjects (73.8 +7.6 mmHg and 74.6 + 8.2 mmHg).
Obstetric history seen from the results of IVA test, number of parities, number of pregnancies, menstrual pattern, desire to have more children and breastfeeding. The results of the analysis showed that the subjects who received Monoplant or Indoplant had an average of children born alive between 2-4 children. The average number of children is found in Makassar centre, especially for Monoplant subjects while for Indoplant subjects in Palembang centre. The Semarang centre has the lowest average child described from Monoplant or Indoplant. There is no difference in the number of children born alive which means between Monoplant and Indoplant subjects, judging from the level of parity.
Most of the total subjects who took part in this study especially 4 research centre did not want to add more children; even Semarang centre are mostly 90 percent (Monoplant) and 84.4 percent (indoplant) already don’t want to add more children.
Seen from breastfeeding status at the time of recruitment, more than half of Monoplant subjects in the Semarang and Jakarta centre were found not breastfeeding, namely 57.8 percent and 52.2 percent. While the other three centre (Makassar, Surabaya and Palembang) mostly breastfeed at the time of recruitment. A similar pattern is found in Indoplant users, in the Surabaya, Semarang, Jakarta centre, which is 51.1 percent, 58.9 percent, 56.7 percent, most of whom are not breastfeeding. While the other two centre (Palembang, Makassar) are mostly breastfeeding at the time of recruitment.
To fulfill the inclusion requirements, the subjects were asked whether in the last 3 months they had used hormonal contraception. According to the history of hormonal use, most subjects had used hormonal contraception more than three months ago, especially the type of injection. Centre with the highest proportion did not use hormones in the past 3 months, for Monoplant subjects namely Semarang and Surabaya centre, 86.7 percent and 80.0 percent. The same centre was also experienced for Indoplant subjects, namely 85.6 percent and 87.8 percent.
Most of the subjects, both Monoplant and Indoplant said that they had no difficulty at the time of the implant placement. Only the Palembang and Semarang centre say there is a 1.1 percent difficulty for Monoplant. As for the installation of Indoplants, there was 1.1 percent difficulty in the Palembang centre, 2.2 percent found in Semarang and Surabaya centre. While this shows the level of difficulty of implant placement of Indoplant stem II is relatively more than Monoplant. There was no significant difference seen from the length of installation of the two types of implants. The median duration of Monoplant installation is 3.23 minutes (minimum 0.48 and maximum 15 minutes) while for Indoplant is 3.75 minutes (minimum 0.83 and maximum 15.25 minutes).
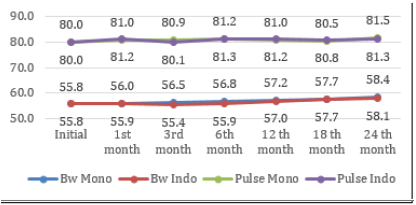
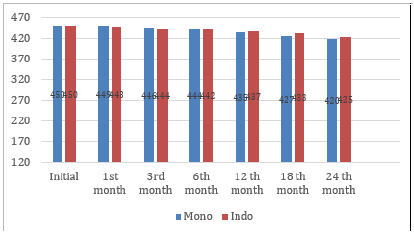
Until now, follow-up or repeat visits from several subjects have been running for 36 months. However, the complete re-visit up to now is a 24-month visit, while the 30-month and 36-month visit is not complete. So that the return visit that can be analyzed in full is data up to a 24-month return visit. For data analysis, further research will be immediately proposed when recording acceptor health data on the 30th birthday visit and the 36th month visit is complete Figure 1 shows that from the start of the implant placement to the 24-month, there did not appear to be a change in the average body weight and pulse. There was a slight fluctuation in the two variables, but still within the normal range for the two study groups.
In addition to body weight and pulse, it turned out that changes in systolic and diastolic blood pressure in the two study groups also tended to be stable from the beginning to the twenty-fourth month (Figure 1).
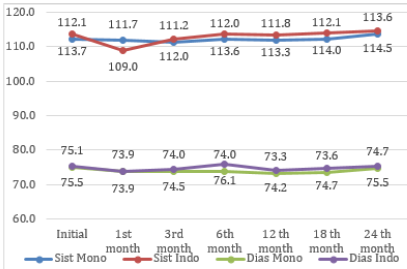
Figure 2 shows that the subject’s systolic and diastolic blood pressure up to a 24-month did not show any significant changes and is still in a normal condition.
The tendency of weight stability, pulse rate, and systolic and diastolic blood pressure shows a high level of security from the cardiovascular aspect for users of the two types of implants being studied. Cardiovascular disorders can be categorized as a systemic disorder that is quite serious and can trigger other organic disorders (Figure 2).
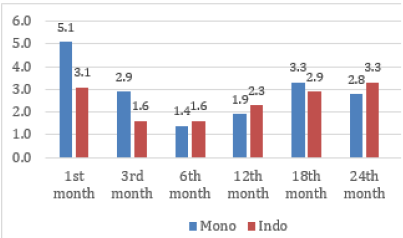
Subject complaints about health in the form of pain around the reproductive organs or known as dyspareunia after the use of Monoplant and Indoplant found in the first month, third, sixth, and twelfth. Dyspareunia complaints tend to be higher in Monoplant subjects than the Indo-plant. Within a period of 6 months, there was a noticeable increase in complaints of dyspareunia for these two implants, but in the 12th month there was a slight increase in complaints of dyspareunia to 1.9 percent for Monoplant and 2.3 percent for Indoplant. In the 18th and 24th months there was also an increase in dyspareunia complaints in both groups which reached 3.3%, but this fluctuation in complaints of dyspareunia was still within normal limits (Figure 3).
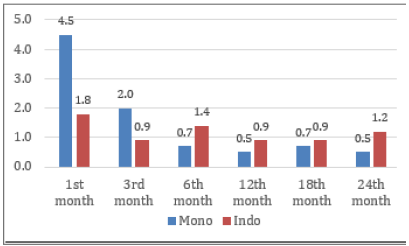
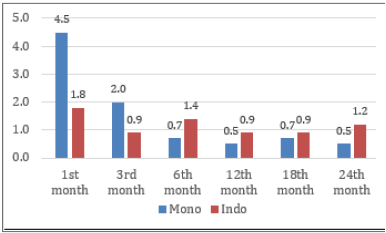
Hormonal use for some subjects can cause complaints of breast pain. The same pattern with dyspareunia complaints also appears here. After observing for 24 months the use of implants of Monoplant and Indoplant, there was a decrease in pain complaints in the breast. In Monoplant subjects, the incidence of breast pain dropped from 4.5 percent in the first month to 0.5 percent in the twelfth month and twenty-four. For Indoplant subjects, from 1.8 percent the subject fell to 0.9 percent in the 12th month even though it rose slightly again to 1.2% in the 24th month (Figure 4).
The proportion of amenorrhea in Monoplant subjects ranged from 24.9 percent (month 1) to 26.8 percent (month 3) and in month 6 to 23.9 percent then slightly increased to 25.3 percent on month 12. Compared to Monoplant, Indoplant KB implants until the 6th month are relatively higher amenorrhea complaints; however, in the 12th month, amenorrhea complaints for Indoplant decreased to 22.7 percent, so complaints of Indoplant amenorrhea were slightly lower than Monoplant. In the 18th to the 24th month, complaints of amenorrhoea were a priority among the two types of contraception, which was around 16.2% (Figure 5).
Up to 24 months of observations, it was seen that the continuity of the use of the Monoplant and Indoplant implants was still high; and there are no significant differences. There was a decrease in users of Monoplant by 30 subjects or 6.7 percent, so 93.3 percent still used in the 24th month. Similarly for Indoplant users, it dropped by 25 subjects or 5.6 percent, so Indoplant users on the 24th month still reached 94.4 percent. From this data it can be seen that the continuity of Monoplant users for 24 months of observation is relatively lower than the implant of Indoplant (Figure 6).
Data on the continuity of Monoplant and Indoplant contraceptive use is very high until the second year of use. Of course, the level of acceptance of these acceptors still has to be analyzed for subsequent years after recording complete research data is completed.
Discussion
There are currently several innovative contraceptive implant systems under development. Monoplant is a single rod contraceptive implant and it provides contraceptive protection for 3 years. Mechanism of action of monoplant® is by ovulation inhibition and increase in viscosity of cervical mucus. The most common side effects associated with Implanon are irregular periods, weight gain, acne, headache and breast tenderness. Single rod implant will have the advantages of insertion and removal easily, the efficacy and safety are similar to Indoplant®. In terms of efficacy a quantitative measurement has been done. We measured Levonogestrel concentration in serum up till 6 months of insertion. The result is LNG serum concentration is far above the contraceptive level with mean of 337.5 pg/ml (min 200 pg/ml), giving suitability in Indonesian people and proving its efficacy. We had already performed research about the efficacy of Monoplant® and Indoplant® as contraceptive methods: a comparative study. Researchers want to determine the effectiveness, safety, and time of insertion between Monoplant® with Indoplant® to prevent pregnancy [9-11].
There was no difficulty or complication during insertion or removal in any subject. Injury to antebrachial cutaneous nerve during insertion or removal has been reported. Injury to ulnar nerve during insertion has been reported also. There was expulsion of monoplant® in one subject on 3rd day of insertion. Commonest side effect was irregular bleeding pv in 27% cases, however, only 5% cases got monoplant® removed due to this disorder. Prolonged spotting was reported in 23% cases but only 1% cases had monoplant® removal due to this disorder. 24% cases complained of amenorrhea but only 4.5% cases got removal done due to amenorrhea. Polymenorrhagia was observed in 22.5 and 16% cases got removal done due to polymenorrhagia. Gezginck et al reported less incidence of irregular bleeding P/v (17.5%), but they reported a much higher incidence of amenorrhea (41.25%). Weight gain 5 kg was observed in 7.5% cases, but no subject got removal done due to this reason. Side effects such as breast tenderness, acne, headache and dizziness, depressive mood disorders, pelvic pain and loss of libido have also been reported, but they were not observed in the present study. Hiddalgo et al. reported ovarian cysts in 5.2% cases of Implanon at 3 months, 7.2% at 6 months and 26.7% at 12 months, but they concluded that these ovarian cysts were transient and should not be interpreted as pathological. No case of ovarian cysts was observed in the present study. There was no failure of contraception in the present study. Harrison-Woolrych and Hill have reported approximate failure rate of 1 per 1000 insertions (218 out of 204486). Pregnancy due to monoplant® failure has also been reported by Hamontri and Weerkul. One case of ectopic pregnancy following monoplant® failure has been reported. Monoplant ® has to be used as a spacing method hence return of ovulation and fertility is an important parameter to be studied. 40% cases had return of ovulation within 1 month which was confirmed by progesterone estimation. Among the cases who did not adopt any alternative contraception methods, 95.8% conceived within 12 months. There was only one case who did not conceive within 1 year and she was suffering from hypothyroidism [12-15].
The data obtained showed no significant difference in the effectiveness of both contraceptive methods. In addition, side effects such as menstrual disorders and weight gain did not differ significantly in those study groups. From that research, both implants have the same effectiveness, safety, no differences in weight gain and menstrual disorders during 1, 3, and 6 months follow-up. However, the insertion of Monoplant® are faster than Indoplant®. Monoplant® can be used as a contraceptive method with the same effectiveness and safety as Indoplant®, yet with shorter insertion time [16,17] We believe that this advancement will bring a new hope to increase the efficacy of national contraception program.
Conclusion
Based on the interim results obtained from examining the twenty- fourth month this can be concluded as follows, there is no significant difference the effectiveness, safety, and time in levonorgestrel levels in the 24th month between single rod implant monoplant® and double rod implant indoplant®. Single rod implant monoplant ® is safe, effective, well accepted method of contraception. We believe that this advancement will bring a new hope to increase the efficacy of national contraception program.
Acknowledgment
Thanks are due to BKKBN, Puslitbang KB and KS team as a coordinator and management data, Triyasa team as a sponsorship and YKB as a CRO.
References
- Brown A (2010) Long-term contraceptives. Best Pract Res Clin Obstet Gynaecol 24(5): 617-631.
- McDonald Mosley R, Burke AE (2010) Contraceptive implants. Semin Reprod Med 28(2): 110-117.
- Brache V, Faundes A, Alvarez F, Garcia AG (2006) Transition from Norplant to Jadelle in a clinic with extensive experience providing contraceptive implants. Contraception 73(4): 364-367.
- Affandi B, Wijayanegara H, Permadi W, Soedarto, Hasan M, et al. (2005) Multi centers research comparative study of Indoplant® and Norplant® in Indonesia.
- Gunardi ER, Affandi B, Juliaan F (2011) Levonorgestrel concentration in a single rod implant users for six months. Indones J Obstet Gynecol 35(3): 122-127.
- Gunardi ER, Affandi B, Muchtar A (2011) Monoplant® the Indonesian implant: the overview of implant and its development. Indones J Obstet Gynecol 35(1): 40-46.
- Lewis RA, Taylor D, Natavio MF, Melamed A, Sokol R, et al. (2009) Effects of the levonorgestrel intrauterine system (LNG-IUS) on cervical mucus quality and sperm penetration. Fertility and Sterility 92(3, Supplement 1): S27.
- Steiner MJ, Lopez LM, Grimes DA, Cheng L, Shelton J, et al. (2010) Sino- implant (II)- a levonorgestrel-releasing two-rod implant: systematic review of the randomized controlled trials. Contraception 81(3): 197- 201.
- Gunardi E, Ballo F (2017) The Efficacy of Monoplant® and Indoplant® as Contraceptive Methods: A Comparative Study. Indones J Obstet Gynecol 5(2): 94-98.
- Gunardi E, Affandi B (2014) Serum levonorgestrel concentration and cervical mucus viscosity after six months of monoplant implantation. Med J Indones 23(1): 25-29.
- Gezginc K, Belci O, Karatayli R, Colakoglu MC (2007) Contraceptive efficacy, side effects of implanon(R). Eur J Contracep Reprod Health Care 12(4): 362-365.
- Hidalgo MM, Lisondo C, Juliato CT, Espejo-Arce X, Monteiro I, et al. (2006) Ovarian cysts in users of Implanon, Jadelle subdermal contraception implants. Contra- caption 73(5): 532-536.
- Harrrison Woolrych M, Hill R (2005) Unintended pregnancies with the etonogestrel implant (Implanon): a case series from postmarket- ing experience in Australia. Contraception 71(4): 306-308.
- Hamontri S, Weerkul W (2007) Implanon failure. J Med Assoc Thai 90(2): 381-383.
- Gunardi E (2019) Indonesian Single Rod Implant: A New Breakthrough. Am J Biomed Sci & Res 1(6): 252-253.
- Curtis KM (2002) Safety of implantable contraceptives for women: data from observational studies. Contraception 65(1): 85-96.
- Gunardi E, Khusen D (2019) New Single Rod Implant Innovation in Indonesia. Am J Biomed Sci & Res 3(4): 347-348.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.