Research Article 
 Creative Commons, CC-BY
Creative Commons, CC-BY
The Incidence of Sarcopenia is Low Among Israeli Liver Transplantation Patients and Associated with Lower Survival Estimates
*Corresponding author: Yael Milgrom, The Liver Unit, Hadassah University Hospital, Ein Kerem, Jerusalem, 911200, Israel.
Received:June 16, 2020;Published: July 07, 2020
DOI: 10.34297/AJBSR.2020.09.001402
Abstract
Purpose: Sarcopenic patients have reduced musculoskeletal capacity and have poorer post- surgical outcomes. High rates of sarcopenia have been reported among Western liver transplantation candidates. The purpose of this study was to determine the incidence of sarcopenia in an Israeli liver transplant program and assess its effect on post-transplant outcomes.
Methods: An observational retrospective study was performed using an updated database which included demographic and outcome measures of 100 consecutive liver transplant patients from deceased donors at the Hadassah Medical Center 2002-2012. Sarcopenia was assessed for those with available pre transplant abdominal CT studies. Psoas muscle measurements were obtained via CT and Lean Psoas Area (LPA) was calculated. Subjects were classified into sarcopenic according to an accepted convention of psoas LPA.
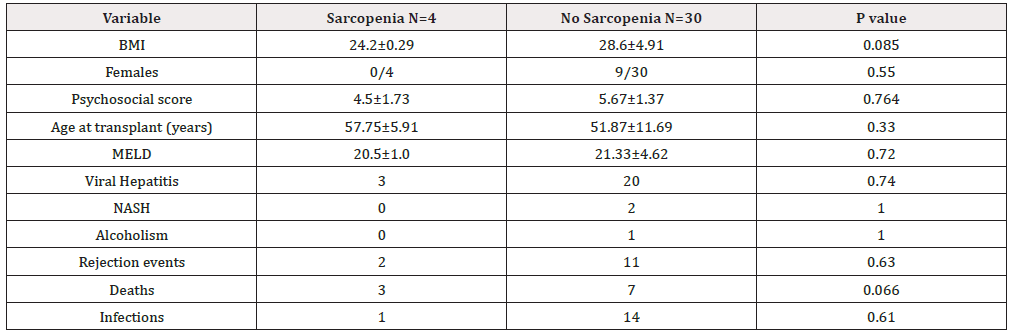
Results: Thirty-four patients had CT studies appropriate for the sarcopenia analysis. Nine were females (26%) and 25 (74%) males. Mean transplant age was 52.6±11.3 and post-transplant follow up 6.3±3.7 years. By the Dolgin criteria only four males (12%) had sarcopenia. The productlimit survival estimate showed a statistically greater estimated survival for non-sarcopenia patients than for sarcopenia patients (log-rank test p=0.045), with a survival estimate at 98 months of 79% for non-sarcopenia patients and 25% for sarcopenia patients.
Conclusions: Because Israeli sarcopenic liver transplantation patients have lower survival estimates along with inherent physical limitations compared to non-sarcopenic patients, it make sarcopenia an additional valuable variable for listing liver transplantation candidates, especially during the COVID-19 pandemic when donor supply to need mismatches have increased.
Keywords: Liver transplantation; Sarcopenia incidence; Outcome; Israel
Introduction
Medicine’s musculoskeletal focus has centered for many decades on the epidemiology, prevention and the associated adverse effects of osteoporosis [1]. It is only more recently that sarcopenia has been acknowledged to be a major musculoskeletal problem and to also be part of the equation of fracture risk [2].
Rosenberg first proposed the term sarcopenia to described the naturally occurring age- related decrease in muscle mass, but sarcopenia is also potentially associated with various medical conditions, patient genetics and lifestyle [3]. The decreased muscle mass of sarcopenia directly results in decreased muscular strength and physical function.
Sarcopenia has also been reported to be associated with poor outcomes and increased mortality after major surgery [4]. In liver transplantation, studies regarding the correlation of sarcopenia with increased transplant and post-transplant mortality, have shown inconsistent results [5,6]. For example, reported that sarcopenia was associated with a higher risk of perioperative bacterial infection and longer post-transplantation hospitalization, but not lower transplant survival. When they did a sub analysis of males with extreme sacropenia they found that they had a significantly inferior post liver transplant survival (71±11 months) than the rest of the liver transplant population (132±11 months).
The presence of sarcopenia has been assessed by calculations derived from either DEXA scans, CT scans, bioelectric impedance measurements, or grip strength and walking speed [6]. What is sarcopenia and what is normal muscle mass is defined for each of the assessment methods, but values often differ between populations. The European Working Group on Sarcopenia in Older People (EWGSOP) has published criteria for sarcopenia for the European population [7]. Because of differences in their physique, different criteria have been published for the Asian population [8]. In some studies sarcopenia has been defined based on the measured value being one standard deviation below the average for the study population. All of this can make comparison of research results between clinical studies problematic.
Pre-transplant sarcopenia has been reported to exist in 50% of liver transplant patients in the US [9]. Similar rates have been reported in a Canadian study as well [6]. An Italian study reported an even higher sarcopenic rate of 76% among patients eligible for liver transplantation [5]. The Israel liver transplant program is of a much smaller scale than that of North American and Western European programs. There are three adult liver transplant centers in Israel compared to 130 in the US. The US active waiting list for liver transplantation exceeded the number of transplants by 60% (13,192 on the waiting list versus 8250 transplantations) in 2018. In Israel the active adult transplantation list exceeded the number of transplants by only 10% in the same year, with 110 patients on the waiting list and 100 liver transplantations performed from cadaver donors. The mismatch between donor supply and need in many countries is likely to increase because of the COVID-19 pandemic.
The current report compares the sarcopenia incidence among Israeli adult liver transplant patients with those from major North American centers. The transplantation outcomes for the Israeli cohort are presented for those with and without sarcopenia.
Materials and Methods
Study Design and Population
The data of 100 successive liver transplant recipients in the Hadassah Medical Center liver transplantation database between 2002-2012 were reviewed. The period was chosen to allow for sufficient long-term follow up to address study endpoints and outcomes. The database includes the reason for transplant, gender, age, height, weight, BMI, MELD (model for end stage liver disease) and psychosocial scoring at transplant [10]. Also listed are the dates of clinic visits, comorbidity and mortality data including death date, rejection, biliary complications, nephrotic complications, infection, hyperkalemia and diabetes. All patients from this cohort who were older than 18 years at transplantation, underwent a first-time liver transplant from a deceased donor and had abdominal CT studies with sagittal reconstruction within the 25 months prior to transplant, were available for sarcopenia assessments. The rate of sarcopenia of the present cohort was compared to that of published American, [9] and Canadian [6] studies.
Sarcopenia measurement
Pre-transplant psoas muscle size (cross-sectional area, in mm2) and quality (density, Hounsfield units [HU]), which included both left and right psoas muscles, were measured at the L4 vertebral level superior plate. To minimize measurement bias, all measurements and calculations where done by the same investigator (YM). The investigator was trained by a senior radiologist to perform calculations. The plane of the L4 vertebral level superior plate was determined on sagittal reconstruction. The axial image related to this level was used to determine the psoas area bilaterally. In accordance with the method of Dolgin et al. [9], psoas muscle measurements were combined to create the following variables: total psoas area (TPA; left + right cross-sectional area, mm2), mean density ([left + right density]/2, HU) and lean psoas area (LPA; TPA × [mean density+85]/170, mm2).
Sarcopenia cutoffs
The presence and absence of sarcopenia was defined for male subjects and female subjects separately according to the staturenormalized lean psoas area cutoff point of 1480.4 mm2 for males and 974.8mm2 for females [9]. These values were determined by Dolgin et al. [8] based on the Michigan Surgical Quality Collaborative database of 1,279 patients who underwent elective surgery at a single institution and had CT based LPA calculations [11]. The cut off point for sarcopenia was determined as one standard deviation below the average of lean psoas area.
Statistical analysis
Statistical analysis was performed using the Statistical Analysis System (SAS Institute Inc., Cary, North Carolina, USA, version 9.4). Nominal data were assessed with the chi-square test and Fischer’s exact test. Normally distributed interval data were compared across the groups, using 2-tail Student’s t-test. Survival estimates for sarcropenic and non-sacropenic patients following transplant were done using the LIFETEST PROCEDURE (SAS).
Results
34 patients had CT studies appropriate for the sarcopenia analysis. Nine were females and 25 males. Based on the Dolgin’s criteria [9] for determining sarcopenia based on lean psosas area measured on CTs, only four patients (12%), all males, had sarcopenia.
No statistically significant difference was found between the major outcome variables studied and patient demographics versus patients with and without sarcopenia (Table 1). There was a trend (p=0.066) for transplantation patients with sarcopenia to have a higher death rate than those without sarcopenia.
The product-limit survival estimate Figure 1 showed a statistically greater estimated survival for non-sarcopenia patients than for sarcopenia patients ( log-rank test p=0.045), with a survival estimate at 98 months of 79% for non-sarcopenia patients and 25% for sarcopenia patients.
Discussion
The presence of sarcopenia in itself affects the quality of life by lowering one’s daily activity level [7]. Worldwide, its prevalence is 5-13% in those in the seventh decade of life and increases to 11-50% in those above 80 years old [12,13]. Adding the physical limitations imposed by sarcopenia to the liver transplantation patient, can compromise the quality of life achievements of the transplant. The current study indicates that this co-morbidity is fortunately present in a minority of Israel liver transplant patients (12%) as opposed to the 50% incidences reported from US and Canadian studies using the same evaluation criteria for sarcopenia [9]. The study also found a statistically greater estimated survival for non sacropenia patients than for sarcopenia patients (p=0.045). At 98 months, survival was 79% for non-sarcopenia patients compared to 25% for sarcopenia patients.
Sarcopenia should not be confused with cachexia. Cachexia has been defined as a multifactorial syndrome with muscle, fat and general weight loss coupled with increased protein catabolism secondary to underlying disease. Sarcopenia is only the loss of muscle mass. The diagnostic criteria for sarcopenia in liver disease patients are either CT psoas muscle mass calculations such as used in this study, bioelectrical impedance analysis or simple grip strength measurements. For any given evaluation method, the sarcopenia cutoff value has been alternately defined as a value either one or two standard deviations from the mean value of a reference group [14]. The reference groups used also vary between studies [4,6-9,13,14]. It would appear that the Israeli incidence of sarcopenia in liver transplant patients in addition to being lower than North American counterparts, is also much lower than the Japanese. The Japan Society of Hepatology has published guidelines for sarcopenia in liver disease [13]. They report the incidence of sarcopenia in Japanese cirrhosis patients to be in the range of 10%-70%. They also found that among patients undergoing liver transplantation, sarcopenia is associated with poor post-transplant prognosis. In all of these assessments the Japanese cutoff values for sarcopenia are lower than those of the values used in Europe [7]. For muscle mass calculations the cut-off values are 4-6% less.
This study is limited in that it is retrospective in nature, based on small numbers when compared with North American Studies and only 34 patients had CT studies of sufficient quality to make the sarcopenia analysis. The statistical finding of lower post-transplant survival among sarcopenic patients, requires further continued study. Nevertheless, the finding of a low incidence of sarcopenia in the Israeli transplant population is an important because it potentially makes it a relevant transplantation screening tool in Israel. A high prevalence of sarcopenia, such as has been reported in North America, would potentially affect transplantation decisions in 50% of patients, making its filtering effect too coarse to be useful for that population. A major difference between Israeli and North American transplant patients is in the etiology of their liver disease. In 24% of US transplant patients [9] and 19% of Canadian patients5 the etiology is alcoholic hepatitis. In this study there was only one patient who suffered from alcoholic hepatitis (2.9%). The composition of the population needing liver transplant is likely to changed in the future. Because of improved antiviral treatment, the incidence of viral hepatitis patients can be expected to decrease. On the other hand, because of the increasing incidence of obesity, the number of non-alcoholic fatty liver disease (NASH) patients can be expected to increase.
Over the decade 2008-2018 the number of liver cadaver transplants performed yearly in Israel has ranged from 31 to 101, with a mean of 61.5±14.3 transplant done a year. At the same time the number of candidates waiting each year for liver transplant has ranged from 101-151. Each year new donors need to be found. The number of donors is a function of multiple factors, many of them entirely based on random events.
Conclusion
Given the possible mismatch between need and supply, this study indicates that sarcopenia might be a factor to consider when determining the list order in liver transplantation programs. During the COVID-19 pandemic this mismatch is likely to become more prevalent.
Acknowledgments
The authors wish to acknowledge the contributions of Yusef Asraq, Mohamad Masarowah and Hadar Merhav to the study. The study was supported by internal funding from the Hadassah Medical Center.
Disclosure
None of the authors has a conflict of interest in this work.
References
- Albright F (1947) Osteoporosis Ann Intern Med 27(6): 861-882.
- Reginster JY, Beaudart C, Buckinx F, Olivier Bruyère (2016) Osteoporosis and sarcopenia: two diseases or one? Curr Opin Clin Nutr Metab Care 19(1): 31-36.
- Rosenberg IH (1989) Epidemiologic and methodologic problems in determining nutritional status of older people. Am J Clin Nutr 50(5 Suppl): 1231-1233.
- Friedman J, Lussiez A, Sullivan J, Stewart Wang, Michael Englesbe (2015) Implications of sarcopenia in major surgery. Nutr Clin Pract 30(2): 175-179.
- Kim G, Kang SH, Kim MY, Soon Koo Baik (2017) Prognostic value of sarcopenia in patients with liver cirrhosis: A systematic review and meta-analysis. PLoS One 12(10): e0186990.
- Montano Loza AJ, Mesa-Junco J, Baracos VE, Carla M M Prado, Mang Ma, et al. (2014) Severe muscle depletion predicts postoperative length of stay but is not associated with survival after liver transplantation. Liver Transpl 20(6): 640-648.
- Cruz Jentoft AJ, Baeyens JP, Bauer JM, Yves Boirie, Tommy Cederholm, et al. (2010) Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing 39(4): 412-423.
- Chen LK, Liu LK, Woo J, Prasert Assantachai, Tung Wai Auyeung, et al. (2014) Sarcopenia in Asia: Consensus report of the Asia working group in sarcopenia. J Am Med Dir Assoc 15(2): 95-101.
- Dolgin NH, Smith AJ, Harrington SG, Babak Movahedi, Paulo N A Martins, et al. (2019) Association between Sarcopenia and Functional Status in Liver Transplant Patients. Exp Clin Transplant 17(5): 653-664.
- Benson AA, Rowe M, Eid A, Keren Bluth, Hadar Merhav, et al. (2017) Pre-liver transplant psychosocial evaluation predicts post-transplantation outcomes. Psychol Health Med 23: 788-796.
- Kirk PS, Friedman JF, Cron DC, Michael N Terjimanian, Stewart C Wang, et al. (2015) One-year postoperative resource utilization in sarcopenic patients. J Surg Res 199(1): 51-55.
- Dodds RM, Roberts HC, Cooper C, Avan Aihie Sayer (2015) The epidemiology of sarcopenia. J Clin Densitom 18(4): 461-468.
- Nishikawa H, Shiraki M, Hiramatsu A, Kyoji Moriya, Keisuke Hino, et al. (2016) Japan Society of Hepatology guidelines for sarcopenia in liver disease (1st edition): Recommendation from the working group for creation of sarcopenia assessment criteria. Hepatol Res 46(10): 951-963.
- Kim TN, Choi KM (2013) Sarcopenia: definition, epidemiology, and pathophysiology. J Bone Metab 20(1): 1-10.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.