Research Article 
 Creative Commons, CC-BY
Creative Commons, CC-BY
The Advantages of The Alpha-Glycosylated Form of Hesperidin, Increased Bioavailability and Beneficial Effects for Human Health
*Corresponding author: Eva Künnemann, MVS Pharma GmbH.
Received: November 14, 2024; Published: November 20, 2024
DOI: 10.34297/AJBSR.2024.24.003250
Abstract
Hesperidin is a bioflavonoid found in high concentrations in citrus fruits. It benefits cardiovascular function, type II diabetes, anti-inflammation, and cutaneous functions, including wound healing, UV protection, anti-inflammation, antimicrobial, anti-skin cancer, and skin lightening. It has low water solubility and poor oral bioavailability. To improve its solubility and bioavailability, Alpha-Glucosyl Hesperidin (AGH) has been synthesized from hesperidin by transglucosylase using cyclodextrin glucotransferase. It is 10,000×more soluble and has 3.7×higher bioavailability than hesperidin. AGH has various pharmacological effects, such as hypolipidemic and hypouricemic effects, and may therefore be a useful supplement or drug for people who are fighting with obesity and fat loss. In this review we summarize the potential benefits of alpha-glucosyl hesperidin and add points for safety and hesperidin use from a commercial point of view.
Keywords: Hesperidin, Bioavailability, Alpha glycosylated hesperidin
Abbreviations: AGH: Alpha Glycosyl Hesperidin.
Origin and Occurrence of Hesperidin
The French pharmacist Lebreton was the first to isolate hesperidin in 1828 from the white inner layer of the peel of oranges, the so-called mesocarp or albedo [1]. It is a major flavonoid compound, present in orange and lemon fruits. Figure 1 shows a citric fruit together with isolated hesperidin. It is also common in other citrus fruits. In the peels of oranges and lemons hesperidin is the main flavonoid and is found there alongside hesperetin. Sweet oranges (Citrus sinensis) and tangelos are considered the most important source of this bioflavonoid. In some orange varieties hesperidin makes up up to 4.1% of the dry matter of the peel. 470–761 mg/L of Hesperidin is normally present in orange juice [2]. It can vary widely between different fruits [3]. If the juice contains crushed fruit it has more of this flavonoid than without pulp. It can be extracted from orange peels, taking care to extract a larger amount of the white inner layer. Naringin, hesperidin, neohesperidin, narirutin, and eriocitrin are the major flavanone-7-O-glycoside constituents in citrus fruit juices and can be separated as diastereomers by multidimensional liquid chromatography [4]. Hesperidin was present in orange juices almost exclusively as the 2S isomer. Hesperidin can also be found in green vegetables and in the uppercase sections of the Rubiceae genus as well as in the roots and entire grasslands of cruciferous plants [5] (Figure 1).
Chemistry
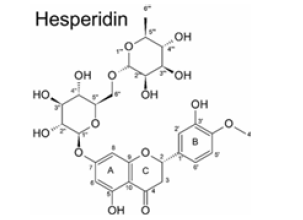
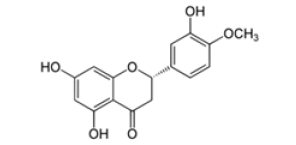
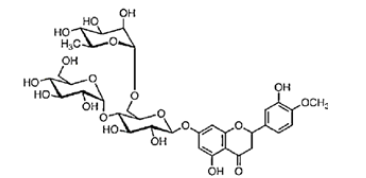
Hesperetin is the aglycon of Hesperidin, that means the two sugar moieties that are part of Hesperidin are absent (Figure 2 a and c). The aglycone is linked to the disaccharide rutinose that is formed by glucose and rhamnose. The sugar rest is attached to hesperitin at position-7 of the A ring (Figure 2 b). Hesperidin has a lot of alternative names: Cirantin, Hesperidosid, Hesperetin-7-O-rutinosid, Hesperetin-7-rhamnoglucosid, (2S)-7-((6-O-(6-Desoxy-α-L-manno¬pyranosyl)-β-D-glucopyranosyl)oxy)-2,3-dihydro-5-hydroxy-2-(3-hydroxy-4-methoxyphenyl)-4H-1-benzopyran-4-on. The sugar residues cause hesperidin to be more water-soluble than hesperitin. The chiral carbon in position 2 of hesperidin generates two diastereoisomers, 2R- and 2S-form. In nature, the predominant form in nature of hesperidin is the 2S-diastereoisomer. Lévèques has found a ratio of 8/92 for 2R-/2S-hesperidin diastereoisomers in fresh fruit products [6]. Alpha Glycosyl Hesperidin [AGH] is a modified form of hesperidin that comprises glucose and hesperidin (Figure 2d). It can be obtained by an enzymatic modification that involves adding an alpha-glucosyl saccharide to the hesperidin molecule. This alteration is often done by a saccharide-transferring enzyme that catalyses the transfer of the alpha-glucosyl group to hesperidin. This modification can enhance the solubility and bioavailability of hesperidin. In fact, it is 10’000 times more soluble in water than native hesperidin [7,8]. This is making it potentially more effective for certain applications, for example in supplements or pharmaceuticals. AGH is a powerful antioxidant with anti-inflammatory effects and is associated with various health benefits as it has many more of the same beneficial effects as hesperidin. Various study show the combined effect of green tea and α-glucosyl hesperidin in preventing obesity [9] (Figure 2a-2d).
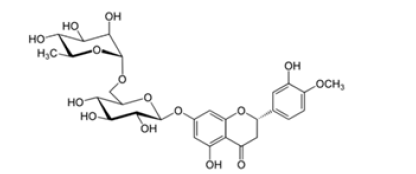
Figure 2: Chemical formula of hesperidin and hesperitin and alpha glycosylated hesperidin A: Chemical Formula Hesperidin (C28H34O15)
Biochemistry
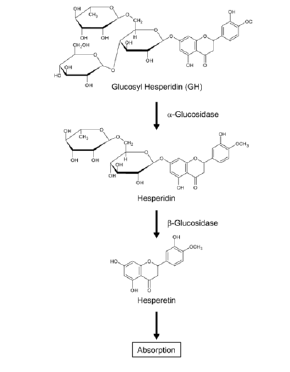
The metabolization of substances depends a lot on the structure of a molecule, how it can be digested by enzymes or pH and on the hydrophilicity or hydrophobicity. In hesperidin the sugar rests of the rutinose moiety are present. These are responsible that most of the ingested hesperidin is metabolized and absorbed in the colon since it cannot be hydrolyzed by the β-glucosidases of the small intestine [10,11]. Whereas in the colon, hesperidin is metabolized and hydrolyzed by the intestinal microbiota. Most of the molecules are deglycosylated to the aglycon hesperitin (Figure 3). In a next step hesperitin is absorbed by the colonocytes. In these cells hesperetin so-called phase II enzymes will continue the metabolization in the colon. These are the important enzymes UDP-glucuronosyl transferases and sulfotransferases. They catalyze the conjugation of hesperetin with glucuronic acid or sulphate and also other subsequent conjugation reactions [12]. The last step of hesperidin uptake into the body is the release of the conjugated hesperitin molecules into the bloodstream. Furthermore, a substantial proportion of hesperetin is directly additionally metabolized by the colon microbiota. From these reactions bioavailable forms result, as for example 3-(3’-Hydroxy-4’Methoxyphenyl) Propanoic Acid (HMPPA) and hippuric acid, 4-hydroxyhippuric acid and 3-Hydroxy-3-(3′-hydroxyphenyl) propanoic acid [1] [13] [12] (Figure 3).
Isolation of Hesperidin and Formation of Alpha- Glycosylated Hesperidin
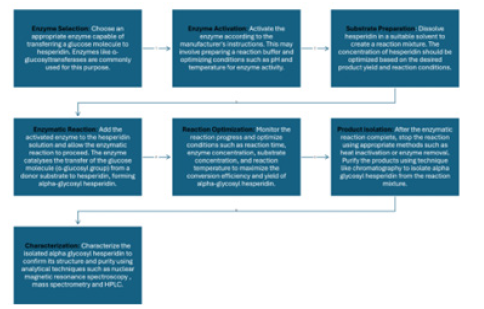
Dried orange peel is the raw material for the extraction of hesperidin from natural sources. Several different methods exist [14]. Often, petroleum ether is used to remove the essential oils and then to extract the glycoside hesperidin by methanol. Another method uses alkaline extraction of chopped orange peel and then an acidification of the extract. From this acidic extract hesperidin can be crystallized. Further extraction procedures include including maceration, extraction with ultrasound, high hydrostatic pressure and microwave assisted extraction, enzymatic processes and extraction supercritical fluids [15]. Because these methods are all not very economically friendly and use organic solvents a new green method with low energy consumption was invented by Stanisic, et al. [15] Alpha-glycosyl hesperidin is obtained from the purified natural substance hesperidin. The essential step to convert normal hesperidin to alpha-glycosyl hesperidin involves enzymatic processes that transfer a glucose molecule (α-glucosyl group) to hesperidin [7]. This modification enhances the solubility and bioavailability of hesperidin, making it more suitable for various applications. Figure 4 shows the sequential process to obtain alpha-glycosyl hesperidin and this general overview of the steps involved in the conversion will be described in the following. In a first step an appropriate enzyme that is capable of transferring a glucose molecule to hesperidin must be chosen. Enzymes like α-glucosyltransferases are commonly used for this catalysis. Next the appropriate enzyme conditions must be applied. Enzymes can be obtained commercially, and the process be done according to the manufacturer's instructions. This may involve preparing a reaction buffer and optimizing conditions such as pH and temperature for enzyme activity.
On the other hand the substrate preparation is necessary. Therefore, hesperidin is dissolved in a suitable solvent to create the reaction mixture. The concentration of hesperidin should be optimized based on the desired product yield and reaction conditions. Subsequently, the enzymatic reaction can start by adding the activated enzyme to the hesperidin solution and allowing the enzymatic reaction to proceed. The enzyme catalyses the transfer of the glucose molecule (α-glucosyl group) from a donor substrate to hesperidin, forming alpha-glycosyl hesperidin. To optimize the reaction the progress is monitored and improved conditions such as reaction time, enzyme concentration, substrate concentration, and reaction temperature are applied to maximize the conversion efficiency and the yield of alpha-glycosyl hesperidin. Finally, the product can be isolated once the enzymatic reaction is completed. To stop the reaction appropriate methods such as heat inactivation or enzyme removal are used. The product can be purified using techniques like chromatography to isolate alpha-glycosyl hesperidin from the reaction mixture. To be sure to have the right product and for quality control the isolated alpha-glycosyl hesperidin is characterized to confirm its structure and purity using analytical techniques such as nuclear magnetic resonance spectroscopy, mass spectrometry and HPLC. Specific details of the enzymatic conversion process may vary depending on factors such as choice of enzyme, reaction conditions and desired product characteristics. Consulting the relevant literature and working with experienced professionals and approved protocols will ensure successful conversion of normal hesperidin to alpha-glycosyl hesperidin (Figure 4).
Beneficial Effects on Human Health
Hesperidin is a citrus flavonoid that has various pharmacological effects, including antioxidative, anti-inflammatory, antidiabetic, antihypertensive, and antihyperlipidemic effects [16], compare Figure 1 It is used as a natural remedy for a number of health problems including allergies (hay fever, sinusitis), hemorrhoids, varicose veins, high blood pressure, high cholesterol levels, hot flashes, symptoms associated with menopausal changes and premenstrual syndrome. Many reports show that naturally occurring flavonoids exhibit a remarkable antiviral activity [17]. In an in-silico comparison study with ten phenolic antiviral agents against SARS-CoV-2, hesperidin showed the highest docking score [18]. The docking study with Covid-19 protease 6lu7 confirmed that hesperidin can form electrostatic interactions with THR 25, LEU 27, His 41and Gly143 residues while form pi-stacking interaction with TYR 118. Hesperidin was also predicted by screening in silico to interact with the site between SARS-CoV-2 Spike and ACE2 receptors. Thereby it blocks the entry of the virus into the human lung cells. For this reason, hesperidin could be a promising prophylactic drug against COVID-19 [18]. The ligand hesperidin causes conformational changes in the three‑dimensional structure of protein ACE2, which is confirmed by molecular docking and molecular dynamics studies [2]. The binding energy of bound structure of ACE2 and spike protein is modulated. It was suggested that due to presence of hesperidin, the bound structure of ACE2 and spike protein fragment becomes unstable. For this reason, the natural product hesperidin can show antiviral activity in SARS CoV2 infection. This was confirmed as hesperidin shows at least three independent effects: blocking of viral entry in human cells, inhibition of the production of inflammatory cytokines, activation of the MAPK pathway to increase cell immunity [19]. Also, in other study by Behloul, et al. hesperidin was identified as potent strong binder of the SARS-CoV-2 Receptor-Binding Domain (RBD) [20]. During the binding, the bioflavonoid forms a high number of hydrogen bonds and is interacting with 13 residues of the SARS-RBD (Figure 5).
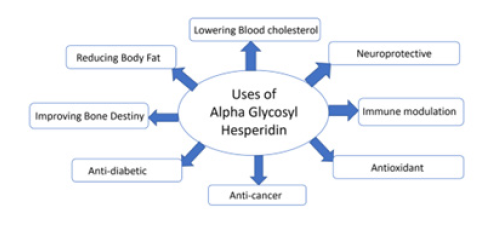
Figure 5: Uses of alpha glycosyl hesperidin. The benefits of hesperidin for human health are possible applications of alpha gycosyl hesperidin.
In vitro antiviral activity of hesperidin was additionally shown against hepatitis A virus as an example of RNA viruses. Flavonoids, which include hesperidin, can inhibit protein phosphorylation, which in turn leads to potent antiviral activity and infection prevention of other cells [18]. In addition to the antiviral activity also anti-bacterial activity of hesperidin is possible as plants can synthesize flavonoids in response to microbial infections and hence these molecules are very effective antimicrobial substances against a variety of microorganisms [21]. The actions of hesperidin are very wide. In vivo and in vitro studies were presented that show that hesperidin has significant antioxidant, anti-inflammatory, anticancer, and hepatoprotective activities as well as anti-diabetes, antiadipogenic, antihypertensive, and antimicrobial activities [21]. The antioxidant activity protects the body against reactive oxygen species, by acting as radicals’ scavengers. This was shown to be achieved by increasing the number of endogenous enzymes that are dependent on glutathione and which have antioxidant activity [1]. Anti-inflammatory activity for hesperidin has been shown, however the specific mechanism of action is not completely known and additional studies are required for elucidation of the molecular targets [22]. Further, hesperidin derivatives like alpha-glycosyl hesperidin have been studied for their immunomodulatory effects. They may help regulate immune responses, reduce inflammation, and enhance the immune response. Associated with its anti-oxidant and anti-inflammatory activities Hesperidin has also been shown to have anti-cancer activity and that it inhibits tumor promotion [23,24]. The substance interacts with several known cellular targets and inhibits cancer cell proliferation by inducing apoptosis and cell cycle arrest. A role for hesperidin to inhibit tumor cell metastasis, angiogenesis, and chemoresistance have been suggested.
Hesperidin has vasoprotective properties as is also thought to have beneficial effects on blood vessels [16]. This can be seen in correlation with the fact that: flavonoids seem to suppress LDL oxidation and inflammatory progression in the artery wall what leads to anti-artherosclerotic effects. In addition to its antioxidant properties, hesperidin and AGH may have neuroprotective effects. It has a role in protection of several neurodegenerative disorders like Parkinson’s and Alzheimer’s Disease [1]. It could help protect neurons from oxidative damage, inflammation, and neurodegenerative processes, potentially benefiting cognitive function and brain health. Hesperidin has impact on glucose and on lipid metabolism and thereby on fat loss [1]. The flavonoid can stimulate the release of Chole Cysto kinin (CCK), an appetite-regulating hormone, in enteroendocrine STC-1 cells, which is ultimately used to treat obesity by suppressing appetite [25]. Hesperidin reduces hepatic steatosis, adipose tissue, and liver weight, and decreases serum total cholesterol and Retinol-Binding Protein (RBP) 4 concentrations in high-fat diets [26]. Further it may improve hypercholesterolemia and fatty liver by inhibiting cholesterol synthesis and absorption, regulating RBP, C-FABP, and H-FABP mRNA expression. The flavonoid hesperidin increases the expression of messenger RNA by hormone-sensitive lipases and stimulates the breakdown of mature adipocytes. A significant down-regulation is seen of the expression of stearoyl-CoA desaturase, fatty-acid desaturase (FAT-6 and FAT-7) by hesperidin, and the reduction of the expression of other genes involved in lipid metabolism, including acetyl-CoA carboxylase-2(POD-2), mediator subunit-15(MDT-15), acyl-CoA synthetase-2(ACS-2) and 3-ketoacyl-CoA thiolase-1 (KAT-1), thereby reducing fat accumulation.
Bioavailability
The use of hesperidin as a drug or supplement is limited because it has low solubility in water and poor bioavailability after oral administration. There are several points that influence the extent to which the substance can be used by the body. As discussed above hesperidin is modified by enzymes of the microbiota. Therefore, its amount, concentration and composition influence the catalytic reactions and the performance of gut microbial enzymes. In the small intestine hesperidin is metabolized to hesperitin by removing the sugar moieties by two gastrointestinal micro-flora’s enzymes, alpha rhamnosidase and beta glucosidase [11]. Especially the performance of alpha rhamnosidase influences the amount of hesperidin that is deglycosylated to hesperitin. If, depending on the microbioty it is inactive, the large intestine stops producing hesperitin [27]. Consequently, as the gut microbes differ between individuals, there is a major variety because of that for the bioavailability of hesperidin. It might be that additionally to the activity of alpha-rhamnosidase other reactions also the bioavailability of hesperidin and results in biologic consequences [1]. Hesperidin can be taken as pure substance, as alpha-glycosylated form or within food where it naturally occurs like orange juice, lemon and citric fruits or the purified product added to food like yoghurt [28]. This might also affect how bioavailable it is in this situation. The phrase “bio accessibility” is used to indicate the portion of a substance which is liberated from its prevailing conditions into the intestinal system and then made accessible for the body. Upon a clinical trial it was concluded that the low level of fats in the yoghurt might contribute to its lack of impact on bioavailability of orange juice flavanones [28].
Furthermore, as stated above hesperidin and alpha-glycosylated hesperidin show extremely different solubilities. This affects how the substance is dissolved and this is a prerequisite for it to reach the circulation and the place in the body where it will have a physiological impact. A study showed that there is a relationship between maximal hesperidin concentration in serum and the concentration of soluble hesperidin in juice that is ingested [1]. The bioavailability of hesperidin, AGH and hesperidin were compared in rats [29]. It was observed that hesperidin was the major form of flavonoid present in plasma, although AGH had similar bioavailability, which suggests that AGH might exhibit good therapeutic potential. Meanwhile also other modifications of hesperidin based on nanotechnology have been designed to boost water solubilization, antioxidant properties, and oral uptake [1]. From an in vivo study with healthy individuals by Crescenti, et al. it was concluded that the bioavailability of 2S-hesperidin extract was higher compared to the standard mixture of 2S-/2R-hesperidin extract due to a greater formation of hesperidin catabolites [30]. Furthermore, the micronation process increased hesperidin bioavailability. Another randomized double-blind crossover trial found that The results of this study demonstrated that the bioavailability of hesperidin was modulated by enzymatic conversion to hesperetin-7glucoside, thus changing the absorption site from the colon to the small intestine [10].
Advantages of Alpha-Glycosylated Hesperidin (AGH)
As already stated, above AGH, a hesperidin derivative, which is synthesized from hesperidin and α-glucosyl saccharide by a transferring enzyme, is about 10’000 times more soluble in water and is absorbed more rapidly and efficiently than hesperidin [7,8]. AGH is readily hydrolysable by α-glucosidases in vivo, yielding hesperidin and D-glucose. Furthermore, hesperidin is hydrolyzed by β-glucosidases to form a hesperetin aglycone before its absorption [31,32] (Figure 2). Previous studies have shown that AGH ameliorates hypertriglyceridemia, hyperuricemia, and the progression of atherosclerosis in animal models and patients [33-36]. Additionally benefits of hesperidin for cutaneous function have been shown [37]. Therefore, AGH may be a new preventive and/or therapeutic agent for various diseases. One of the effects for AGH is lowering blood cholesterol [29]. Some studies suggest that hesperidin derivatives, including AGH, may help lower Low-Density Lipoprotein (LDL) cholesterol levels. This effect is beneficial for cardiovascular health, as high LDL cholesterol is a risk factor for heart disease. Additionally, an improvement of bone density has been observed upon administration of AGH. There is emerging research indicating that hesperidin and its derivatives could have positive effects on bone health [38]. These compounds may enhance bone density and strength, which is important for preventing osteoporosis and maintaining skeletal integrity. Diabetes is one of the diseases that occur mostly. Therefore, the observed anti-diabetic of AGH are of great interest. AGH has been studied for its potential anti-diabetic properties [39]. It may help improve insulin sensitivity, regulate blood glucose levels, and reduce complications associated with diabetes. Of special importance are also the anticancer properties of hesperidin and AGH that were explored in several studies [23]. These compounds exhibit anti-proliferative and apoptotic effects on cancer cells, making them potential candidates for cancer prevention and treatment.
Yoshida, and his team et al.,2020 evaluated the effects of long- and short-term intake of AGH on hyperglycemia and macrophage infiltration into the adipose tissue of High-Fat Diet (HFD)-fed mice [40]. Results show that long-term (11-week) consumption of AGH tends to reduce body weight and the fasting blood glucose concentration of the HFD-fed mice. It ameliorated glucose intolerance and insulin resistance, according to glucose and insulin tolerance tests. Additionally, it reduced HFD-induced macrophage infiltration into adipose tissue. These findings suggest that daily consumption of AGH may have preventive and/or therapeutic effects on obesity-related diseases, such as diabetes mellitus [40]. Additionally, AGH has been reported to reduce serum levels of triglyceride in animals’ hypertriglyceridemia [41-43]. The mechanism of the TG-lowering effect of G-hesperidin is thought to be the downregulation of the synthesis/secretion of very-low-density lipoprotein in hepatocytes, inhibition of lipogenesis, and induction of beta-oxidation of fatty acids in high-fat diet-fed rats [41,22]. In a study conducted by Nagayama, et al., (2022) scientists assessed whether continuous (14 days) intake of glucosyl hesperidin improves the aerobic exercise capacity of rats during long-term acute exercise [44]. Results have shown that glucosyl hesperidin intake did not improve the performance of high-intensity running (30m/min), but improvement was observed in low-intensity running (15m/min) (p<0.05). This study demonstrated that in sedentary rats, AGH intake increased β-oxidation and oxidative phosphorylation in the skeletal muscle (p<0.05 and p<0.01, respectively). AGH intake also created a metabolic state useful for long-term exercise. In conclusion, the continuous intake of AGH improved the aerobic exercise capacity of rats during long-term acute exercise. AGH has been investigated for its potential to reduce body fat accumulation. It may help modulate lipid metabolism, leading to decreased fat storage and potentially aiding in weight management. Several reports indicate that AGH significantly decreases body fat, but the underlying molecular mechanism remains unclear. A study by Nishikawa has shown that the antiobesity effects of AGH occur through the induced formation of brown-like adipocytes [45]. This study verified that dietary AGH induces brown-like adipocytes to form in mouse inguinal white adipose tissue (iWAT), thereby significantly decreasing the weight of White Adipose Tissue (WAT). Furthermore, dietary AGH significantly induced thermogenesis in iWAT. Dietary AGH also significantly suppressed high-fat-diet-induced WAT accumulation in mice, which may be mediated by brown-like adipocyte formation. These results indicate that dietary AGH induces increased energy expenditure by stimulating the formation of brown-like adipocytes [45].
Safety
Since centuries hesperidin has been used as a herbal drug in medicine. The Federation of American Societies of Experimental Biology (FASEB) confirmed the safety of hesperidin upon request of the FDA [46,47]. Toxicity studies have proven the high safety profile of hesperidin after oral intake. Results from oral toxicity studies showed the absence of adverse side effects after oral hesperidin ingestion of more than 2g/kg. Evaluation of the cytotoxicity of hesperidin gainst Vero cells showed for 50% cell cytotoxic concentration (CC50) = 620.8±34.6 µg/mL [18]. According to the oral toxicity study of hesperidin, it can be concluded that this phytochemical can be safely used in herbal formulations with its LD50 value higher than 2000mg/kg [2]. Because it doesn’t accumulate, hesperidin is regarded as safe when used as a supplement or nutritional additive, with nearly no negative impacts [48] Hajialyani, et al., (2019). Hesperidin can act as a blood thinner. It can slow down the process of blood coagulation. And for this reason, Hesperidin is not recommended to people suffering from bleeding disorders or taking anticoagulants. Hesperidin is also not recommended after undergoing surgery to prevent bleeding complications. Alpha glycosyl hesperidin, also known as α-glucosyl-hesperidin, is a derivative of hesperidin, a flavonoid found primarily in citrus fruits. It is produced by enzymatically combining natural hesperidin with a glucose molecule, resulting in a product with very high-water solubility and retaining the antioxidative activity of native hesperidin. The safety of alpha glycosyl hesperidin has been evaluated in the context of its use as a food ingredient. A GRAS (Generally Recognized as Safe) notice has been submitted to the FDA, which includes a comprehensive review of in vitro, animal, and human information related to the safety of natural hesperidin, hesperidin-containing complexes, and alpha glycosyl hesperidin. Caution is urged in its use in pregnancy or when breastfeeding, bleeding or blood clotting disorders, and allergy or intolerance to flavonoids. Hesperidin may interact with prescription drugs, such as blood thinners, blood pressure-lowering drugs (including calcium channel blockers), and certain anticancer drugs.
Commercial Aspects
Crucial factors for sourcing alpha-glycosyl hesperidin or any other specialized compound are enumerated in the following. A first point is to ensure that the alpha-glycosyl hesperidin meets high-quality standards. This includes factors such as purity, absence of contaminants, and adherence to established specifications. Next the purity of alpha-glycosyl hesperidin is essential. This refers to the percentage of the compound in its isolated form without impurities or contaminants. Higher purity levels typically indicate better quality and efficacy. Suppliers often provide Certificates of Analysis (COA) detailing the purity of their alpha-glycosyl hesperidin products. The purity of alpha-glycosyl hesperidin can significantly impact its effectiveness and safety. Look for suppliers that provide information on the purity level of their product, typically expressed as a high percentage (e.g., 98% purity). To assure regulatory compliance verify that the supplier complies with relevant regulatory requirements and industry standards. This includes certifications, such as Good Manufacturing Practices (GMP) for pharmaceuticals or ISO standards for quality management systems. A further necessary point for a good product is supplier reliability. Therefore, it is recommended to choose a reputable and reliable supplier with a track record of delivering high-quality products and excellent customer service. Check customer reviews, industry reputation, and the supplier's experience in providing alpha-glycosyl hesperidin. All people working with or using the product should look for suppliers that provide detailed product information, including technical specifications, Certificates of Analysis (COA), Safety Data Sheets (SDS), and product documentation. This information helps to assess the quality and suitability of the alpha-glycosyl hesperidin for the specific applications. It is helpful if suppliers offer technical support and expertise to assist in selecting the right alpha-glycosyl hesperidin for individual needs. This can include guidance on formulation, dosage, stability, and compatibility with other ingredients. Comparing prices and availabilities from multiple suppliers is essential to ensure that one is getting competitive pricing without compromising quality. Consideration of factors such as shipping costs, minimum order quantities, and lead times is additionally a quality point. By evaluating these factors and working with reliable suppliers that prioritize quality, compliance, and customer support, companies and individual customers can confidently source alpha-glycosyl hesperidin for all applications while ensuring product efficacy and safety.
Scope
The worldwide scope for alpha-glycosyl hesperidin is significant, driven by several factors that contribute to its potential in various industries and markets globally [49]. Worldwide there is a growing emphasis on health and wellness, with consumers seeking natural and functional substances to support their well-being. From this point of view alpha-glycosyl hesperidin, as a bioactive compound derived from citrus fruits, can be marketed as a natural and beneficial ingredient in dietary supplements, functional foods, and beverages. The nutraceutical and dietary supplement industry is experiencing steady growth worldwide, driven by increasing awareness of preventive healthcare and lifestyle-related health concerns. Alpha-glycosyl hesperidin offers potential health benefits such as antioxidant, anti-inflammatory, anti-obesity and cardiovascular support, making it a valuable ingredient for formulations in supplements targeting these areas. The demand for functional foods and beverages is expanding globally, fueled by consumer interest in products that offer added health benefits beyond basic nutrition. Alpha-glycosyl hesperidin can be incorporated into a variety of functional food and beverage products, including energy drinks, health juices, snacks, and meal replacements, to enhance their nutritional profile and appeal to health-conscious consumers. Hesperidin and its derivatives, including alpha-glycosyl hesperidin, have potential applications in the anti-aging and skincare industry [24]. These compounds possess antioxidant properties that can help protect the skin from oxidative damage and support overall skin health. Therefore, alpha-glycosyl hesperidin may find use in skincare formulations targeting anti-aging effects and skin protection.
Ongoing research and innovation in the field of bioactive compounds like alpha-glycosyl hesperidin contribute to its worldwide scope. Studies exploring its bioavailability, mechanisms of action, potential health benefits, and synergistic effects with other compounds drive interest from researchers, product developers, and manufacturers globally. Concerning regulatory considerations: Like in any market, regulatory compliance is essential for the global acceptance of alpha-glycosyl hesperidin products. Adhering to regulatory standards and obtaining necessary approvals in different countries and regions is crucial for ensuring product safety, quality, and market access.
Conflicts of Interest
None.
Acknowledgements
None.
References
- Ahmad A, Afzaal M, Saeed F, Ali SW, Imran A, et al. (2023) A comprehensive review of the therapeutic potential of citrus bioflavonoid hesperidin against lifestyle-related disorders. Cogent Food Agric 9(1): 2226427.
- Basu A, Sarkar A, Maulik U (2020) Molecular docking study of potential phytochemicals and their effects on the complex of SARS-CoV2 spike protein and human ACE2. Sci Rep 10(1): 17699.
- Bellavite P, Donzelli A (2020) Hesperidin and SARS-CoV-2: New Light on the Healthy Function of Citrus Fruits. Antioxidants 9(8): 742.
- Aturki Z, Brandi V, Sinibaldi M (2004) Separation of flavanone-7-O-glycoside diastereomers and analysis in citrus juices by multidimensional liquid chromatography coupled with mass spectrometry. J Agric Food Chem 52(17): 5303-8530.
- Gupta S, Singh V, Varadwaj PK, Chakravartty N, Katta AVSKM, et al. (2022) Secondary metabolites from spice and herbs as potential multitarget inhibitors of SARS-CoV-2 proteins. J Biomol Struct Dyn 40(5): 2264-2283.
- Lévèques A, Actis Goretta L, Rein MJ, Williamson G, Dionisi F, et al. (2012) UPLC-MS/MS quantification of total hesperetin and hesperetin enantiomers in biological matrices. J Pharm Biomed Anal 57: 1-6.
- Hijiya, Hiromi. Alpha-Glycosyl hesperidin, and its preparation and uses. EP0402049A2.
- Yamada M, Tanabe F, Arai N, Mitsuzumi H, Miwa Y, et al. (2006) Bioavailability of Glucosyl Hesperidin in Rats. Biosci Biotechnol Biochem 70(6): 1386-1394.
- Yoshitomi R, Yamamoto M, Kumazoe M, Fujimura Y, Yonekura M, et al. (2021) The combined effect of green tea and α-glucosyl hesperidin in preventing obesity: a randomized placebo-controlled clinical trial. Sci Rep 11(1): 19067.
- Nielsen ILF, Chee WSS, Poulsen L, Offord Cavin E, Rasmussen SE, et al. (2006) Bioavailability Is Improved by Enzymatic Modification of the Citrus Flavonoid Hesperidin in Humans: A Randomized, Double-Blind, Crossover Trial. J Nutr 136(2): 404-408.
- Borges G, Lean MEJ, Roberts SA, Crozier A (2013) Bioavailability of dietary (poly)phenols: a study with ileostomists to discriminate between absorption in small and large intestine. Food Funct 4(5): 754-762.
- Pereira Caro G, Ludwig IA, Polyviou T, Malkova D, García A, et al. (2016) Identification of Plasma and Urinary Metabolites and Catabolites Derived from Orange Juice (Poly)phenols: Analysis by High-Performance Liquid Chromatography-High-Resolution Mass Spectrometry. J Agric Food Chem 64(28): 5724-5735.
- Pereira Caro G, Fernández Quirós B, Ludwig IA, Pradas I, Crozier A, et al. (2018) Catabolism of citrus flavanones by the probiotics Bifidobacterium longum and Lactobacillus rhamnosus. Eur J Nutr 57(1): 231-242.
- Pyrzynska K (2022) Hesperidin: A Review on Extraction Methods, Stability and Biological Activities. Nutrients 14(12): 2387.
- Stanisic D, Liu LHB, Dos Santos RV, Costa AF, Durán N, et al. (2020) New Sustainable Process for Hesperidin Isolation and Anti-Ageing Effects of Hesperidin Nanocrystals. Molecules 25(19): 4534.
- Mas Capdevila A, Teichenne J, Domenech Coca C, Caimari A, Del Bas JM, et al. (2020) Effect of Hesperidin on Cardiovascular Disease Risk Factors: The Role of Intestinal Microbiota on Hesperidin Bioavailability. Nutrients 12(5): 1488.
- Adhikari B, Marasini BP, Rayamajhee B, Bhattarai BR, Lamichhane G, et al. (2021
) Potential roles of medicinal plants for the treatment of viral diseases focusing on COVID‐19: A review. Phytother Res 35(3): 1298-1312. - Attia GH, Moemen YS, Youns M, Ibrahim AM, Abdou R, et al. (2021) Antiviral zinc oxide nanoparticles mediated by hesperidin and in silico comparison study between antiviral phenolics as anti-SARS-CoV-2. Colloids Surf B Biointerfaces 203: 111724.
- Haggag YA, El-Ashmawy NE, Okasha KM (2020) Is hesperidin essential for prophylaxis and treatment of COVID-19 Infection? Med Hypotheses 144: 109957.
- Behloul N, Baha S, Guo Y, Yang Z, Shi R, Meng J (2021) In silico identification of strong binders of the SARS-CoV-2 receptor-binding domain. Eur J Pharmacol 890: 173701.
- Donia T, Dabbour NM, Loutfy SA (2023) Hesperidin: Advances on Resources, Biosynthesis Pathway, Bioavailability, Bioactivity, and Pharmacology. In: Xiao J, Herausgeber. Handbook of Dietary Flavonoids [Internet]. Cham: Springer International Publishing; 1-55.:
- Tejada S, Pinya S, Martorell M, Capó X, Tur JA, et al. (2018) Potential Anti-inflammatory Effects of Hesperidin from the Genus Citrus. Curr Med Chem 25(37): 4929-4945.
- Aggarwal V, Tuli HS, Thakral F, Singhal P, Aggarwal D, et al. (2020) Molecular mechanisms of action of hesperidin in cancer: Recent trends and advancements. Exp Biol Med 245(5): 486-497.
- Rodrigues CV, Pintado M (2024) Hesperidin from Orange Peel as a Promising Skincare Bioactive: An Overview. Int J Mol Sci 25(3): 1890.
- Kim HY, Park M, Kim K, Lee YM, Rhyu MR (2013) Hesperetin Stimulates Cholecystokinin Secretion in Enteroendocrine STC-1 Cells. Biomol Ther 21(2): 121-125.
- Wang X, Hasegawa J, Kitamura Y, Wang Z, Matsuda A, et al. (2011) Effects of hesperidin on the progression of hypercholesterolemia and fatty liver induced by high-cholesterol diet in rats. J Pharmacol Sci 117(3): 129-138.
- Amaretti A, Raimondi S, Leonardi A, Quartieri A, Rossi M (2015) Hydrolysis of the Rutinose-Conjugates Flavonoids Rutin and Hesperidin by the Gut Microbiota and Bifidobacteria. Nutrients 7(4): 2788-2800.
- Mullen W, Archeveque MA, Edwards CA, Matsumoto H, Crozier A (2008) Bioavailability and metabolism of orange juice flavanones in humans: impact of a full-fat yogurt. J Agric Food Chem 56(23): 11157–11164.
- Tomazini Gonçalves T, Lazaro C, De Mateo F, Campos M, Mezencio J, et al. (2018) Effects of glucosyl-hesperidin and physical training on body weight, plasma lipids, oxidative status and vascular reactivity of rats fed with high-fat diet. Diabetes Metab Syndr Obes 11: 321-332.
- Crescenti A, Caimari A, Alcaide Hidalgo JM, Mariné Casadó R, Valls RM, et al. (2022) Hesperidin Bioavailability Is Increased by the Presence of 2S-Diastereoisomer and Micronization-A Randomized, Crossover and Double-Blind Clinical Trial. Nutrients 14(12): 2481.
- Matsumoto H, Ikoma Y, Sugiura M, Yano M, Hasegawa Y (2004) Identification and quantification of the conjugated metabolites derived from orally administered hesperidin in rat plasma. J Agric Food Chem 52(21): 6653-6659.
- Ameer B, Weintraub RA, Johnson JV, Yost RA, Rouseff RL (1996) Flavanone absorption after naringin, hesperidin, and citrus administration. Clin Pharmacol Ther 60(1): 34-40.
- Miwa Y, Yamada M, Sunayama T, Mitsuzumi H, Tsuzaki Y, et al. (2004) Effects of glucosyl hesperidin on serum lipids in hyperlipidemic subjects: preferential reduction in elevated serum triglyceride level. J Nutr Sci Vitaminol 50(3): 211-218.
- Miwa Y, Mitsuzumi H, Sunayama T, Yamada M, Okada K, et al. (2005) Glucosyl hesperidin lowers serum triglyceride level in hypertriglyceridemic subjects through the improvement of very low-density lipoprotein metabolic abnormality. J Nutr Sci Vitaminol 51(6): 460-470.
- Ota Kontani A, Hirata H, Ogura M, Tsuchiya Y, Harada Shiba M (2020) Comprehensive analysis of mechanism underlying hypouricemic effect of glucosyl hesperidin. Biochem Biophys Res Commun 521(4): 861-867.
- Sugasawa N, Katagi A, Kurobe H, Nakayama T, Nishio C, et al. (2019) Inhibition of Atherosclerotic Plaque Development by Oral Administration of α-Glucosyl Hesperidin and Water-Dispersible Hesperetin in Apolipoprotein E Knockout Mice. J Am Coll Nutr 38(1): 15-22.
- Man MQ, Yang B, Elias PM (2019) Benefits of Hesperidin for Cutaneous Functions. Evid Based Complement Alternat Med 2019: 2676307.
- Ortiz ADC, Fideles SOM, Reis CHB, Bellini MZ, Pereira EDSBM, et al. (2022) Therapeutic Effects of Citrus Flavonoids Neohesperidin, Hesperidin and Its Aglycone, Hesperetin on Bone Health. Biomolecules 12(5): 626.
- Xiong H, Wang J, Ran Q, Lou G, Peng C, et al. (2019) Hesperidin: A Therapeutic Agent For Obesity. Drug Des Devel Ther 13: 3855-3866.
- Yoshida H, Tsuhako R, Sugita C, Kurokawa M (2021) Glucosyl Hesperidin Has an Anti-diabetic Effect in High-Fat Diet-Induced Obese Mice. Biol Pharm Bull 44(3): 422-430.
- Miwa Y, Mitsuzumi H, Yamada M, Arai N, Tanabe F, et al. (2006) Suppression of Apolipoprotein B Secretion from HepG2 Cells by Glucosyl Hesperidin. J Nutr Sci Vitaminol 52(3): 223-231.
- Akiyama S, Katsumata S ichi, Suzuki K, Nakaya Y, Ishimi Y, et al. (2009) Hypoglycemic and hypolipidemic effects of hesperidin and cyclodextrin-clathrated hesperetin in Goto-Kakizaki rats with type 2 diabetes. Biosci Biotechnol Biochem 73(12): 2779-2782.
- Chiba H, Uehara M, Wu J, Wang X, Masuyama R, et al. (2003) Hesperidin, a citrus flavonoid, inhibits bone loss and decreases serum and hepatic lipids in ovariectomized mice. J Nutr 133(6): 1892–1897.
- Nagayama S, Aoki K, Komine S, Arai N, Endo S, et al. (2023) Improvement of low‐intensity long‐time running performance in rats by intake of glucosyl hesperidin. Physiol Rep 11(2): 15413.
- Nishikawa S, Hyodo T, Nagao T, Nakanishi A, Tandia M, et al. (2019) α-Monoglucosyl Hesperidin but Not Hesperidin Induces Brown-Like Adipocyte Formation and Suppresses White Adipose Tissue Accumulation in Mice. J Agric Food Chem 67(7): 1948–54.
- Yoko Nagasako-Akazome. Safety of High and Long-term Intake of Polyphenols. Polyphenols Hum Health Dis. Chapter 58:747–56.
- (2019) Generally Recognized As Safe Notice for Glucosyl Hesperidin (GH).
- Hajialyani M, Hosein Farzaei M, Echeverría J, Nabavi SM, Uriarte E, et al. (2019) Hesperidin as a Neuroprotective Agent: A Review of Animal and Clinical Evidence. Molecules 2019;24(3): 648.
- (2023) Global Alpha Glucosyl Hesperidin Market by Type (Purity: 98-99%, Purity: ≥99%), By Application (Skin Care Products, Hair Care Products), By Geographic Scope and Forecast [Internet].




 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.