Mini Review 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Medical Theory: Why Does Progesterone Not Work After a Traumatic Brain Injury in Humans?
*Corresponding author:Eduardo de Oliveira Duque Estrada, Faculdade de Medicina do ABC (FMABC), Santo André, São Paulo, Brasil
Received: September 18, 2019;Published: September 30, 2019
DOI: 10.34297/AJBSR.2019.05.000929
Introduction
The evidences suggest progesterone has a neuroprotective action in preclinical studies [1], but in some recent clinical studies, this hormone did not benefit people involved in traumatic brain injury. [2,3] So, why progesterone does not work correctly after a traumatic brain injury in humans?
Traumatic Brain Injury
Traumatic brain injury (TBI) is a great public health problem. More than 50 million head injuries occur each year worldwide. [4] In 2010, a study pointed to ECT in 89% of trauma-related deaths in low and middle-income countries. And according to the same study in the spectrum of trauma-related injuries, traumatic brain injury was considered a major cause of death and disability. [5] In the United States, traumatic brain injury is serious long-term morbidity. Between 1.6 and 3.8 million sports-related injuries are reported yearly, while an estimate of 5.3 million people lives with long-term cognitive and psychological impairment. [6] The pathophysiology of traumatic brain injury is complex and involves primary and secondary mechanisms of injury. The primary lesion induces biochemical and cellular changes leading to lesions that evolve over hours, days, months or even years with definitive neuronal damage and early death. [7] These traumatic injuries are caused by direct and indirect biomechanical forces and result in an intense neurometabolic event in the brain. [8] The main process after traumatic brain injury affecting the results is acute inflammation, involving mediators such as TNF-α, IL-6, IL-8, and IL-10. [9] After brain injury, including traumatic brain injury, GFAP expression increases in astrocytes which in turn release inflammatory cytokines and disrupt functional recovery. [10,11]
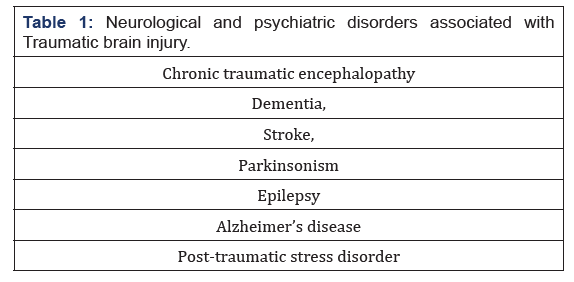
Multiple lesions in trauma are commonly associated with hypotension, hypoxia, hyperpyrexia, and coagulopathy that can adversely affect the brain and have long-term consequences. In addition, a growing body of evidence indicates that traumatic brain injury patients are susceptible to other neurological and psychiatric disorders (Table 1). In many cases, the manifestation of these associated conditions occurs several years after the injury. The disease’s level of complexity limits the development of strategies and effective treatment models to predict the outcome of patients with traumatic brain injury [12-19].
Progesterone
Some clinical studies have indicated that progesterone is a potent anti-inflammatory agent that modulates microglial activity after brain injury [20-22]. Perilesional reactive gliosis decreased broadly after progesterone administration (GFAP and Iba1 immunolabeling), with previous studies demonstrating a progesterone-induced reduction in GFAP expression after traumatic brain injury [23]. Progesterone is a pleiotropic hormone that uses multiple signaling pathways, including the regulation of gene expression after binding to intracellular progesterone receptors. In addition to the classical receptor (PR), progesterone also interacts with other signal transduction mechanisms, such as the s1 receptor (Ps1) that is a competitive inhibitor by which it can reduce N-methyl-D-aspartate (NMDA) glutamate). [24,25] Other studies have reported the attenuating effects of progesterone on edema in various animal models of brain injury. [26,27] An inverse correlation between serum progesterone level and degree of edema has been reported. [28] A study, evaluating progesterone versus dexamethasone to reduce neurosurgical brain edema, demonstrated the safety of progesterone use and equal efficacy in cerebral edema and attenuation of acute cellular inflammatory responses. [29]. Some authors evaluated edema and brain water concentration (BWC), with progesterone analogous in guinea pigs, and reported the same result. [30, 31] Positive results from preclinical studies and phase II studies [33-35] were contradicted in phase III studies, where progesterone failed to prove its efficacy in traumatic brain injury in humans. [36, 37].
Pharmacology of Progesterone
When we evaluated the pharmacological profile of the presentations tested for clinical efficacy, most of the available drugs are poorly acidic or poorly basic and have low aqueous solubility. These poorly soluble drugs in water evolve with low absorption rates, which can result in low tissue bioavailability, being critical for their rapid and effective action, as in brain injury. [38]. The solubility dilemma is a major challenge for its formulation. [38-40] Solubility is an important parameter in obtaining drugs with the ability to achieve the desired concentration in the brain and other tissues. Due to progesterone’s plasma half-life of only 25 min, it is necessary to take it to the brain rapidly, which in practical conditions would require immediate continuous IV post-trauma treatment. [41] It can be stated that the dripping or use of multiple injections in a lipid-based vehicle delays the release into the systemic circulation and results in a consequent reduction of the expected protective properties of progesterone in the acute phase of traumatic brain injury. This feature is important and makes it difficult to “replicate” the compelling results obtained in pre-clinical trials in guinea pigs. [42-44]
Conclusion
After two decades of preclinical research on the use of progesterone, it can be said that it is more than a simple reproductive hormone. [45,46] More than 100 articles on beneficial effects of progesterone have been published in four different species, including humans and 22 different models of traumatic brain injury lesions. [45-48] Although there are questions about the design and execution of clinical trials, it is important to realize that progesterone itself is not adequately soluble, thus configuring a challenge for its emergency use. [1,19,49-51] The drug solubility and the time necessary to reach concentrations in damaged brain tissue may contribute to the variability of response in patients and mask potential benefits in the injury’s acute phase. We consider Hill’s sentence: “All scientific work is incomplete - whether it be observational or experimental. All scientific work is liable to be upset or modified by advancing knowledge. That does not confer upon us a freedom to ignore the knowledge we already have, or to postpone the action that it appears to demand at a given time.” [52].
References
- Schumacher M, Denier, C, Oudinet JP, Adams D, Guennoun R (2016) Progesterone neuroprotection: the background of clinical trial failure. J Steroid Biochem Mol Biol 160: 53-66.
- Skolnick BE, Mass AI, Narayan RK, Van der Hoop RG, Mac Allister T, et al. (2014) A clinical trial of progesterone for severe traumatic brain injury. NEJM 25: 371(26):2467-2476.
- Wright DW, Yeatts SD, Silbergleit R, Palesch YY, Hertberg VS, et al. (2014) Very early administration of progesterone for acute traumatic brain injury. NEJM 371(26): 2457-66.
- Feigin VL, Theadom A, Barker Collo S, Starkey NJ, Mc Pherson K, et al. (2013) Incidence of traumatic brain injury in New Zealand: a population-based study. Lancet Neurol 12(1): 53-64.
- Rubiano AM, Carney N, Chesnut R, Puyana JC (2015) Global neurotrauma research challenges and opportunities. Nature 527(7578): S193-S197.
- Selassie AW, Eduard Zaloshnja, Jean Langlois, Ted Miller, Paul Jones, et al. (2008) Incidence of long-term disability following traumatic brain injury hospitalization, United States, 2003. J. Head Trauma Rehabil 23(2): 123-131.
- Cox CS (2018) Cellular therapy for traumatic neurological injury. Pediatr Res 83(1-2): 325-332.
- Giza CC. and Hovda DA (2014) The new neurometabolic cascade of concussion. Neurosurgery 75(Suppl.4): S24-S33.
- Woodcock T, Morganti Kossmann MC. (2013). The role of markers of inflammation in traumatic brain injury. Front Neurol 4:18.
- Hoane MR, Akstulewicz SL, Toppen J (2003) Treatment with vitamin B3 improves functional recovery and reduces GFAP expression following traumatic brain injury in rats. J Neurotrauma 20(11): 1189-99
- Hoane MR, Knotts AA, Akstulewicz SL, Aquilano M, Means LW (2003) The behavioral effects of magnesium therapy on recovery of function following bilateral anterior medial cortex lesions in the rat. Brain Res Bull. 60(1-2): 105-14.
- Stern RA, Riley DO, Daneshvar DH, Nowinski CJ, Cantu RC, et al. (2011) Long-term consequences of repetitive brain trauma: chronic traumatic encephalopathy. PM R 3(10 Suppl 2): S460-S467.
- Van Den Heuvel C, Thornton E, Vink R (2007) Traumatic brain injury and Alzheimer’s disease: a review. Prog Brain Res 161: 303-316.
- Levin HS, Diaz Arrastia RR (2015) Diagnosis, prognosis, and clinical management of mild traumatic brain injury. Lancet Neurol 14(5): 506-517.
- Fleminger S, Oliver DL, Lovestone S, Rabe Hesketh S, Giora A (2003) Head injury as a risk factor for Alzheimer’s disease: the evidence 10 years on; a partial replication. J Neurol Neurosurg Psychiatry 74(7): 857-862.
- Burke JF, Stulc JL, Skolarus LE, Sears ED, Zahuranec DB, et al. (2013) Traumatic brain injury may be an independent risk factor for stroke. Neurology 81(1): 33-39.
- Jafari S, Etminan M, Aminzadeh F, Samii A (2013) Head injury and risk of Parkinson disease: a systematic review and meta-analysis. Mov Disord 28(9): 1222-1229.
- Crane PK, Gibbons LE, Dams O Connork, Trittschuh E, Leverenz JB, et al. (2016) Association of traumatic brain injury with latelife neurodegenerative conditions and neuropathologic findings. JAMA Neurol 73(9): 1062-1069.
- Stein DG, Howard RB, 2017. Why did the phase III clinical trials for progesterone in tbi fail? An analysis of three potentially critical factors. In: Heidenreich, K.A. (Ed.), New Therapeutics for Traumatic Bbrain Injury: Prevention of Secondary Brain Damage and Enhancement of Repair and Regeneration. Elsevier, San Diego, CA, pp. 3-18.
- Stein DG (2007) Progesterone exerts neuroprotective effects after brain injury. Brain Res Rev 57(2): 386-97.
- Schumacher M, Denier C, Oudinet JP, Adams D, Guennoun R (2016) Progesterone neuroprotection: the background of clinical trial failure. J Steroid Biochem Mol Biol 160: 53-66.
- Drew PD, Chavis JA (2000) Female sex steroids: effects upon microglial cell activation. J Neuroimmunol 111(1-2): 77-85.
- Liu F, Liao F, Li W, Han Y, Liao D (2004) Progesterone alters Nogo-A, GFAP and GAP-43 expression in a rat model of traumatic brain injury. Mol Med Rep 9(4): 1225-1231.
- Hanner M, F F Moebius, A Flandorfer, H G Knaus, J Striessnig, et al. (1996) Purification, molecular cloning, and expression of the mammalian sigma1-binding site. Proc Natl Acad Sci USA 93(15): 8072-8077.
- Bergeron R, de Montigny C, Debonnel G (1999) Pregnancy reduces brain sigma receptor function. Br J Pharmacol 127(8): 1769-1776.
- Guo Q, Sayeed I, Baronne LM, Hoffman SW, Guennoun R, et al. (2006) Progesterone administration modulates AQP4 expression and edema after traumatic brain injury in male rats. Exp Neurol 198(2): 469-78.
- Maghool F, Khaksari M, Siahposht Khachki A (2013) Differences in brain edema and intracranial pressure following traumatic brain injury across the estrous cycle: involvement of female sex steroid hormones. Brain Res 1497: 61-72.
- Wright DW, Bauer ME, Hoffman SW, Stein DG (2001) Serum progesterone levels correlate with decreased cerebral edema after traumatic brain injury in male rats. J Neurotrauma 18(9): 901-909
- Xu FF, Sun S, Ho AS, Lee D, Kiang KM, et. al. (2014) Effects of progesterone vs. dexamethasone on brain oedema and inflammatory responses following experimental brain resection. Brain Inj 28(12): 1594-1601.
- MacNevin CJ, Atif F, Sayeed I, Stein DG, Liotta DC (2009) Development and screening of water-soluble analogues of progesterone and allopregnanolone in models of brain injury. J Med Chem 52(19): 6012-6023.
- Guthrie DB, Stein DG, Liotta DC, Lockwood MA, Sayeed I, et al. (2012) Water-soluble progesterone analogues are effective, injectable treatments in animal models of traumatic brain injury. ACS Med Chem Lett 3(5): 362-366.
- Vandromme M, Melton SM, Kerby JD (2008) Progesterone in traumatic brain injury: time to move on to phase III trials. Crit Care 12(3):153.
- Xiao G, Wei J, Yan W, Wang W, Lu, Z, (2008) Improved outcomes from the administration of progesterone for patients with acute severe traumatic brain injury: a randomized controlled trial. Crit Care 12(2): R61.
- Junpeng M, Huang S, Qin S, You C, Zeng Y (2011) Progesterone for acute traumatic brain injury. Cochrane Database Syst. Rev 12: CD008409.
- Skolnick BE, Maas AI, Narayan RK, van der Hoop RG, MacAllister T, et al. (2014) A clinical trial of progesterone for severe Traumatic Brain Injury. NEJM 371(26): 2467-2476.
- Wright DW, Yeatts SD, Silbergleit R, Palesch YY, Hertzberg VS, et al. (2014) Very early administration of progesterone for acute traumatic brain injury. NEJM 371(26): 2457-2466.
- Kalepu S, Nekkanti V (2015) Insoluble drug delivery strategies: review of recent advances and business prospects. Acta Pharm Sin B 5(5): 442-53.
- Spellacy WN (1969) A review of carbohydrate metabolism and the oral contraceptives. Am J Obstet Gynecol 104(3): 448-460.
- Savjani KT, Gajjar AK, Savjani JK (2012) Drug solubility: importance and enhancement techniques. ISRN Pharm 195727: Pp.10.
- Thijssen JH, Zander J (1966) Progesterone-4-14c and its metabolites in the blood after intravenous injection into women. Acta Endocrinol (Copenh) 51(4): 563-577.
- Whitehead MI, Hillard TC, Crook D (1990) The role and use of progestogens. Obstet Gynecol 75 (4 suppl): 59S-76S.
- Misra A, Ganesh S, Shahiwala, A, Shah SP (2003) Drug delivery to the central nervous system: a review. J Pharm Pharm Sci 6(2): 252-273.
- Rankovic Z (2015) Cns drug design: balancing physicochemical properties for optimal brain exposure. J Med Chem 58(6): 2584-2608.
- Stein DG, Hurn PD (2009) Effects of sex steroids on damaged neural systems. In Pfaff DW, Arnold AP, Etgen AM, et al., editors, Hormones, brain and behavior. Pp. 2223-2258.
- Sayeed I, Stein DG (2009) Progesterone as a neuroprotective factor in traumatic and ischemic brain injury. Prog Brain Res 175: 219-237.
- Vink R, Nimmo AJ (2009) Multifunctional drugs for head injury. Neurotherapeutics 6(1): 28-42.
- Schumacher M, Guennoun R, Stein DG, De Nicola AF (2007) Progesterone: therapeutic opportunities for neuroprotection and myelin repair. Pharmacol Ther 116 (1): 77-106.
- Stein DG (2015) Embracing failure: what the phase III progesterone studies can teach about tbi clinical trials. Brain Inj 29(11): 1259-1272.
- Howard RB, Sayeed I, Stein DG (2017) Suboptimal dosing parameters as possible factors in the negative phase III clinical trials of progesterone in TBI. J Neurotrauma 34(11): 1915-1918.
- Sayeed I, Atif F, Espinosa Garcia C, Wali B, Turan N, et al. (2018) Prehospital intubation: further confounders in trial results. Prehosp Emerg Care 22(4): 535-536.
- Stein, DG, Sayeed I, Espinosa Garcia C, Atif F, Sergeeva EG (2017) Goldstein et al.'s Secondary Analysis of Progesterone Clinical Trial for Traumatic Brain Injury Can Only Reflect the Same Trial Design Flaws: A Response to "Very Early Administration of Progesterone Does Not Improve Neuropsychological Outcomes in Subjects with Moderate to Severe Traumatic Brain Injury" 2017 J. Neurotrauma 34(13): 2192-2193.
- Hill AB (2015) The environment and disease: Association or causation? J R Soc Med 108 (1): 32-7.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.